top of page
Urinary System
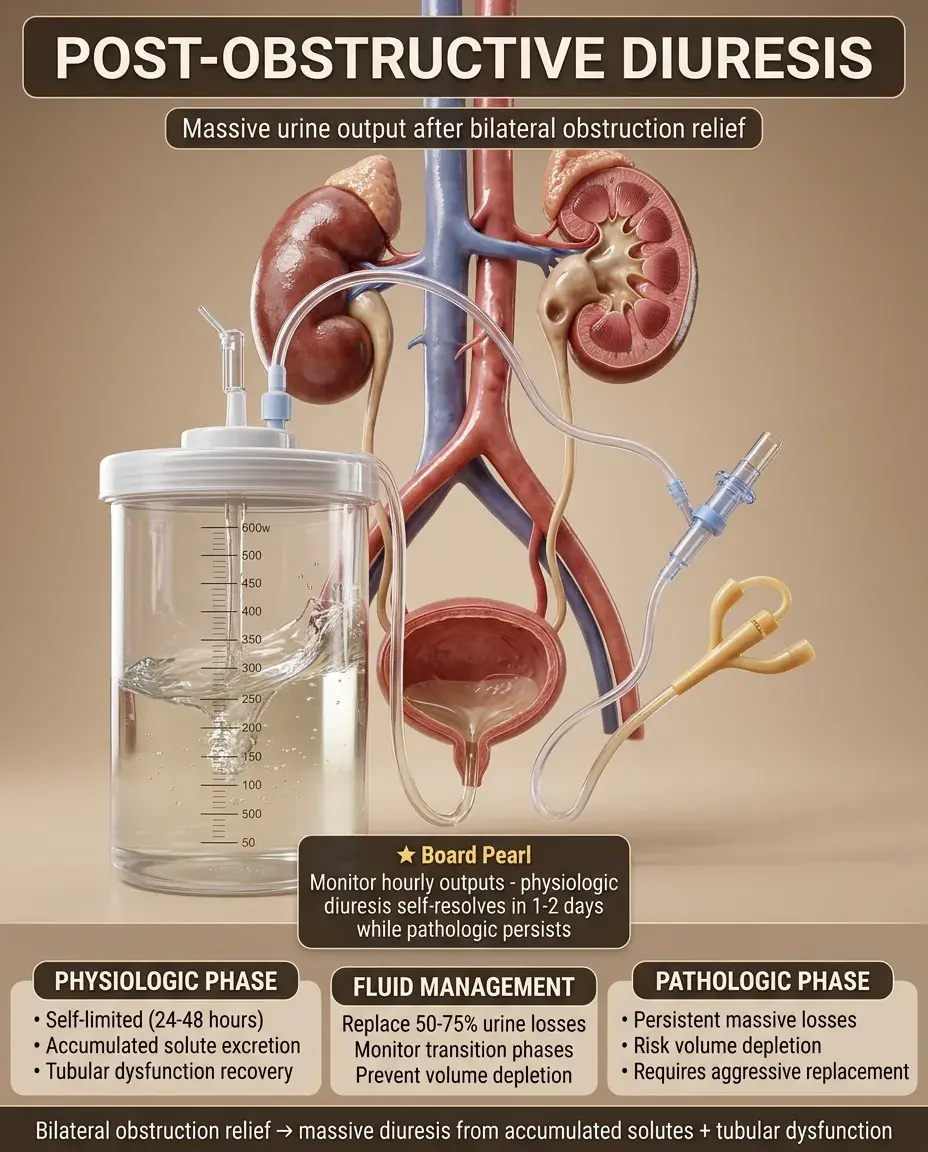
Post-obstructive diuresis
Core Principle of Post-obstructive Diuresis
🧷
Post-obstructive diuresis is the excessive urine output (>200 mL/hr or >3 L/day) that occurs after relief of bilateral urinary obstruction or unilateral obstruction in a solitary kidney.
🧷
The mechanism involves accumulated solutes (urea, sodium) and volume that were retained during obstruction, combined with tubular dysfunction from prolonged back-pressure.
🧷
This represents both a physiologic response to clear retained waste products and a pathologic impairment of tubular concentrating ability.
🧷
Board pearl: The diuresis is self-limited in most cases but can become pathologic if tubular damage prevents appropriate concentration of urine.

Pathophysiology of Tubular Dysfunction
📍
During obstruction, increased intratubular pressure damages the medullary concentration gradient and impairs tubular responsiveness to ADH.
📍
Chronic obstruction causes tubular atrophy, interstitial fibrosis, and downregulation of aquaporin channels → nephrogenic diabetes insipidus-like state.
📍
Accumulated urea acts as an osmotic diuretic once flow is restored, pulling water into the tubular lumen.
📍
The kidney cannot immediately restore its concentrating ability even after obstruction relief — recovery takes days to weeks.
📍
Board clue: The severity of diuresis correlates with the duration and completeness of the preceding obstruction.

Types of Post-obstructive Diuresis
🔹
Physiologic diuresis: appropriate excretion of retained sodium, water, and urea. Self-limited, lasting 24-48 hours. No intervention needed beyond monitoring.
🔹
Pathologic diuresis: inappropriate ongoing losses despite normalization of volume status. Due to severe tubular damage. Requires careful fluid replacement.
🔹
The distinction is made by monitoring urine output, electrolytes, and volume status after the initial diuretic phase.
🔹
Board distinction: Physiologic diuresis stops when excess solute is cleared; pathologic diuresis continues despite euvolemia.

Clinical Presentation and Timing
⭐
Diuresis typically begins within hours of obstruction relief (catheter placement, nephrostomy tube, surgical intervention).
⭐
Urine output can reach 500-1000 mL/hr in severe cases — a dramatic increase that alarms clinicians.
⭐
Patients may develop orthostatic symptoms, tachycardia, and signs of volume depletion if losses exceed intake.
⭐
Laboratory findings: initial improvement in creatinine, followed by potential worsening if volume depletion occurs.
⭐
Board pearl: The most common cause is bladder outlet obstruction from BPH in elderly men.

Risk Factors and Predisposing Conditions
✅
Bilateral ureteral obstruction: stones, retroperitoneal fibrosis, malignancy, iatrogenic ligation.
✅
Bladder outlet obstruction: BPH (most common), prostate cancer, neurogenic bladder, urethral stricture.
✅
Solitary kidney with obstruction: previous nephrectomy, congenital absence, or non-functioning contralateral kidney.
✅
Duration of obstruction: longer obstruction → more severe tubular damage → greater risk of pathologic diuresis.
✅
Pre-existing CKD: less renal reserve to handle the stress of obstruction and subsequent diuresis.

Initial Evaluation and Monitoring
🧠
Measure urine output hourly for the first 24-48 hours after obstruction relief.
🧠
Check serum electrolytes (Na⁺, K⁺, HCO₃⁻, Mg²⁺, PO₄³⁻) every 6-8 hours initially.
🧠
Monitor vital signs for evidence of volume depletion: orthostatic hypotension, tachycardia.
🧠
Daily weights provide the most accurate assessment of net fluid balance.
🧠
Serial creatinine to ensure improving renal function — rising creatinine suggests inadequate volume replacement.

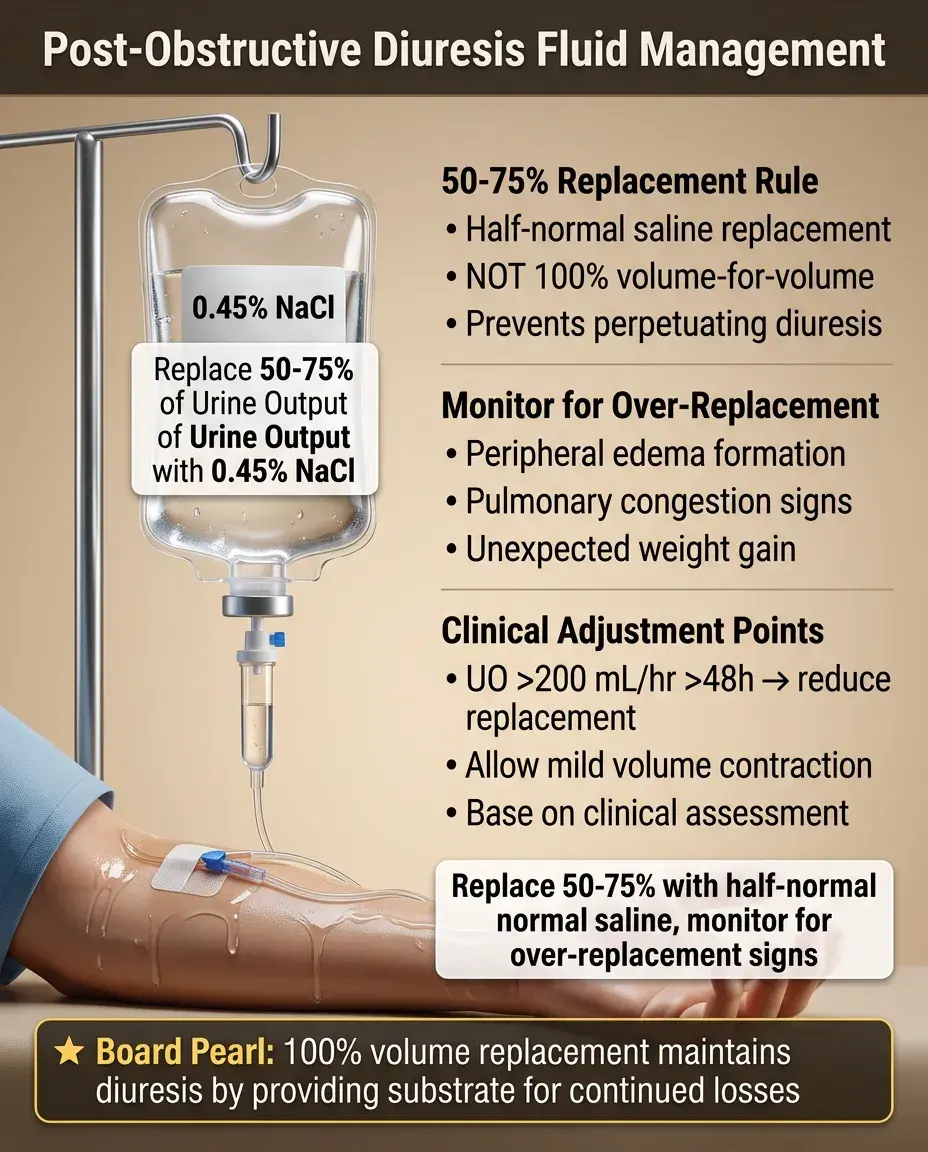
Fluid Management Strategy
⚡
Replace 50-75% of urine output with 0.45% NaCl (half-normal saline) — not 100% replacement which perpetuates diuresis.
⚡
If urine output >200 mL/hr persists beyond 48 hours, consider reducing replacement to allow mild volume contraction.
⚡
Monitor for signs of over-replacement: peripheral edema, pulmonary congestion, weight gain.
⚡
Board pearl: Complete volume-for-volume replacement maintains the diuresis by providing substrate for continued losses.
⚡
Adjust replacement based on clinical assessment, not solely on urine output.
Electrolyte Abnormalities and Management
📌
Hyponatremia: from excessive free water losses. Treat with normal saline if hypovolemic.
📌
Hypokalemia: from urinary K⁺ losses and secondary hyperaldosteronism. Replace cautiously — rapid correction risks arrhythmias.
📌
Hypomagnesemia: often overlooked but common. Contributes to refractory hypokalemia and hypocalcemia.
📌
Metabolic acidosis: from HCO₃⁻ losses if severe tubular dysfunction. Usually mild and self-limited.
📌
Board distinction: Electrolyte abnormalities reflect tubular dysfunction, not just volume losses.

Distinguishing Physiologic from Pathologic Diuresis
📣
Calculate electrolyte-free water clearance: if urine is hypotonic relative to plasma, excessive free water loss is occurring.
📣
Urine osmolality <300 mOsm/kg suggests impaired concentrating ability.
📣
Urine sodium: high (>40 mEq/L) suggests salt wasting; low (<20 mEq/L) suggests appropriate conservation.
📣
Response to fluid restriction trial: physiologic diuresis will slow; pathologic continues.
📣
Board approach: After 48 hours, if high urine output persists despite clinical euvolemia → pathologic diuresis.

Complications of Post-obstructive Diuresis
🔸
Volume depletion → prerenal azotemia → worsening renal function despite relief of obstruction.
🔸
Electrolyte disturbances → cardiac arrhythmias (especially from K⁺ and Mg²⁺ depletion).
🔸
Postural hypotension → falls and fractures in elderly patients.
🔸
Overcorrection with aggressive fluid replacement → volume overload, CHF exacerbation.
🔸
Board pearl: The goal is to prevent complications while allowing the physiologic diuresis to occur.

Recovery of Tubular Function
🧷
Concentrating ability (response to ADH) recovers slowly over days to weeks if tubular damage is reversible.
🧷
Persistent concentrating defect suggests chronic tubular atrophy and interstitial fibrosis.
🧷
GFR improvement follows a predictable pattern: rapid initial improvement, then plateau.
🧷
Maximum recovery typically achieved by 7-10 days; further improvement unlikely.
🧷
Degree of recovery inversely correlates with duration of obstruction — complete obstruction >2 weeks often causes permanent damage.

Special Populations: Solitary Kidney
📍
Post-obstructive diuresis is more severe when a solitary kidney was obstructed — no contralateral kidney to compensate.
📍
Higher risk of pathologic diuresis due to greater tubular damage from handling the entire filtered load.
📍
More aggressive initial monitoring required — these patients can decompensate rapidly.
📍
Consider nephrology consultation early for complex fluid and electrolyte management.
📍
Board scenario: Patient with previous nephrectomy develops acute obstruction → expect severe diuresis after relief.

Role of Diuretics and Medications
🔹
Diuretics are contraindicated during post-obstructive diuresis — they worsen volume depletion.
🔹
Hold ACE inhibitors/ARBs initially — they impair renal autoregulation when GFR is recovering.
🔹
NSAIDs should be avoided — they decrease renal blood flow and impair recovery.
🔹
Nephrotoxic medications (aminoglycosides, contrast) pose higher risk during recovery period.
🔹
Resume medications cautiously once urine output normalizes and renal function plateaus.

When to Consult Nephrology
⭐
Pathologic diuresis persisting >48-72 hours despite appropriate management.
⭐
Severe electrolyte disturbances requiring complex replacement strategies.
⭐
Rising creatinine despite adequate volume replacement.
⭐
Unclear etiology of obstruction requiring further workup.
⭐
Pre-existing CKD with limited renal reserve — these patients have less margin for error.

Long-term Sequelae
✅
Chronic tubulointerstitial disease: permanent concentrating defect, salt wasting, mild proteinuria.
✅
Increased risk of CKD progression, especially if obstruction was prolonged.
✅
Persistent polyuria/polydipsia from acquired nephrogenic diabetes insipidus.
✅
Recurrent UTIs from bladder dysfunction and incomplete emptying.
✅
Board concept: Even after successful relief, some degree of permanent tubular dysfunction often remains.

Prevention of Recurrent Obstruction
🧠
Address underlying cause: TURP for BPH, stone prevention protocols, treatment of neurogenic bladder.
🧠
Regular monitoring with renal ultrasound to detect silent re-obstruction.
🧠
Patient education about early symptoms: decreased urine output, flank pain, uremic symptoms.
🧠
Prophylactic measures: adequate hydration for stone formers, intermittent catheterization for neurogenic bladder.
🧠
Board pearl: Preventing re-obstruction is crucial as repeated episodes cause cumulative renal damage.

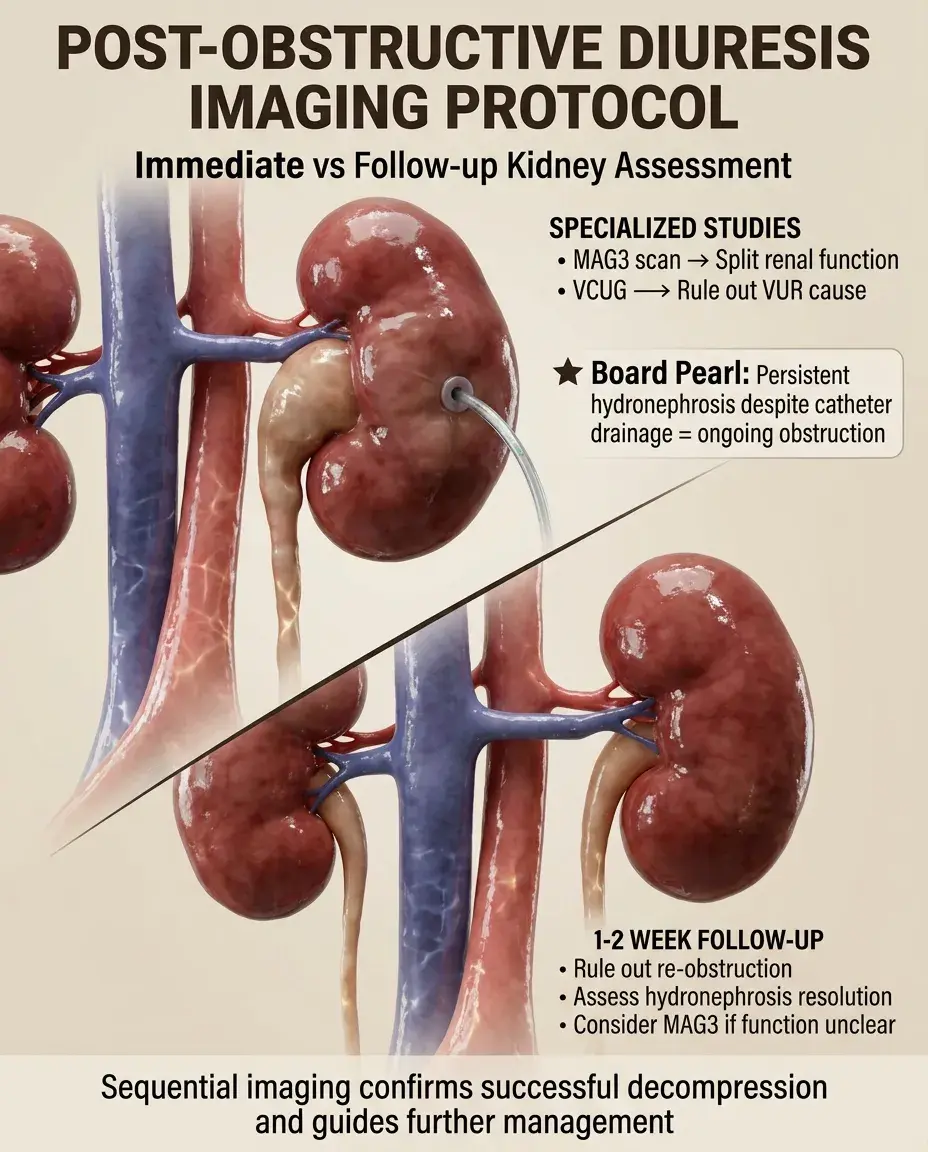
Imaging and Follow-up
⚡
Immediate post-relief imaging: confirm decompression of collecting system.
⚡
Follow-up ultrasound at 1-2 weeks: ensure no re-obstruction, assess for hydronephrosis resolution.
⚡
MAG3 renal scan if differential function needed: determines split renal function after recovery.
⚡
Voiding cystourethrogram if vesicoureteral reflux suspected as cause.
⚡
Board clue: Persistent hydronephrosis on imaging despite catheter drainage suggests ongoing obstruction.
Prognosis and Recovery Predictors
📌
Duration of obstruction: <1 week → full recovery likely; >2 weeks → permanent damage common.
📌
Degree of initial renal impairment: mild elevation in creatinine has better prognosis.
📌
Patient age: younger patients have better regenerative capacity.
📌
Presence of infection: pyonephrosis causes more severe and permanent damage.
📌
Completeness of obstruction: partial obstruction has better outcome than complete.

Board Question Stem Patterns
📣
Elderly man with BPH, acute urinary retention, catheter placed → massive urine output → post-obstructive diuresis.
📣
Urine output 500 mL/hr after nephrostomy tube placement → monitor and replace 50-75% of losses with 0.45% NaCl.
📣
Persistent polyuria 72 hours after obstruction relief with urine osmolality 250 → pathologic diuresis from tubular damage.
📣
Rising creatinine despite catheter drainage and IV fluids → inadequate volume replacement or irreversible renal damage.
📣
Severe hypokalemia and hypomagnesemia after obstruction relief → tubular dysfunction with urinary electrolyte wasting.

One-Line Recap
🔸
Post-obstructive diuresis results from accumulated solute excretion and tubular dysfunction after relief of bilateral obstruction, managed by replacing 50-75% of urine losses while monitoring for the transition from physiologic (self-limited) to pathologic (persistent) diuresis, with the goal of preventing volume depletion while allowing appropriate excretion of retained waste products.
bottom of page

