top of page
Reproductive & Endocrine Systems
Ovarian Follicular Development
Core Principle of Ovarian Follicular Development
🧷
Ovarian follicular development is the cyclical maturation process that transforms primordial follicles containing immature oocytes into mature Graafian follicles capable of ovulation.
🧷
This process integrates oocyte maturation, granulosa and theca cell proliferation, antrum formation, and hormonal responsiveness to create a functional unit for reproduction.
🧷
The journey from primordial to preovulatory follicle takes approximately 85 days, with only the final 14 days being gonadotropin-dependent.
🧷
Board pearl: Most follicular development occurs independent of FSH/LH — only antral to preovulatory stages require gonadotropins.

Primordial Follicle Pool and Activation
📍
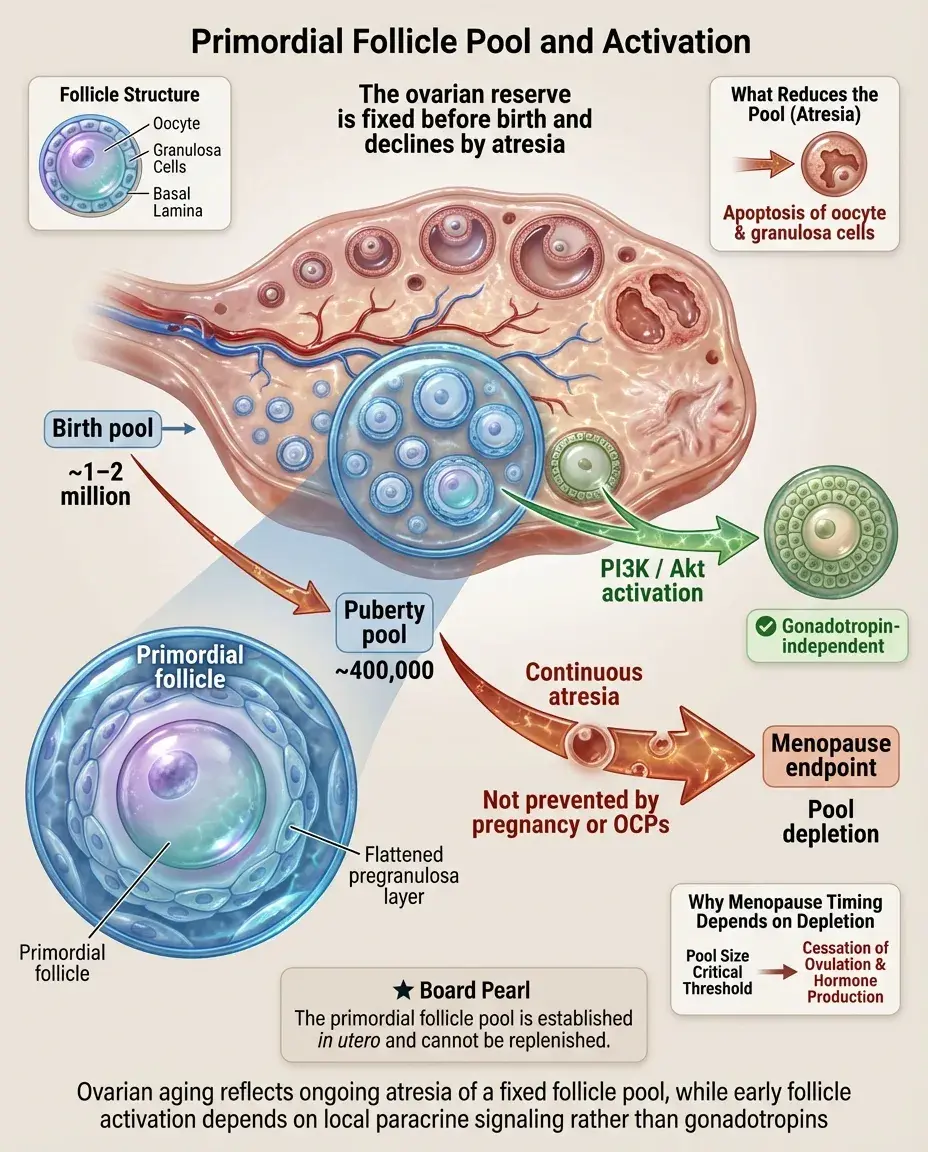
At birth, ovaries contain ~1–2 million primordial follicles, each consisting of an oocyte arrested in meiosis I surrounded by a single layer of flattened pregranulosa cells.
📍
By puberty, this number decreases to ~400,000 through atresia — a continuous process unaffected by pregnancy, OCP use, or ovulation suppression.
📍
Primordial follicle activation occurs through paracrine signals (particularly PI3K/Akt pathway), not gonadotropins, making it a gonadotropin-independent process.
📍
Board pearl: The follicle pool is established in utero and cannot be replenished — depletion defines menopause timing.
Primary Follicle Formation
🔹
Activation transforms primordial follicles into primary follicles through morphologic changes: oocyte enlargement, zona pellucida formation, and conversion of flat pregranulosa cells to cuboidal granulosa cells.
🔹
The zona pellucida is a glycoprotein matrix secreted by the oocyte that will later serve as the binding site for sperm during fertilization.
🔹
Gap junctions form between the oocyte and granulosa cells, establishing bidirectional communication essential for oocyte competence.
🔹
This stage remains gonadotropin-independent, regulated by local factors including GDF9 and BMP15 from the oocyte.

Secondary (Preantral) Follicle Development
⭐
Secondary follicles form when granulosa cells proliferate to create multiple layers around the oocyte, while maintaining the basement membrane (basal lamina) intact.
⭐
Theca cells differentiate from surrounding ovarian stroma, organizing into theca interna (steroidogenic) and theca externa (structural support) layers.
⭐
FSH receptors appear on granulosa cells, but the follicle doesn't yet require FSH for survival — it's in transition to gonadotropin dependence.
⭐
Board pearl: The appearance of theca cells marks the beginning of the two-cell model of steroidogenesis.

Two-Cell Model of Ovarian Steroidogenesis
✅
Theca cells express LH receptors and possess all enzymes needed to convert cholesterol to androgens (17α-hydroxylase/17,20-lyase) but lack aromatase.
✅
Granulosa cells express FSH receptors and aromatase but lack 17α-hydroxylase, so they cannot produce androgens de novo.
✅
LH stimulates theca cells to produce androgens (androstenedione, testosterone) which diffuse to granulosa cells where FSH-induced aromatase converts them to estrogens.
✅
Board pearl: Neither cell type alone can produce estradiol — both are required, explaining why both LH and FSH are necessary for normal ovarian function.

Antral (Tertiary) Follicle Formation
🧠
Antral follicles form when fluid-filled spaces (Call-Exner bodies) coalesce between granulosa cells to create a single antrum containing follicular fluid.
🧠
The oocyte becomes eccentrically positioned, surrounded by specialized granulosa cells called the cumulus oophorus, with the innermost layer forming the corona radiata.
🧠
Follicular fluid contains hormones, growth factors, and hyaluronic acid, creating a specialized microenvironment for oocyte maturation.
🧠
This stage marks full gonadotropin dependence — withdrawal of FSH leads to atresia within days.

Follicular Selection and Dominance
⚡
During early follicular phase, rising FSH rescues a cohort of ~15–20 antral follicles from atresia, initiating the selection process.
⚡
By day 5–7, one follicle emerges as dominant through increased FSH sensitivity via upregulation of FSH receptors and local IGF-1 production.
⚡
The dominant follicle produces inhibin B and estradiol, suppressing FSH below the threshold needed by other follicles, ensuring mono-ovulation.
⚡
Board pearl: The FSH threshold concept explains why most FSH suppression (OCPs, pregnancy) prevents follicular development.

Preovulatory (Graafian) Follicle Maturation
📌
The dominant follicle grows to 18–24 mm diameter, with granulosa cells acquiring LH receptors under estradiol and FSH influence.
📌
Rising estradiol (>200 pg/mL for >50 hours) triggers the LH surge through positive feedback at the hypothalamus and pituitary.
📌
The LH surge induces three critical events: resumption of meiosis I in the oocyte, luteinization of granulosa cells, and rupture of the follicle wall.
📌
Board pearl: LH receptor acquisition by granulosa cells only occurs in the preovulatory follicle, explaining why only the dominant follicle responds to the LH surge.

Ovulation: The Inflammatory Cascade
📣
The LH surge triggers an inflammatory-like reaction: prostaglandin synthesis, protease activation, and vascular changes leading to follicle rupture ~36 hours later.
📣
The oocyte completes meiosis I, forming a secondary oocyte and first polar body, then arrests in metaphase of meiosis II until fertilization.
📣
Cumulus expansion occurs through hyaluronic acid production, creating a gel-like matrix that facilitates oocyte pickup by the fallopian tube.
📣
Board pearl: NSAIDs can inhibit ovulation by blocking prostaglandin synthesis — relevant for emergency contraception timing.

Corpus Luteum Formation and Function
🔸
After ovulation, the collapsed follicle reorganizes into the corpus luteum through luteinization — granulosa and theca cells hypertrophy and vascularize.
🔸
Luteinized cells produce progesterone (predominant), estradiol, and inhibin A, maintaining the endometrium for potential implantation.
🔸
Without hCG from pregnancy, the corpus luteum involutes after 14 days due to falling LH levels and prostaglandin F2α, forming the corpus albicans.
🔸
Board pearl: The 14-day lifespan of the corpus luteum determines luteal phase length — it's remarkably constant across cycles.

Hormonal Feedback Loops Throughout the Cycle
🧷
Early follicular phase: Low estradiol and inhibin B allow FSH rise, recruiting the antral follicle cohort.
🧷
Mid-follicular phase: Rising estradiol and inhibin B from growing follicles suppress FSH (negative feedback), establishing selection.
🧷
Late follicular phase: High estradiol from the dominant follicle switches to positive feedback, triggering the LH surge.
🧷
Luteal phase: Progesterone, estradiol, and inhibin A suppress both LH and FSH, preventing new follicle recruitment.
🧷
Board pearl: The estradiol switch from negative to positive feedback is dose and duration dependent — the key to understanding cycle control.

Follicular Atresia: Programmed Cell Death
📍
>99% of follicles undergo atresia through apoptosis rather than reaching ovulation — this is the default pathway.
📍
Atresia can occur at any stage but predominantly affects antral follicles that fail to acquire sufficient FSH sensitivity.
📍
Molecular markers include granulosa cell apoptosis (caspase activation), basement membrane breakdown, and oocyte degeneration.
📍
Androgens promote atresia while FSH prevents it through anti-apoptotic signaling (PI3K/Akt pathway activation).
📍
Board clue: In PCOS, excess intraovarian androgens promote premature follicular atresia, contributing to anovulation.

Oocyte Maturation and Competence
🔹
Nuclear maturation involves progression from prophase I arrest (germinal vesicle stage) through meiosis I completion at ovulation.
🔹
Cytoplasmic maturation includes organelle redistribution, mRNA accumulation, and protein synthesis necessary for fertilization and early embryogenesis.
🔹
The oocyte directs follicular development through paracrine factors (GDF9, BMP15) that regulate granulosa cell proliferation and differentiation.
🔹
Board pearl: Oocyte quality, not just chromosomal normalcy, determines fertilization success — explaining age-related fertility decline beyond aneuploidy.

Clinical Correlations: Ovulation Induction
⭐
Clomiphene citrate blocks estrogen receptors at hypothalamus → perceived low estrogen → increased GnRH/FSH → multiple follicle development.
⭐
Letrozole inhibits aromatase → reduced estradiol → increased FSH through reduced negative feedback.
⭐
Exogenous gonadotropins directly stimulate multiple follicles, bypassing endogenous feedback — requires monitoring to prevent ovarian hyperstimulation.
⭐
Board distinction: Clomiphene may thin endometrium (anti-estrogenic effect) while letrozole does not — relevant for implantation.

Ovarian Reserve and Aging
✅
Ovarian reserve reflects the quantity and quality of remaining follicles, declining with age due to continuous atresia and accumulated oocyte damage.
✅
Day 3 FSH elevation indicates diminished reserve — fewer follicles produce less inhibin B, requiring higher FSH for recruitment.
✅
Anti-Müllerian hormone (AMH), produced by preantral and small antral follicles, provides a gonadotropin-independent marker of reserve.
✅
Board pearl: AMH doesn't fluctuate with cycle day unlike FSH — can be measured anytime for reserve assessment.

Pathologic Disruptions of Folliculogenesis
🧠
PCOS: Arrested development at small antral stage due to hyperandrogenism and abnormal FSH:LH ratio → multiple small follicles on ultrasound.
🧠
Hypothalamic amenorrhea: Insufficient GnRH → low FSH/LH → no follicle recruitment beyond preantral stage.
🧠
Premature ovarian insufficiency: Accelerated follicle depletion → elevated FSH, low AMH before age 40.
🧠
Hyperprolactinemia: Suppresses GnRH → low gonadotropins → arrested follicular development.
🧠
Board clue: Follicle arrest stage helps identify the disruption level in the HPO axis.

Ultrasound Monitoring of Follicular Development
⚡
Follicles become visible on transvaginal ultrasound at ~4–5 mm (early antral stage).
⚡
Growth rate is ~1–2 mm/day once selection occurs, reaching 18–24 mm at maturity.
⚡
Multiple follicles 10–14 mm suggest PCOS; single dominant follicle >15 mm with others <10 mm indicates normal selection.
⚡
Endometrial thickness correlates with estradiol production — trilaminar pattern and >7 mm thickness suggest adequate estrogenization.
⚡
Board pearl: Follicle size predicts maturity better than estradiol levels — 18 mm is the minimum for ovulation trigger.

Molecular Markers and Growth Factors
📌
FSH induces aromatase, LH receptors, and inhibin production in granulosa cells through cAMP signaling.
📌
IGF-1 amplifies FSH action, with bioavailability regulated by IGF binding proteins that are modulated by follicular androgens.
📌
Activins promote FSH receptor expression while inhibins suppress FSH secretion — creating local and systemic feedback loops.
📌
VEGF increases during follicular growth, preparing for corpus luteum vascularization post-ovulation.
📌
Board pearl: Growth factor dysregulation (particularly IGF and VEGF systems) contributes to PCOS and ovarian hyperstimulation pathophysiology.

Board Question Stem Patterns
📣
Day 3 FSH of 15 mIU/mL in a 38-year-old trying to conceive → diminished ovarian reserve.
📣
Multiple 8–10 mm follicles with elevated LH:FSH ratio → PCOS with arrested follicular development.
📣
Failure to menstruate after progesterone withdrawal with low FSH → hypothalamic/pituitary dysfunction preventing follicle development.
📣
Enlarged multicystic ovaries during fertility treatment → ovarian hyperstimulation from excessive follicular response.
📣
High estradiol with single 22 mm follicle → imminent ovulation, LH surge expected.
📣
Post-pill amenorrhea with low FSH and normal prolactin → hypothalamic suppression, not ovarian failure.
📣
AMH <1.0 ng/mL in a 35-year-old → low ovarian reserve despite regular cycles.

One-Line Recap
🔸
Ovarian follicular development transforms primordial follicles through primary, secondary, and antral stages via coordinated oocyte-granulosa-theca cell interactions, with FSH/LH dependence emerging at the antral stage, culminating in dominant follicle selection, LH surge-triggered ovulation, and corpus luteum formation through an intricate interplay of endocrine feedback and paracrine growth factors.

bottom of page

