top of page
Cardiovascular System
Renovascular hypertension
Core Principle of Renovascular Hypertension
🧷
Renovascular hypertension occurs when reduced renal perfusion activates the renin-angiotensin-aldosterone system (RAAS), leading to systemic hypertension.
🧷
The kidney interprets decreased blood flow as systemic hypotension and compensates by increasing renin release, which drives angiotensin II production → vasoconstriction and aldosterone secretion → sodium retention and volume expansion.
🧷
This is a secondary form of hypertension with an identifiable and potentially correctable cause — renal artery stenosis.
🧷
The two main etiologies are atherosclerotic disease (90% of cases) and fibromuscular dysplasia (10% of cases), each with distinct patient demographics and angiographic patterns.

Pathophysiology of RAAS Activation
📍
Renal artery stenosis → decreased afferent arteriolar pressure → juxtaglomerular cells sense hypoperfusion → renin release.
📍
Renin converts angiotensinogen to angiotensin I → ACE converts angiotensin I to angiotensin II in pulmonary circulation.
📍
Angiotensin II effects: systemic vasoconstriction, aldosterone release from zona glomerulosa, ADH release, increased sympathetic activity, and direct sodium reabsorption in proximal tubule.
📍
Aldosterone → principal cell Na⁺ reabsorption and K⁺ secretion → volume expansion and hypokalemia.
📍
Board pearl: The stenotic kidney has high renin production while the contralateral kidney suppresses renin due to systemic hypertension.

Atherosclerotic Renal Artery Stenosis
🔹
Accounts for 90% of renovascular hypertension cases, typically affecting older patients (>55 years) with cardiovascular risk factors.
🔹
Atherosclerotic plaques develop at the ostium or proximal third of the renal artery — same pathophysiology as coronary and carotid disease.
🔹
Often bilateral but asymmetric; associated with aortic atherosclerosis, coronary artery disease, peripheral vascular disease, and carotid stenosis.
🔹
Progressive stenosis can lead to ischemic nephropathy — irreversible parenchymal damage from chronic hypoperfusion.
🔹
Board clue: Elderly patient with new or worsening hypertension + cardiovascular risk factors + abdominal bruit = atherosclerotic RAS.

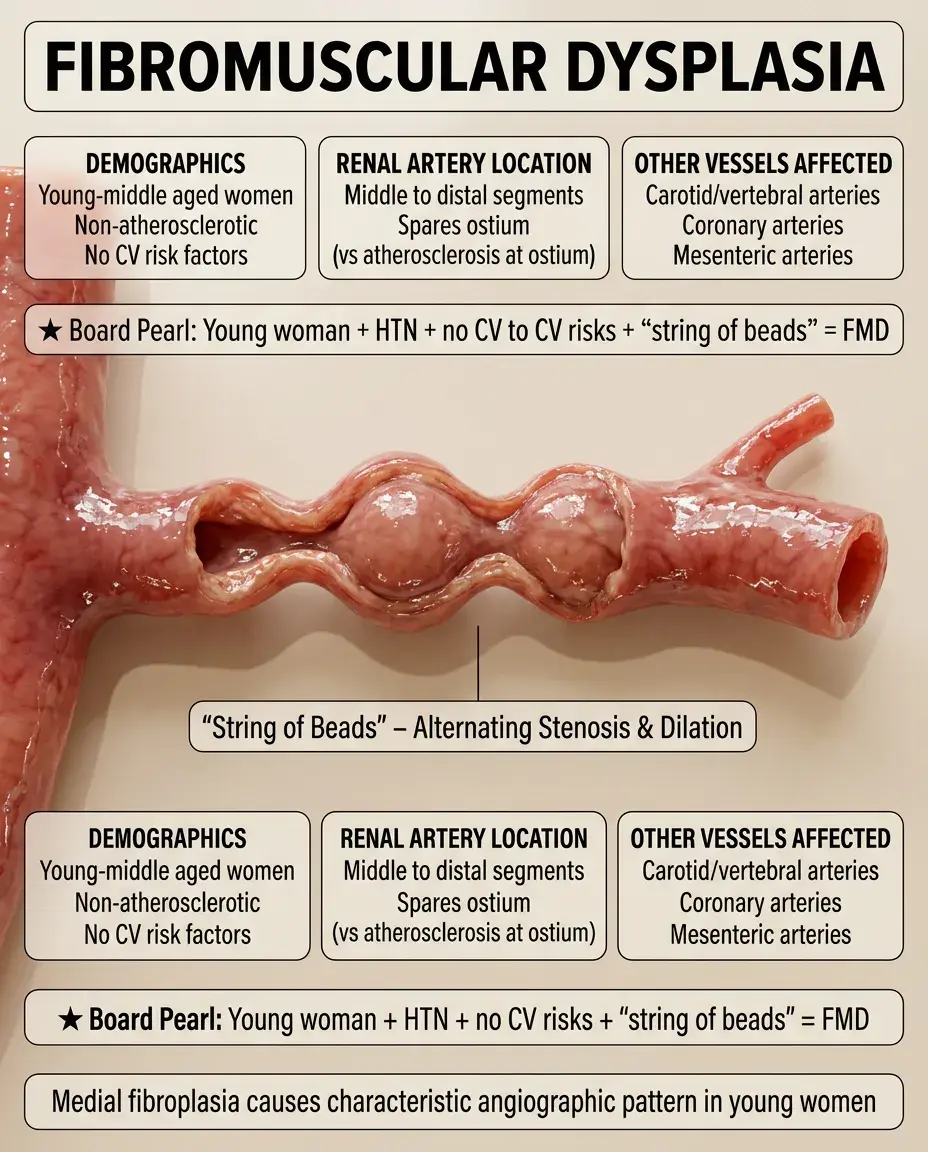
Fibromuscular Dysplasia
⭐
Non-atherosclerotic, non-inflammatory vascular disease causing arterial stenosis, predominantly affecting young to middle-aged women.
⭐
Medial fibroplasia (most common subtype) creates the classic "string of beads" appearance on angiography — alternating areas of stenosis and dilation.
⭐
Typically affects the middle to distal portions of the renal artery, sparing the ostium (unlike atherosclerosis).
⭐
Can affect other vascular beds: carotid and vertebral arteries (causing headaches, pulsatile tinnitus, stroke), coronary arteries, mesenteric arteries.
⭐
Board pearl: Young woman with hypertension + no cardiovascular risk factors + "string of beads" on imaging = fibromuscular dysplasia.
Clinical Presentation Red Flags
✅
Onset of hypertension before age 30 (suggests FMD) or after age 55 (suggests atherosclerotic disease).
✅
Resistant hypertension despite three antihypertensive medications including a diuretic.
✅
Abrupt worsening of previously well-controlled hypertension.
✅
Hypertension with unexplained hypokalemia (from secondary hyperaldosteronism).
✅
Acute kidney injury precipitated by ACE inhibitor or ARB initiation — suggests bilateral RAS or unilateral RAS in a solitary kidney.
✅
Asymmetric kidney size on imaging (>1.5 cm difference) — the ischemic kidney undergoes atrophy.

Physical Examination Findings
🧠
Abdominal bruit: systolic-diastolic bruit heard over the flank or epigastrium, present in 40% of cases. High specificity but low sensitivity.
🧠
Findings of severe hypertension: S4 gallop, hypertensive retinopathy (AV nicking, copper wire changes, flame hemorrhages, papilledema).
🧠
Evidence of atherosclerosis elsewhere: carotid bruits, diminished peripheral pulses, femoral bruits.
🧠
Flash pulmonary edema in bilateral RAS — sudden LV failure from acute afterload increase.
🧠
Board pearl: An abdominal bruit in a hypertensive patient should always prompt evaluation for renovascular disease.

Laboratory Findings and RAAS Profile
⚡
Hypokalemia from secondary hyperaldosteronism — but can be masked by concurrent diuretic use.
⚡
Elevated serum creatinine, especially if bilateral disease or solitary kidney affected.
⚡
Plasma renin activity (PRA): elevated in the affected kidney's renal vein, suppressed on the contralateral side. Lateralization ratio >1.5 is significant.
⚡
Captopril stimulation test: exaggerated rise in PRA after captopril administration due to interruption of angiotensin II negative feedback.
⚡
Board distinction: Primary hyperaldosteronism has high aldosterone but LOW renin; renovascular hypertension has high aldosterone AND high renin.

Diagnostic Imaging Modalities
📌
Duplex ultrasonography: non-invasive screening test. Peak systolic velocity >200 cm/s suggests significant stenosis. Limited by operator dependence and patient body habitus.
📌
CT angiography: excellent sensitivity and specificity for detecting stenosis. Provides anatomic detail but requires iodinated contrast — risky in renal insufficiency.
📌
MR angiography: avoids radiation and can use gadolinium (though risk of nephrogenic systemic fibrosis in severe CKD). Less spatial resolution than CTA.
📌
Renal angiography: gold standard for diagnosis and allows simultaneous intervention. Reserved for cases planned for revascularization.
📌
Board pearl: In patients with renal insufficiency, start with duplex ultrasound to avoid contrast nephropathy.

ACE Inhibitor Challenge and Renal Function
📣
ACE inhibitors/ARBs can precipitate acute kidney injury in bilateral RAS or unilateral RAS with solitary kidney.
📣
Mechanism: Angiotensin II normally maintains GFR by preferential efferent arteriolar constriction. Blocking this in the setting of decreased afferent flow → GFR collapse.
📣
A rise in creatinine >30% after starting ACE inhibitor suggests hemodynamically significant RAS.
📣
Captopril renal scintigraphy: decreased uptake or delayed excretion after captopril indicates functional significance of stenosis.
📣
Board pearl: New AKI after starting lisinopril in a patient with atherosclerotic disease → suspect bilateral renal artery stenosis.

Ischemic Nephropathy Pathophysiology
🔸
Chronic renal hypoperfusion from progressive RAS leads to irreversible parenchymal injury — tubular atrophy, interstitial fibrosis, glomerulosclerosis.
🔸
The kidney shrinks and becomes echogenic on ultrasound. Function does not recover even after revascularization once atrophy develops.
🔸
Bilateral disease or unilateral disease in a solitary kidney can progress to end-stage renal disease.
🔸
Atheroembolic disease during intervention can accelerate renal failure — cholesterol crystals shower distal vessels.
🔸
Board distinction: Acute tubular necrosis is reversible; ischemic nephropathy from chronic RAS causes irreversible structural damage.

Medical Management Principles
🧷
Goal: control blood pressure and preserve renal function while considering revascularization in appropriate candidates.
🧷
First-line agents in unilateral RAS with normal contralateral kidney: ACE inhibitors or ARBs — they reduce intraglomerular pressure and provide cardiovascular protection.
🧷
Avoid ACE inhibitors/ARBs in bilateral RAS or solitary kidney RAS — use calcium channel blockers, beta-blockers, or direct vasodilators.
🧷
Aggressive management of atherosclerotic risk factors: statins, antiplatelet therapy, smoking cessation.
🧷
Board pearl: The choice of antihypertensive depends on whether the patient has one or two functioning kidneys.

Indications for Revascularization
📍
Resistant hypertension despite optimal medical therapy (≥3 drugs including a diuretic).
📍
Progressive renal insufficiency, especially if bilateral disease or solitary kidney affected.
📍
Recurrent flash pulmonary edema or unstable angina from hypertensive surges.
📍
Viable kidney parenchyma on imaging — kidney length >8 cm suggests potential for functional recovery.
📍
Board pearl: Revascularization is less likely to improve renal function if the kidney is <8 cm or shows cortical thinning, indicating irreversible ischemic damage.

Revascularization Techniques
🔹
Percutaneous transluminal renal angioplasty (PTRA): first-line for fibromuscular dysplasia with excellent long-term patency rates (>90%).
🔹
PTRA with stenting: preferred for atherosclerotic RAS, especially ostial lesions. Stenting reduces restenosis compared to angioplasty alone.
🔹
Surgical revascularization: aortorenal bypass or endarterectomy. Reserved for complex anatomy, failed endovascular therapy, or need for concurrent aortic surgery.
🔹
Complications: contrast nephropathy, atheroembolic disease, renal artery dissection or rupture, restenosis.
🔹
Board pearl: FMD responds better to angioplasty alone; atherosclerotic disease usually requires stenting for durable results.

Differential Diagnosis of Secondary Hypertension
⭐
Primary hyperaldosteronism: low renin, high aldosterone, adrenal adenoma or bilateral hyperplasia on imaging.
⭐
Pheochromocytoma: episodic hypertension, headaches, palpitations, elevated plasma/urine metanephrines.
⭐
Cushing syndrome: central obesity, purple striae, proximal muscle weakness, elevated 24-hour urine cortisol.
⭐
Coarctation of aorta: upper extremity hypertension, delayed femoral pulses, rib notching on CXR.
⭐
Hyperthyroidism: wide pulse pressure, tachycardia, tremor, elevated free T4 and suppressed TSH.
⭐
Board pearl: Renovascular disease is the most common cause of secondary hypertension in elderly patients.

Pediatric Considerations
✅
Renovascular hypertension is the most common cause of secondary hypertension in children after coarctation of the aorta.
✅
Fibromuscular dysplasia is the predominant etiology in children, though congenital anomalies and neurofibromatosis-associated vasculopathy also occur.
✅
Children may present with growth retardation, headaches, or hypertensive encephalopathy rather than asymptomatic hypertension.
✅
Takayasu arteritis should be considered in adolescents, especially those of Asian descent, with constitutional symptoms and elevated inflammatory markers.
✅
Board clue: Child with neurofibromatosis type 1 + hypertension = renal artery stenosis from vascular dysplasia.

Pregnancy and Renovascular Hypertension
🧠
Renovascular hypertension can worsen during pregnancy due to increased cardiac output and blood volume.
🧠
ACE inhibitors and ARBs are absolutely contraindicated in pregnancy — teratogenic effects include renal dysgenesis, oligohydramnios, and IUGR.
🧠
Safe antihypertensives: methyldopa, labetalol, nifedipine, hydralazine.
🧠
MR angiography without gadolinium is the preferred imaging modality if diagnosis needed during pregnancy.
🧠
Board pearl: Young pregnant woman with severe hypertension and abdominal bruit → consider fibromuscular dysplasia, manage with methyldopa or labetalol.

Atheroembolic Renal Disease
⚡
Cholesterol crystal embolization to small renal arteries, typically after vascular procedures or anticoagulation in patients with severe atherosclerosis.
⚡
Clinical triad: acute/subacute renal failure + livedo reticularis + eosinophilia. May see blue toe syndrome from distal embolization.
⚡
Laboratory findings: eosinophilia, hypocomplementemia, elevated ESR/CRP. Urinalysis shows minimal proteinuria.
⚡
Renal biopsy shows cholesterol clefts (needle-shaped spaces) in small arteries — pathognomonic finding.
⚡
Board pearl: AKI + livedo reticularis + eosinophilia after cardiac catheterization = atheroembolic disease, not contrast nephropathy.

Monitoring and Prognosis
📌
Blood pressure response to revascularization: cure (normotensive off medications) in 35%, improvement (better control with fewer medications) in 35%, no benefit in 30%.
📌
Predictors of good response: short duration of hypertension, FMD etiology, absence of renal atrophy, young age.
📌
Renal function may transiently worsen after revascularization due to reperfusion injury before improving.
📌
Surveillance imaging at 1, 6, and 12 months, then annually to detect restenosis — duplex ultrasound for screening, angiography if intervention planned.
📌
Board pearl: Best outcomes occur when revascularization performed before irreversible ischemic nephropathy develops.

Board Question Stem Patterns
📣
Young woman + severe hypertension + abdominal bruit + "string of beads" on imaging → fibromuscular dysplasia.
📣
Elderly man + resistant hypertension + atherosclerotic disease + AKI after ACE inhibitor → bilateral atherosclerotic RAS.
📣
Hypertension + hypokalemia + high renin + high aldosterone → renovascular hypertension (not primary aldosteronism).
📣
Flash pulmonary edema + severe hypertension + bilateral renal artery stenosis → immediate revascularization indicated.
📣
Child with neurofibromatosis + new hypertension → screen for renal artery stenosis.
📣
Renal atrophy + cortical thinning + small kidney → irreversible ischemic nephropathy, poor revascularization candidate.

One-Line Recap
🔸
Renovascular hypertension results from renal hypoperfusion activating RAAS, caused by atherosclerotic disease in older patients or fibromuscular dysplasia in younger patients, presenting with resistant hypertension and diagnosed by imaging showing renal artery stenosis, with management ranging from medical therapy to revascularization based on clinical severity and kidney viability.

bottom of page

