top of page
Behavioral Health & Nervous System
Pharmacologic Modulation of Sleep
Core Principle of Sleep Pharmacology
🧷
Sleep-wake regulation involves complex interactions between neurotransmitter systems: wake-promoting (histamine, orexin, norepinephrine, dopamine, acetylcholine) and sleep-promoting (GABA, adenosine, melatonin).
🧷
Pharmacologic modulation targets these systems to either promote sleep (sedative-hypnotics) or wakefulness (stimulants), with different drug classes affecting sleep architecture differently.
🧷
Understanding receptor mechanisms predicts both therapeutic effects and side effect profiles — GABAergic drugs cause anterograde amnesia, antihistamines cause anticholinergic effects, and orexin antagonists preserve sleep architecture.
🧷
Board pearl: Sleep medications work by either enhancing inhibitory tone (GABA, melatonin) or blocking excitatory signals (histamine, orexin).

GABA-A Receptor and Benzodiazepines
📍
Benzodiazepines bind to the α-γ interface of GABA-A receptors, increasing the frequency of chloride channel opening when GABA is present — they are positive allosteric modulators.
📍
This enhanced chloride influx hyperpolarizes neurons, reducing neuronal excitability throughout the CNS, producing sedation, anxiolysis, muscle relaxation, and anticonvulsant effects.
📍
All benzodiazepines decrease sleep latency and increase total sleep time but suppress REM sleep and slow-wave sleep, altering normal sleep architecture.
📍
Board pearl: Benzodiazepines require endogenous GABA to work — they enhance but cannot replace GABA signaling, explaining why they are safer in overdose than barbiturates.

Z-Drugs: Selective GABA-A Modulation
🔹
Zolpidem, zaleplon, and eszopiclone ("Z-drugs") selectively bind to GABA-A receptors containing α1 subunits, which are enriched in sleep-promoting brain regions.
🔹
This selectivity produces hypnotic effects with less muscle relaxation and anticonvulsant activity compared to benzodiazepines, though the clinical significance remains debated.
🔹
Zaleplon has the shortest half-life (1 hour) — ideal for sleep initiation; zolpidem is intermediate (2.5 hours); eszopiclone is longest (6 hours) — maintains sleep throughout the night.
🔹
Board pearl: Despite marketed differences, Z-drugs share the same dependence potential and amnestic side effects as benzodiazepines because they work through the same GABA-A mechanism.

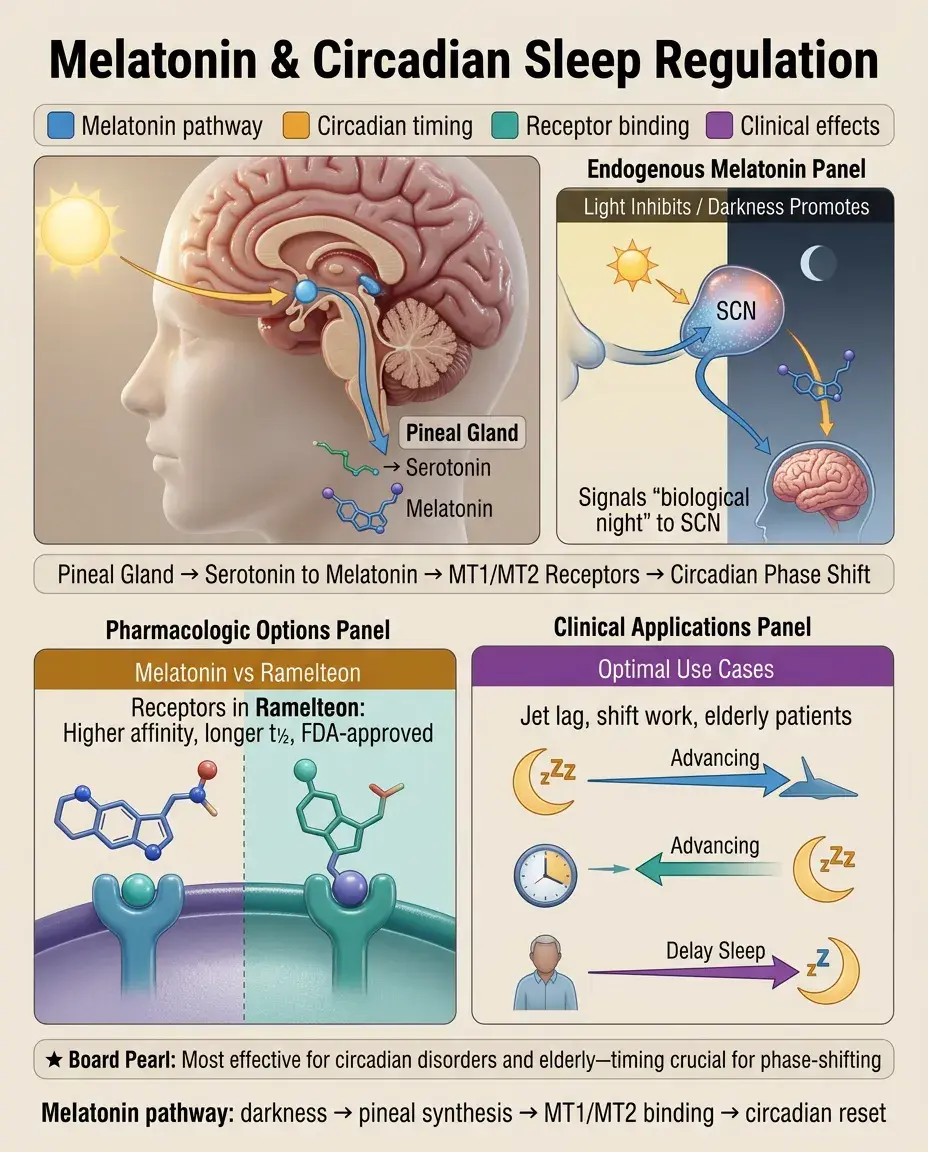
Melatonin and Circadian Regulation
⭐
Melatonin is synthesized from serotonin in the pineal gland, with secretion inhibited by light and promoted by darkness — it signals "biological night" to the body.
⭐
Exogenous melatonin acts on MT1 and MT2 receptors in the suprachiasmatic nucleus to phase-shift circadian rhythms and promote sleep onset, but has minimal effect on sleep maintenance.
⭐
Ramelteon is a selective MT1/MT2 agonist with higher affinity and longer half-life than melatonin, FDA-approved for sleep-onset insomnia without abuse potential.
⭐
Board pearl: Melatonin is most effective for circadian rhythm disorders (jet lag, shift work) and in elderly patients with reduced endogenous production — timing is critical for phase-shifting effects.
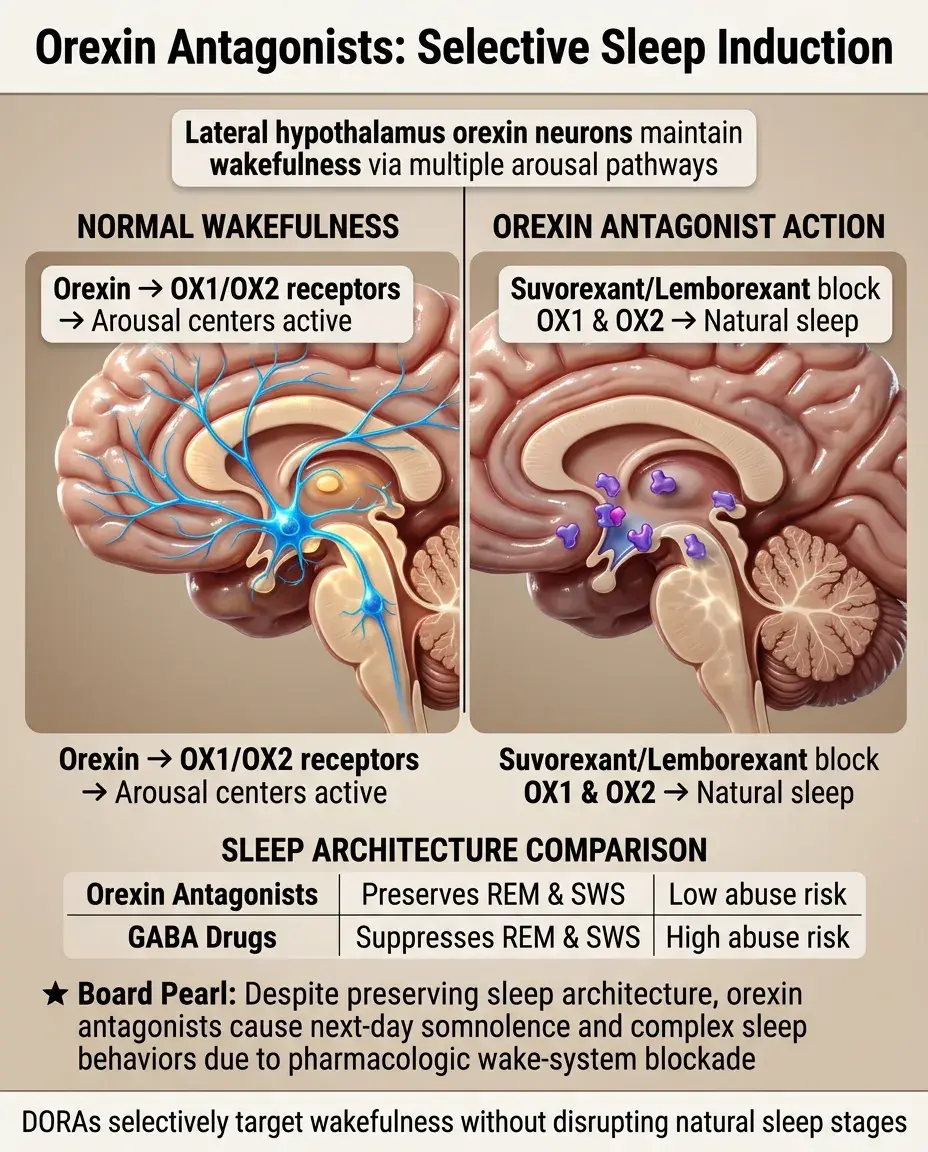
Orexin Antagonists: Preserving Sleep Architecture
✅
Orexin (hypocretin) neurons in the lateral hypothalamus promote wakefulness by activating multiple arousal centers — their loss causes narcolepsy with cataplexy.
✅
Suvorexant and lemborexant are dual orexin receptor antagonists (DORAs) that block both OX1 and OX2 receptors, promoting sleep without suppressing REM or slow-wave sleep.
✅
Unlike GABAergic drugs, orexin antagonists preserve normal sleep architecture and lack abuse potential, making them attractive for chronic insomnia treatment.
✅
Board pearl: Orexin antagonists can cause next-day somnolence and complex sleep behaviors (sleep-driving) despite preserving sleep architecture — the wake-promoting system is pharmacologically blocked.
Antihistamines and OTC Sleep Aids
🧠
First-generation H1 antagonists (diphenhydramine, doxylamine) cross the blood-brain barrier and block histamine's wake-promoting effects, causing sedation.
🧠
These drugs also block muscarinic, α-adrenergic, and serotonin receptors, producing anticholinergic effects (dry mouth, urinary retention, confusion), orthostatic hypotension, and weight gain.
🧠
Tolerance to sedative effects develops rapidly (within days), while anticholinergic side effects persist, making them poor choices for chronic insomnia.
🧠
Board pearl: Diphenhydramine is contraindicated in elderly patients due to anticholinergic effects increasing fall risk and precipitating delirium — appears on the Beers Criteria.

Antidepressants for Sleep
⚡
Trazodone, mirtazapine, and doxepin are sedating antidepressants commonly used off-label for insomnia at doses below their antidepressant range.
⚡
Trazodone blocks 5-HT2A receptors and H1 receptors; mirtazapine blocks 5-HT2A, 5-HT2C, and H1 receptors; doxepin is a potent H1 antagonist at low doses (3-6 mg).
⚡
These drugs improve sleep continuity without significant REM suppression, but can cause morning grogginess, orthostatic hypotension (trazodone), and weight gain (mirtazapine).
⚡
Board pearl: Trazodone can cause priapism via α1-adrenergic blockade — patients should be counseled about this rare but serious adverse effect requiring emergency treatment.

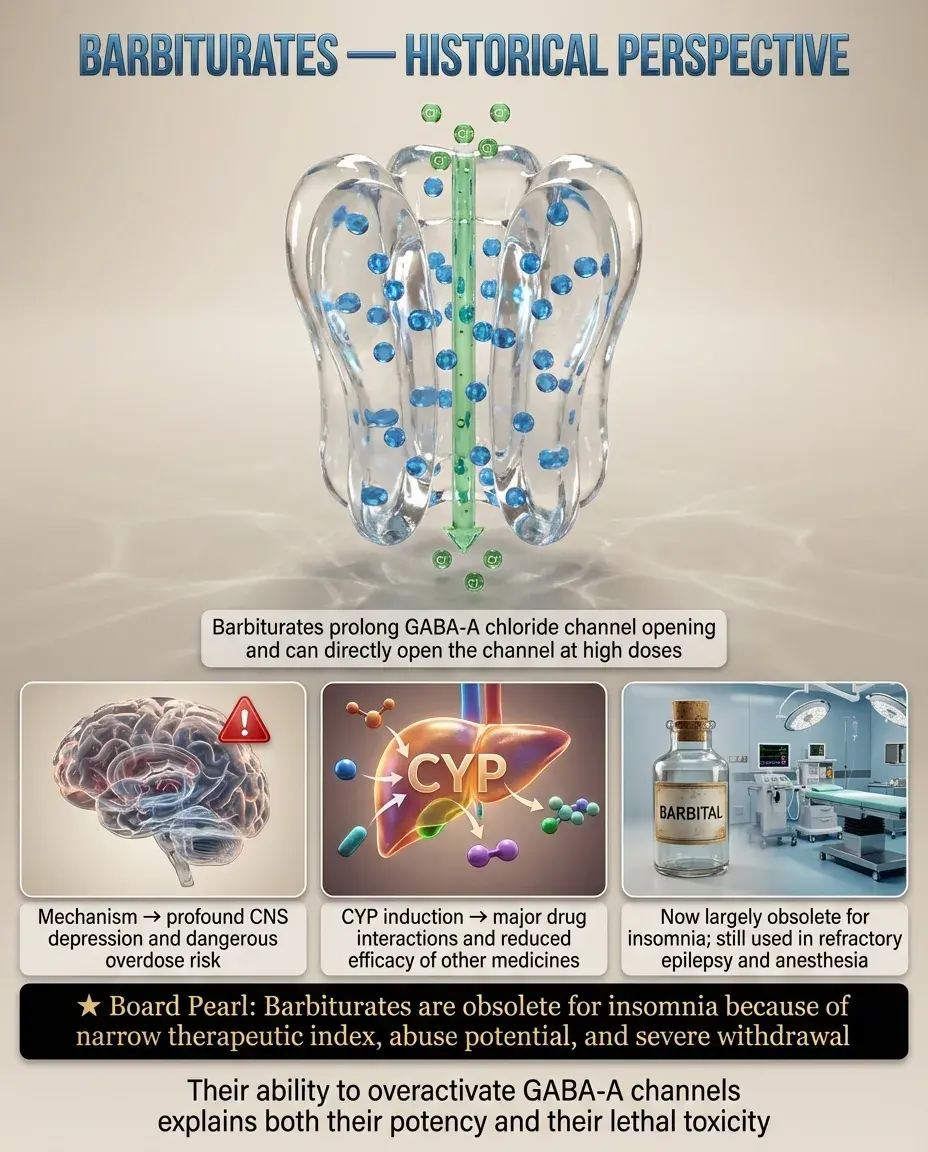
Barbiturates: Historical Perspective
📌
Barbiturates enhance GABA-A receptor function by increasing the duration of chloride channel opening and, at high doses, directly opening channels without GABA.
📌
This dual mechanism makes barbiturates dangerous in overdose — they can silence all CNS activity, causing respiratory depression and death, unlike benzodiazepines.
📌
Barbiturates induce cytochrome P450 enzymes (especially CYP3A4), causing numerous drug interactions and reducing effectiveness of oral contraceptives, warfarin, and other medications.
📌
Board pearl: Barbiturates are obsolete for insomnia due to narrow therapeutic index, high abuse potential, and severe withdrawal syndrome — now used only for refractory epilepsy and anesthesia.
Stimulants and Wake-Promoting Agents
📣
Modafinil and armodafinil promote wakefulness through unclear mechanisms involving dopamine reuptake inhibition, histamine release, and orexin activation without classical stimulant effects.
📣
Traditional stimulants (amphetamines, methylphenidate) increase synaptic dopamine and norepinephrine, treating narcolepsy and shift work disorder but with higher abuse potential.
📣
Caffeine blocks adenosine receptors, preventing sleep pressure accumulation — half-life of 5-6 hours means afternoon coffee can disrupt nighttime sleep.
📣
Board pearl: Modafinil induces CYP3A4 and reduces oral contraceptive effectiveness — women require backup contraception, distinguishing it from traditional stimulants.

Sleep Architecture and Drug Effects
🔸
Normal sleep cycles through NREM stages 1-3 and REM every 90-120 minutes, with more slow-wave sleep early and more REM sleep toward morning.
🔸
GABAergic drugs (benzodiazepines, Z-drugs) suppress REM sleep and decrease slow-wave sleep, leading to REM rebound when discontinued.
🔸
Most antidepressants suppress REM sleep (except trazodone, mirtazapine, nefazodone), which may contribute to their therapeutic effects in depression.
🔸
Board pearl: Alcohol initially promotes sleep through GABAergic effects but causes fragmented sleep and REM suppression, with REM rebound producing vivid dreams/nightmares during withdrawal.

Tolerance, Dependence, and Withdrawal
🧷
Tolerance to hypnotic effects of GABAergic drugs develops through GABA-A receptor downregulation and subunit changes, requiring dose escalation to maintain efficacy.
🧷
Physical dependence manifests as withdrawal syndrome upon discontinuation: insomnia, anxiety, tremor, seizures (severe cases) — severity correlates with dose and duration.
🧷
Rebound insomnia occurs with all GABAergic hypnotics, where sleep is worse than baseline for several nights after stopping, reinforcing continued use.
🧷
Board pearl: Benzodiazepine withdrawal can be life-threatening due to seizures — requires gradual taper over weeks to months, unlike opioid withdrawal which is uncomfortable but not dangerous.

Special Populations: Elderly
📍
Elderly patients have decreased drug metabolism, increased sensitivity to sedatives, and higher fall risk, requiring lower doses and careful drug selection.
📍
Benzodiazepines and anticholinergics are particularly dangerous, increasing risk of cognitive impairment, delirium, falls, and hip fractures — avoid if possible.
📍
Preferred options include low-dose doxepin (3-6 mg), ramelteon, or orexin antagonists, which have better safety profiles in older adults.
📍
Board pearl: The Beers Criteria lists benzodiazepines and diphenhydramine as potentially inappropriate medications in elderly due to increased risk of adverse events.

Parasomnias and Sleep-Related Behaviors
🔹
Complex sleep behaviors (sleep-walking, sleep-eating, sleep-driving) can occur with all sedative-hypnotics but are most associated with zolpidem, especially at higher doses.
🔹
These behaviors occur during partial arousals from NREM sleep with amnesia for the event — patients have driven cars and prepared meals with no memory.
🔹
Risk factors include concurrent CNS depressants, sleep deprivation, and higher doses — FDA required boxed warnings for all sedative-hypnotics.
🔹
Board pearl: Zolpidem-induced complex sleep behaviors are dose-dependent and more common in women due to slower metabolism — FDA lowered recommended doses for women.

Drug Interactions and Metabolism
⭐
Most benzodiazepines and Z-drugs are metabolized by CYP3A4 — levels increased by inhibitors (ketoconazole, macrolides) and decreased by inducers (rifampin, carbamazepine).
⭐
Lorazepam, oxazepam, and temazepam undergo direct glucuronidation without CYP metabolism, making them safer in liver disease and with fewer drug interactions.
⭐
Combining sedative-hypnotics with other CNS depressants (alcohol, opioids) causes synergistic respiratory depression — major cause of overdose deaths.
⭐
Board pearl: "LOT" drugs (Lorazepam, Oxazepam, Temazepam) are preferred in liver disease because they bypass hepatic oxidation and undergo only conjugation.

Newer Mechanisms and Pipeline Drugs
✅
Selective GABA-A α2/α3 agonists aim to preserve anxiolytic effects while minimizing sedation and amnesia associated with α1 activation.
✅
Orexin-2 selective antagonists may reduce cataplexy in narcolepsy while preserving some wake-promoting function through OX1 receptors.
✅
5-HT2A inverse agonists like pimavanserin may improve sleep without causing sedation or altering sleep architecture.
✅
Board pearl: Drug development focuses on "cleaner" mechanisms that preserve normal sleep architecture while avoiding tolerance, dependence, and next-day impairment.

Clinical Pearls for Insomnia Management
🧠
Chronic insomnia requires addressing underlying causes (depression, anxiety, sleep apnea, medications) before prescribing hypnotics — treat the cause, not just the symptom.
🧠
Cognitive behavioral therapy for insomnia (CBT-I) is first-line treatment with better long-term outcomes than medications and no side effects.
🧠
Hypnotics should be used at lowest effective dose for shortest duration — ideally less than 4 weeks to minimize tolerance and dependence.
🧠
Board pearl: If a patient on chronic benzodiazepines develops new-onset depression, consider medication-induced depression before adding antidepressants.

Specific Clinical Scenarios
⚡
Jet lag: melatonin 0.5-3 mg taken at destination bedtime for eastward travel; bright light exposure in evening for westward travel.
⚡
Shift work disorder: modafinil for wakefulness during work; melatonin or ramelteon for daytime sleep; avoid benzodiazepines due to residual effects.
⚡
REM sleep behavior disorder: clonazepam 0.5-2 mg or melatonin 3-12 mg — benzodiazepines paradoxically effective despite usually suppressing REM.
⚡
Board pearl: High-dose melatonin (10-15 mg) for REM sleep behavior disorder works through different mechanism than low-dose for insomnia — not just sedation.

Overdose and Reversal
📌
Benzodiazepine overdose alone rarely fatal due to ceiling effect on respiratory depression, but often fatal when combined with alcohol or opioids.
📌
Flumazenil competitively antagonizes benzodiazepines at GABA-A receptors but can precipitate seizures in dependent patients — use cautiously.
📌
No reversal agents exist for Z-drugs (despite GABA-A mechanism), barbiturates, or other sedative-hypnotics — supportive care only.
📌
Board pearl: Flumazenil is contraindicated in mixed overdoses involving tricyclic antidepressants or in chronic benzodiazepine users — seizure risk outweighs benefits.

Board Question Stem Patterns
📣
Elderly patient with hip fracture after nighttime fall → benzodiazepine or anticholinergic sleep aid.
📣
Patient reports driving to work with no memory → zolpidem-induced complex sleep behavior.
📣
Chronic benzodiazepine user with new seizures → withdrawal due to missed doses or drug interaction reducing levels.
📣
Shift worker with excessive daytime sleepiness → modafinil preferred over traditional stimulants.
📣
Patient on warfarin with increased INR after starting sleep medication → barbiturate enzyme induction wearing off.
📣
Vivid dreams and poor sleep after stopping alcohol → REM rebound.
📣
Young woman on oral contraceptives needing narcolepsy treatment → modafinil requires backup contraception.

One-Line Recap
🔸
Sleep pharmacology centers on enhancing inhibitory tone (GABA via benzodiazepines/Z-drugs, adenosine blockade) or reducing excitatory drive (antihistamines, orexin antagonists), with key board concepts including receptor mechanisms predicting side effects, age-related considerations, drug interactions through CYP3A4, withdrawal syndromes, and newer agents like orexin antagonists that preserve sleep architecture without dependence potential.

bottom of page

