top of page
Cardiovascular System
Preload, afterload, contractility, compliance
Core Principle of Cardiac Hemodynamics
🧷
The heart functions as a pressure-volume pump whose output depends on four interdependent determinants: preload (ventricular filling), afterload (resistance to ejection), contractility (intrinsic pump strength), and compliance (ventricular distensibility).
🧷
These parameters interact through the Frank-Starling mechanism, Laplace's law, and pressure-volume relationships to determine stroke volume and cardiac output.
🧷
Understanding how each parameter affects the pressure-volume loop is essential for predicting hemodynamic responses to pathology and therapeutic interventions.
🧷
Board pearl: Changes in any one parameter alter the others — increased afterload reduces stroke volume, triggering compensatory increases in preload and contractility.

Preload: The Ventricular Filling Determinant
📍
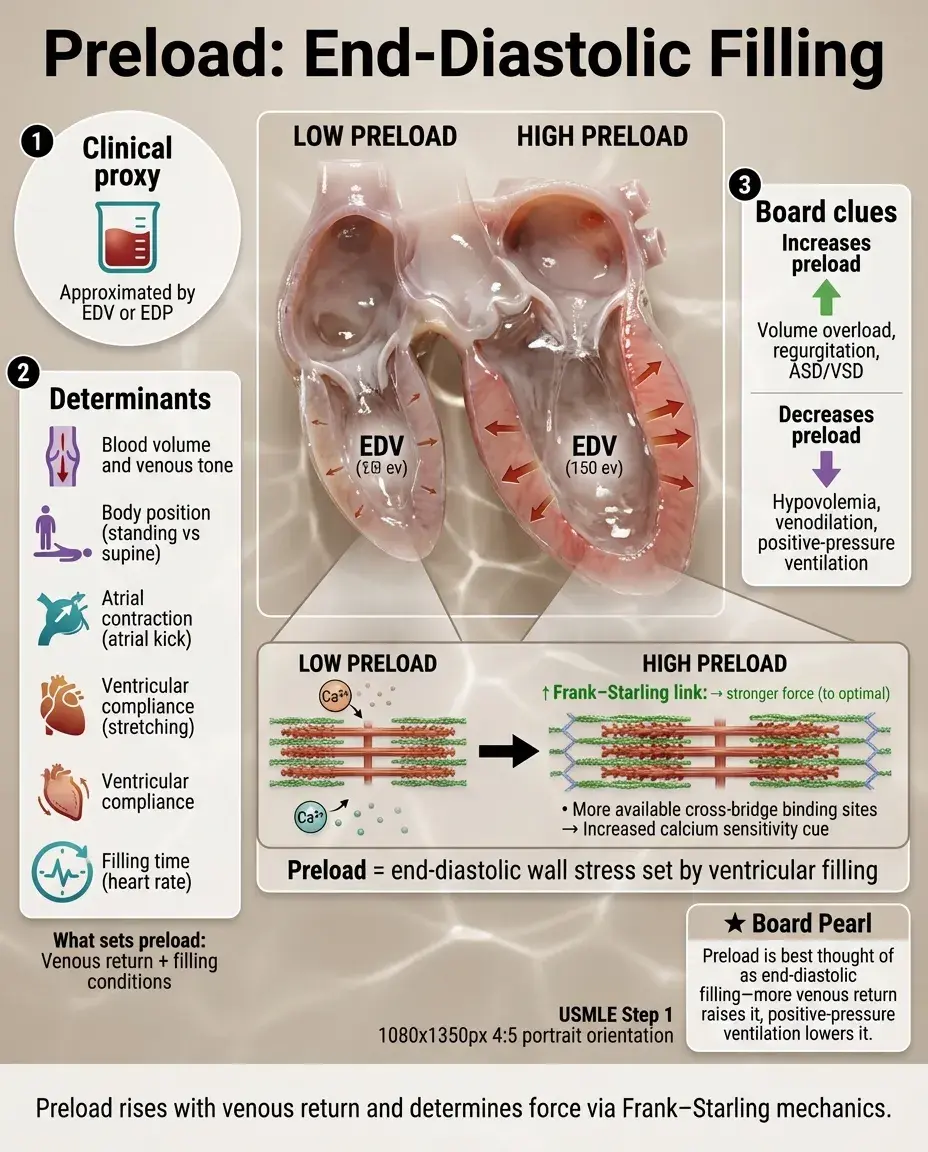
Preload is the ventricular wall stress at end-diastole, clinically approximated by end-diastolic volume (EDV) or end-diastolic pressure (EDP).
📍
Determinants: venous return (affected by blood volume, venous tone, body position), atrial contraction (lost in atrial fibrillation), ventricular compliance, and filling time (heart rate).
📍
The Frank-Starling mechanism: increased preload → increased sarcomere length → enhanced calcium sensitivity → increased contractile force up to an optimal length.
📍
Board clue: Conditions increasing preload include volume overload, mitral/tricuspid regurgitation, and ASD/VSD; conditions decreasing preload include hypovolemia, venodilation, and positive-pressure ventilation.
Afterload: The Resistance to Ejection
🔹
Afterload is the ventricular wall stress during systolic ejection, clinically approximated by aortic pressure (LV) or pulmonary artery pressure (RV).
🔹
By Laplace's law: Wall stress = (Pressure × Radius) / (2 × Wall thickness), explaining why dilated ventricles face higher afterload despite similar pressures.
🔹
Primary determinants: systemic vascular resistance (SVR) for the LV, pulmonary vascular resistance (PVR) for the RV, and ventricular radius.
🔹
Board pearl: Afterload reducers (ACE inhibitors, ARBs, hydralazine) improve cardiac output in heart failure by decreasing the work required for ejection, particularly beneficial when contractility is impaired.

Contractility: The Intrinsic Pump Strength
⭐
Contractility (inotropy) is the intrinsic ability of cardiac muscle to generate force independent of preload and afterload changes.
⭐
Molecular basis: calcium availability to troponin C determines contractile force — more Ca²⁺ binding = stronger contraction.
⭐
Positive inotropes: β₁-agonists (↑ cAMP → ↑ Ca²⁺), digoxin (↑ intracellular Na⁺ → ↑ Ca²⁺), PDE inhibitors (↑ cAMP).
⭐
Negative inotropes: β-blockers, calcium channel blockers (verapamil/diltiazem), ischemia, acidosis.
⭐
Board distinction: Unlike preload and afterload which can be measured, contractility must be inferred from ejection fraction at given loading conditions.

Compliance: The Ventricular Distensibility
✅
Compliance = ΔVolume/ΔPressure; it determines how much the ventricle fills at a given filling pressure.
✅
Normal ventricle: high compliance allows large volume changes with minimal pressure increase.
✅
Decreased compliance (diastolic dysfunction): restrictive cardiomyopathy, ventricular hypertrophy, ischemia, aging → higher filling pressures needed for same EDV.
✅
Board pearl: In diastolic heart failure (HFpEF), ejection fraction is preserved but compliance is reduced → pulmonary congestion occurs at lower filling volumes due to elevated filling pressures.

The Pressure-Volume Loop: Integrating All Four Parameters
🧠
The PV loop graphically displays the cardiac cycle with pressure on y-axis, volume on x-axis.
🧠
Loop boundaries: ESPVR (end-systolic pressure-volume relationship) represents contractility; EDPVR (end-diastolic pressure-volume relationship) represents compliance.
🧠
Width = stroke volume; area = stroke work; rightward shift = increased preload; upward shift = increased afterload.
🧠
Board approach: Given a PV loop change, identify which parameter changed — wider loop suggests increased preload or contractility; taller/narrower suggests increased afterload; steeper EDPVR suggests decreased compliance.

Frank-Starling Mechanism and Its Limits
⚡
Increased ventricular filling (preload) → increased sarcomere length (1.6→2.2 μm) → optimal actin-myosin overlap → increased force generation.
⚡
This intrinsic property ensures that increased venous return automatically increases cardiac output without neural input.
⚡
Beyond optimal length (~2.2 μm), further stretch decreases force — the descending limb of the Starling curve seen in decompensated heart failure.
⚡
Board pearl: In early heart failure, the Frank-Starling mechanism maintains cardiac output; in advanced failure, the ventricle operates on the descending limb where increased preload worsens function.

Clinical Assessment of Preload
📌
Right heart: central venous pressure (CVP), jugular venous pressure (JVP), inferior vena cava diameter on ultrasound.
📌
Left heart: pulmonary capillary wedge pressure (PCWP), left atrial pressure, E/e' ratio on echocardiography.
📌
Volume status clues: orthostatic vital signs, skin turgor, mucous membranes, urine output, BUN/Cr ratio.
📌
Board distinction: CVP reflects right atrial pressure and RV preload; PCWP reflects left atrial pressure and LV preload — they can diverge in isolated right or left heart failure.

Afterload in Specific Conditions
📣
Hypertension: chronically elevated afterload → LV hypertrophy → preserved ejection fraction initially but eventual systolic dysfunction.
📣
Aortic stenosis: fixed obstruction creates extreme afterload → concentric hypertrophy → eventual decompensation with reduced stroke volume.
📣
Mitral regurgitation: reduced effective afterload (blood flows backward into low-pressure LA) → initially preserved ejection fraction masking LV dysfunction.
📣
Board clue: Sudden afterload reduction (e.g., starting vasodilator) in severe aortic stenosis can cause cardiovascular collapse — afterload must match the fixed obstruction.

Contractility Assessment and Modulation
🔸
Clinical surrogates: ejection fraction (load-dependent), dP/dt max (rate of pressure rise), fractional shortening.
🔸
Dobutamine (β₁ agonist): ↑ contractility > ↑ heart rate; used in cardiogenic shock and stress testing.
🔸
Milrinone (PDE-3 inhibitor): ↑ contractility + vasodilation (inodilator); useful when β-receptors are downregulated.
🔸
Digoxin: mild ↑ contractility via Na⁺/K⁺-ATPase inhibition; narrow therapeutic window, watch for toxicity.
🔸
Board pearl: In chronic heart failure, long-term positive inotropes (except digoxin) increase mortality despite short-term hemodynamic improvement.

Compliance Patterns in Disease States
🧷
Hypertrophic cardiomyopathy: markedly decreased compliance → small LV cavity, diastolic dysfunction, preserved ejection fraction.
🧷
Restrictive cardiomyopathy (amyloid, hemochromatosis): stiff ventricles → rapid early filling then abrupt stop (square root sign on catheterization).
🧷
Constrictive pericarditis: normal myocardial compliance but external constraint → equalization of diastolic pressures, ventricular interdependence.
🧷
Board distinction: Restriction vs. constriction — both show square root sign, but respiratory variation in mitral/tricuspid inflow (pulsus paradoxus) suggests constriction.

Integrated Responses to Volume Loading
📍
Initial response: ↑ preload → ↑ stroke volume via Frank-Starling mechanism.
📍
If compliance is normal: large volume increase with modest pressure rise → good cardiac output augmentation.
📍
If compliance is reduced: small volume increase with large pressure rise → pulmonary congestion before adequate output increase.
📍
Chronic adaptation: ventricular dilation to accommodate volume (eccentric hypertrophy) → eventual systolic dysfunction.
📍
Board pearl: Rapid IV fluids in diastolic dysfunction → flash pulmonary edema due to steep EDPVR curve.

Hemodynamic Effects of Common Medications
🔹
ACE inhibitors/ARBs: ↓ afterload (vasodilation) + ↓ preload (less aldosterone → less volume retention).
🔹
β-blockers: ↓ contractility + ↓ heart rate → allow more filling time → may initially ↓ cardiac output but long-term benefit in heart failure.
🔹
Diuretics: ↓ preload via volume reduction; monitor for excessive preload reduction → ↓ cardiac output.
🔹
Calcium channel blockers: non-dihydropyridines ↓ contractility; all ↓ afterload via vasodilation.
🔹
Board distinction: Verapamil/diltiazem decrease contractility (avoid in systolic heart failure); amlodipine is purely vasodilatory (safe in systolic dysfunction).

Right Ventricular vs. Left Ventricular Hemodynamics
⭐
RV: thin-walled, compliant, sensitive to afterload changes; normally works against low resistance (PVR ~10% of SVR).
⭐
RV afterload sensitivity: acute PE → RV dilation → septal shift → ↓ LV filling → obstructive shock.
⭐
LV: thick-walled, generates high pressure, tolerates afterload better but sensitive to volume overload.
⭐
Ventricular interdependence: pericardial constraint means RV dilation → ↓ LV compliance and filling.
⭐
Board pearl: RV infarct presents with hypotension, clear lungs, elevated JVP — preload dependent state requiring IV fluids despite high CVP.

Exercise Physiology and Hemodynamic Reserve
✅
Normal exercise response: ↑ venous return (muscle pump) → ↑ preload; ↑ contractility (sympathetic activation); ↓ afterload (skeletal muscle vasodilation); maintained compliance.
✅
Heart failure: limited preload reserve (already volume overloaded), reduced contractility reserve, inability to reduce afterload → exertional dyspnea.
✅
Chronotropic incompetence: inability to increase heart rate → reliance on stroke volume alone → early fatigue.
✅
Board clue: Fixed cardiac output states (severe AS, MS, constrictive pericarditis) → syncope with exertion due to inability to increase output despite peripheral vasodilation.

Shock States and Hemodynamic Patterns
🧠
Cardiogenic: ↓ contractility → ↓ cardiac output despite ↑ preload (elevated PCWP) and ↑ afterload (compensatory vasoconstriction).
🧠
Hypovolemic: ↓ preload → ↓ cardiac output; compensatory ↑ heart rate and ↑ afterload (cool extremities).
🧠
Distributive (septic): ↓ afterload (vasodilation) → ↑ cardiac output initially; ↓ preload (venous pooling); may have ↓ contractility (septic cardiomyopathy).
🧠
Obstructive: mechanical impediment (PE, tamponade) → ↓ preload and/or ↑ afterload → ↓ cardiac output.
🧠
Board pearl: Swan-Ganz patterns — high PCWP + low CO = cardiogenic; low PCWP + low CO = hypovolemic; low SVR + high/normal CO = distributive.

Valvular Disease Effects on Loading Conditions
⚡
Aortic stenosis: ↑ afterload → concentric hypertrophy → ↓ compliance → preload dependent but prone to pulmonary edema.
⚡
Aortic regurgitation: ↑ preload (volume overload) + ↓ effective afterload (low diastolic pressure) → eccentric hypertrophy.
⚡
Mitral stenosis: ↓ LV preload (obstruction to filling) → ↓ cardiac output; ↑ LA pressure → pulmonary hypertension → RV afterload stress.
⚡
Mitral regurgitation: ↓ forward afterload + ↑ preload → initially preserved ejection fraction masking LV dysfunction.
⚡
Board distinction: Chronic MR → normal LA pressure (dilated, compliant LA); acute MR → very high LA pressure (non-compliant LA) → pulmonary edema.

Heart Failure Compensatory Mechanisms
📌
Immediate: Frank-Starling (↑ preload → maintain stroke volume); sympathetic activation (↑ heart rate + contractility).
📌
Short-term: RAAS activation → salt/water retention (↑ preload) + vasoconstriction (maintain perfusion pressure).
📌
Long-term maladaptive: chronic ↑ preload → ventricular dilation; chronic ↑ afterload → hypertrophy; chronic ↑ contractility demand → energy depletion.
📌
Neurohormonal antagonism rationale: block RAAS (ACE-I/ARB) and sympathetic (β-blockers) to prevent remodeling.
📌
Board pearl: BNP release triggered by ventricular stretch (↑ preload) → promotes natriuresis to counteract volume overload.

Board Question Stem Patterns
📣
Elderly woman with exertional dyspnea, normal EF, small LV cavity → diastolic dysfunction from ↓ compliance.
📣
Post-MI patient with new murmur and pulmonary edema → acute MR causing ↓ afterload but overwhelming ↑ preload.
📣
Marathon runner with bradycardia and large stroke volume → ↑ preload tolerance and ↑ contractility reserve.
📣
Septic patient with warm extremities and wide pulse pressure → ↓ afterload from vasodilation.
📣
Patient with cannon A waves and hypotension during VT → loss of atrial kick → ↓ preload contribution.
📣
Squatting improving murmur intensity → ↑ preload and afterload effects on HOCM or MVP.
📣
Valsalva decreasing murmur → ↓ preload reducing flow-dependent murmurs (AS, MR) but worsening HOCM.

One-Line Recap
🔸
Cardiac output depends on four parameters — preload (end-diastolic wall stress determining filling via Frank-Starling), afterload (systolic wall stress opposing ejection via Laplace's law), contractility (calcium-mediated intrinsic force generation), and compliance (diastolic pressure-volume relationship) — whose integration explains hemodynamic responses to disease, exercise, medications, and shock states.

bottom of page

