top of page
Cardiovascular System
Microscopic anatomy of myocardium, endocardium, pericardium
Core Principle of Cardiac Histology
🧷
The heart wall consists of three distinct layers — endocardium (inner), myocardium (middle), and epicardium (outer) — each with specialized structure matching its function.
🧷
The myocardium contains cardiac muscle cells (cardiomyocytes) that generate contractile force, the endocardium provides a non-thrombogenic surface for blood flow, and the epicardium (visceral pericardium) forms the protective outer covering.
🧷
Understanding the microscopic anatomy explains cardiac pathology: endocarditis affects the endocardium, myocardial infarction damages the myocardium, and pericarditis inflames the pericardium.
🧷
Board pearl: Questions often show histologic images asking you to identify the layer and predict which disease process would affect it.

Cardiomyocyte Structure and Function
📍
Cardiac muscle cells are striated, branched, and connected end-to-end by intercalated discs — the defining histologic feature distinguishing cardiac from skeletal muscle.
📍
Each cardiomyocyte typically contains 1-2 centrally located nuclei (unlike multinucleated skeletal muscle fibers with peripheral nuclei).
📍
The striation pattern results from organized sarcomeres containing actin (thin) and myosin (thick) filaments arranged identically to skeletal muscle.
📍
Cardiomyocytes are rich in mitochondria (30-40% of cell volume) reflecting their enormous energy demands and obligate aerobic metabolism.
📍
Board clue: Central nuclei + striations + branching + intercalated discs = cardiac muscle.

Intercalated Discs: The Cardiac Syncytium
🔹
Intercalated discs are specialized cell-cell junctions appearing as dark, irregular transverse lines between cardiomyocytes under light microscopy.
🔹
Three junction types comprise each disc: gap junctions (electrical coupling), desmosomes (mechanical adhesion), and adherens junctions (anchoring actin filaments).
🔹
Gap junctions contain connexin proteins (especially connexin 43) that form channels allowing rapid spread of action potentials between cells.
🔹
This electrical coupling creates a functional syncytium — the entire myocardium contracts as a coordinated unit.
🔹
Board pearl: Mutations in connexin proteins cause arrhythmogenic cardiomyopathy due to disrupted electrical conduction.

Myocardial Architecture and Fiber Orientation
⭐
Myocardial fibers are arranged in complex spiraling patterns — not simple concentric layers — optimizing ejection fraction through a twisting motion during systole.
⭐
The ventricular myocardium has three layers: subendocardial (longitudinal fibers), middle (circular fibers), and subepicardial (oblique fibers).
⭐
Atrial myocardium is thinner (2-3 mm) than ventricular myocardium (left ventricle 10-15 mm) reflecting lower pressure generation requirements.
⭐
Specialized conduction system cardiomyocytes (Purkinje fibers) are larger, paler, and contain more glycogen but fewer myofibrils than working cardiomyocytes.
⭐
Board distinction: Purkinje fibers stain lighter than regular cardiomyocytes due to glycogen content.

Endocardium: The Heart's Inner Lining
✅
The endocardium consists of three layers from lumen to myocardium: endothelium, subendothelial connective tissue, and subendocardium.
✅
The endothelial layer is continuous with vascular endothelium and provides the critical non-thrombogenic surface preventing intracardiac clot formation.
✅
Subendothelial connective tissue contains elastic and collagen fibers plus scattered smooth muscle cells.
✅
The subendocardium houses the Purkinje fibers of the conduction system and small blood vessels.
✅
Board pearl: Endocarditis vegetation appears as fibrin, platelets, bacteria, and inflammatory cells adherent to damaged endocardium — not normal tissue.

Valvular Histology
🧠
Cardiac valves are avascular structures consisting of a dense fibrous core (fibrosa) covered by endocardium on both surfaces.
🧠
The atrioventricular valves (mitral, tricuspid) have three layers: atrialis (elastic fibers), spongiosa (proteoglycans), and fibrosa (dense collagen).
🧠
Semilunar valves (aortic, pulmonic) are thinner with a similar three-layer structure optimized for high-pressure closure.
🧠
Valve leaflets contain interstitial cells that maintain the extracellular matrix but lack blood vessels — nutrition comes via diffusion from surrounding blood.
🧠
Board clue: Rheumatic heart disease shows Aschoff bodies (granulomas with giant cells) in the myocardium, not the valve itself.

Epicardium and Pericardial Anatomy
⚡
The epicardium (visceral pericardium) is the heart's outermost layer, consisting of mesothelium (simple squamous epithelium) overlying fibroelastic connective tissue.
⚡
Epicardial adipose tissue accumulates along coronary vessels and the atrioventricular groove, serving metabolic and mechanical functions.
⚡
The parietal pericardium mirrors the epicardium structurally — mesothelium facing the pericardial cavity backed by fibrous connective tissue.
⚡
The pericardial cavity normally contains 15-50 mL of serous fluid serving as lubricant.
⚡
Board distinction: Pericardial effusion accumulates between visceral and parietal layers; hemopericardium specifically indicates blood in this space.

Cardiac Capillaries and Microcirculation
📌
The myocardium has the highest capillary density of any tissue — approximately one capillary per cardiomyocyte ensuring adequate oxygen delivery.
📌
Cardiac capillaries are continuous (non-fenestrated) with tight junctions, forming a blood-heart barrier analogous to the blood-brain barrier.
📌
The capillary-to-cardiomyocyte ratio approaches 1:1, with intercapillary distance rarely exceeding 20 μm (the diffusion limit for oxygen).
📌
Coronary capillaries have prominent pericyte coverage providing structural support and regulating blood flow.
📌
Board pearl: Myocardial ischemia occurs when oxygen demand exceeds supply — the high baseline extraction means little reserve exists.

Cardiac Extracellular Matrix
📣
The cardiac ECM consists primarily of type I collagen (85%) and type III collagen (11%), forming a structural scaffold supporting cardiomyocytes.
📣
This collagenous network prevents overstretching during diastole and coordinates force transmission during systole.
📣
Cardiac fibroblasts are the most numerous cell type in the heart (though cardiomyocytes occupy more volume) and produce the ECM components.
📣
Matrix metalloproteinases (MMPs) and their tissue inhibitors (TIMPs) regulate ECM turnover — imbalance leads to pathologic remodeling.
📣
Board clue: Post-MI remodeling involves fibroblast activation → excessive collagen deposition → ventricular stiffness and dysfunction.

Specialized Conduction System Histology
🔸
The sinoatrial (SA) node contains small, pale cardiomyocytes with few myofibrils embedded in dense connective tissue near the superior vena cava-right atrial junction.
🔸
Atrioventricular (AV) node cells are similar but more densely packed, located in the interatrial septum near the coronary sinus opening.
🔸
Bundle of His cells transition to Purkinje fibers — large, pale cells with peripheral myofibrils and central glycogen-rich cytoplasm.
🔸
Purkinje fibers have the fastest conduction velocity (2-4 m/s) due to abundant gap junctions and large cell diameter.
🔸
Board distinction: Conduction system cells stain pale with PAS due to glycogen; working myocardium stains dark.

Age-Related Cardiac Histologic Changes
🧷
Lipofuscin (wear-and-tear pigment) accumulates in cardiomyocytes with age, appearing as golden-brown perinuclear granules.
🧷
Cardiac amyloidosis shows eosinophilic, acellular deposits in the interstitium that stain with Congo red and show apple-green birefringence.
🧷
Basophilic degeneration represents accumulation of glycogen and mucopolysaccharides in aging cardiomyocytes.
🧷
The number of cardiomyocytes decreases with age while remaining cells hypertrophy — total muscle mass is maintained.
🧷
Board pearl: Lipofuscin is a normal aging finding; amyloid deposition is always pathologic and causes restrictive cardiomyopathy.

Myocardial Hypertrophy Patterns
📍
Concentric hypertrophy (pressure overload) shows increased cardiomyocyte diameter with parallel addition of sarcomeres — wall thickness increases, chamber size normal or decreased.
📍
Eccentric hypertrophy (volume overload) shows increased cardiomyocyte length with series addition of sarcomeres — chamber dilation with proportional wall thickening.
📍
Hypertrophied cardiomyocytes develop enlarged, hyperchromatic, irregularly shaped nuclei (boxcar nuclei).
📍
Myocyte diameter normally 10-20 μm can increase to 30-40 μm in hypertrophy.
📍
Board clue: Hypertension → pressure overload → concentric hypertrophy; mitral regurgitation → volume overload → eccentric hypertrophy.

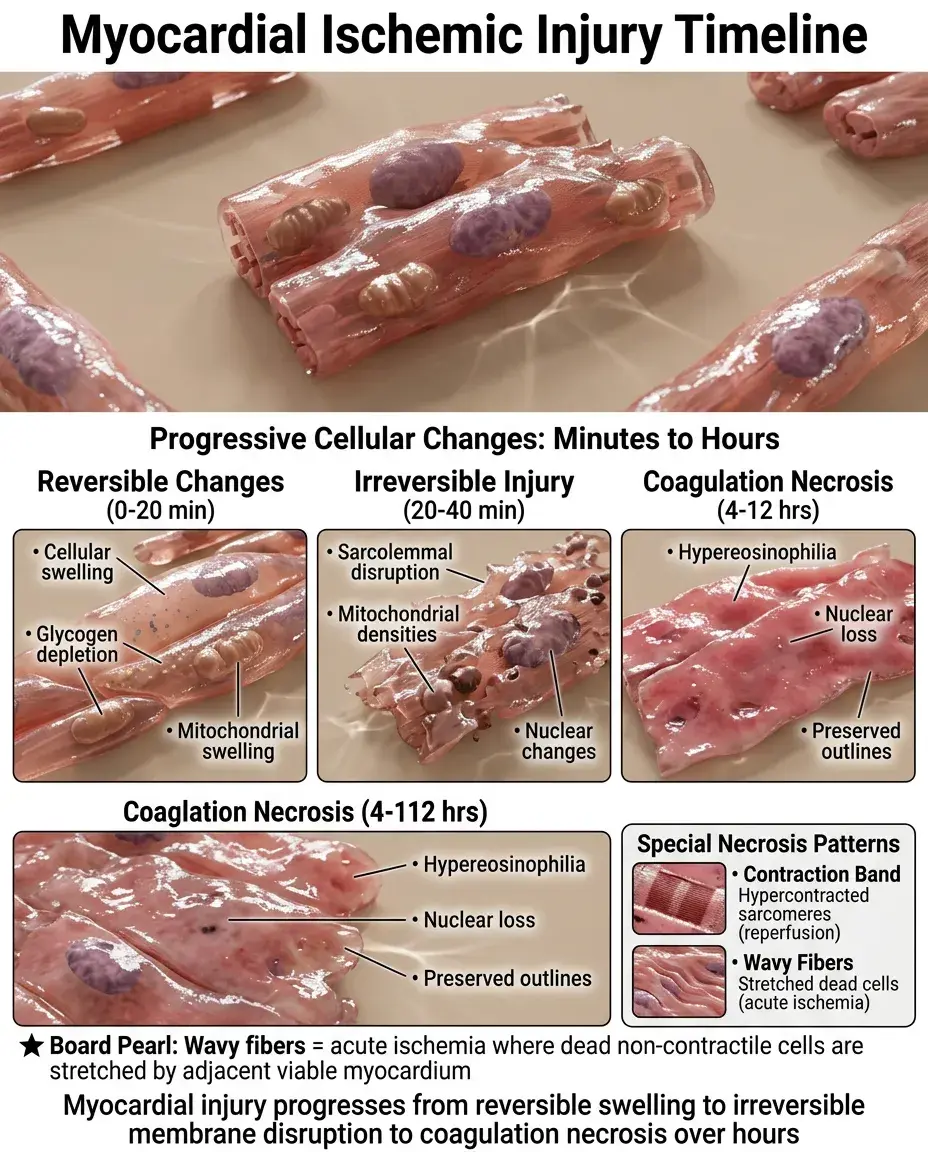
Pathologic Myocardial Changes
🔹
Myocardial ischemia first causes reversible changes: cellular swelling, glycogen depletion, and mitochondrial swelling within minutes.
🔹
Irreversible injury appears by 20-40 minutes: sarcolemmal disruption, mitochondrial amorphous densities, and nuclear changes.
🔹
Coagulation necrosis becomes visible by 4-12 hours with hypereosinophilia, loss of nuclei, and preserved cell outlines.
🔹
Contraction band necrosis shows hypercontracted sarcomeres — indicates reperfusion injury or catecholamine excess.
🔹
Board pearl: Wavy fibers indicate acute ischemia where non-contractile dead cells are stretched by adjacent viable myocardium.
Inflammatory Cell Patterns in Cardiac Disease
⭐
Acute myocarditis shows lymphocytic infiltration (usually T cells) with myocyte necrosis — viral etiology most common.
⭐
Giant cell myocarditis displays multinucleated giant cells with extensive myocyte necrosis — poor prognosis, consider transplant.
⭐
Hypersensitivity myocarditis shows eosinophilic infiltration without myocyte necrosis — drug reaction until proven otherwise.
⭐
Sarcoidosis produces non-caseating granulomas preferentially affecting the basal septum and conduction system.
⭐
Board distinction: Lymphocytes + myocyte necrosis = viral myocarditis; eosinophils without necrosis = drug reaction.

Endocardial Pathology Patterns
✅
Acute rheumatic carditis shows Anitschkow cells (activated macrophages with caterpillar nuclei) in Aschoff bodies within myocardium.
✅
Chronic rheumatic heart disease causes endocardial thickening with neovascularization — MacCallum plaques in the left atrium.
✅
Infective endocarditis vegetations contain fibrin, platelets, bacteria, and neutrophils — destroy underlying valve architecture.
✅
Marantic endocarditis (NBTE) shows sterile fibrin-platelet vegetations on structurally normal valves — associated with malignancy.
✅
Board clue: Vegetation + bacteria + neutrophils = infective; sterile vegetation + cancer = marantic.

Pericardial Histopathology
🧠
Acute pericarditis shows neutrophilic infiltration, fibrin deposition, and vascular congestion of both pericardial layers.
🧠
Chronic pericarditis displays lymphocytes, plasma cells, and progressive fibrosis potentially leading to constrictive physiology.
🧠
Fibrinous pericarditis creates a bread-and-butter appearance grossly due to fibrin strands between visceral and parietal layers.
🧠
Hemorrhagic pericarditis indicates blood vessel involvement — consider malignancy, tuberculosis, or uremia.
🧠
Board pearl: Pericardial fluid cytology showing malignant cells confirms neoplastic pericarditis — often adenocarcinoma.

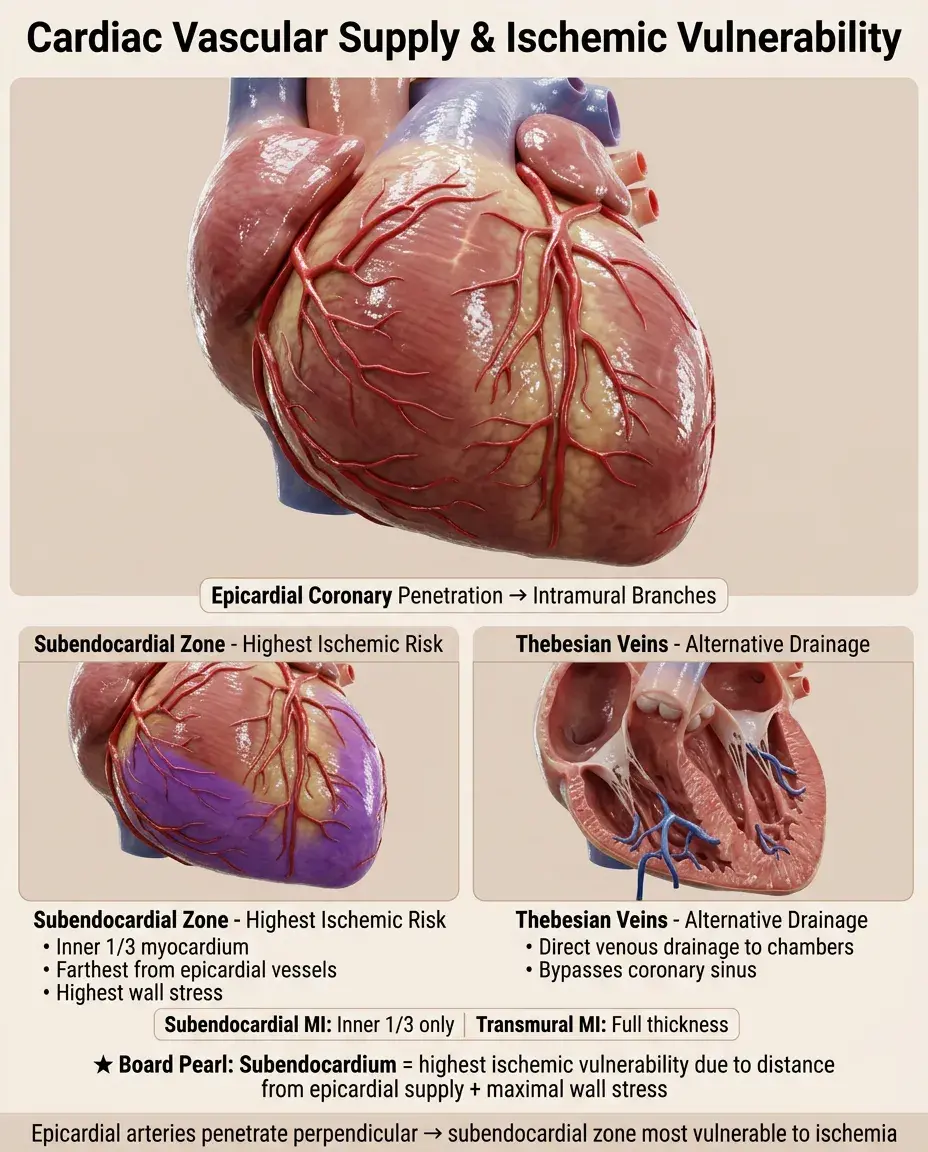
Vascular Supply of Cardiac Layers
⚡
The myocardium receives blood from epicardial coronary arteries that penetrate perpendicularly as intramural branches.
⚡
The inner third of myocardium (subendocardium) is most vulnerable to ischemia — farthest from epicardial vessels with highest wall stress.
⚡
Thebesian veins drain directly into cardiac chambers, providing an alternative venous drainage route.
⚡
The endocardium and inner myocardium can receive some oxygen directly from ventricular blood — insufficient during stress.
⚡
Board distinction: Subendocardial infarction affects inner third only; transmural infarction involves full thickness.
Immunohistochemical Markers
📌
Troponin I and T specifically identify cardiomyocytes — nuclear and cytoplasmic staining confirms cardiac origin of cells.
📌
Desmin marks the intermediate filaments of all muscle types; myoglobin is similarly non-specific for cardiac muscle.
📌
CD31 and von Willebrand factor identify endothelial cells lining the endocardium and blood vessels.
📌
Smooth muscle actin marks myofibroblasts activated during cardiac remodeling and fibrosis.
📌
Board pearl: A spindle cell tumor staining positive for troponin = cardiac rhabdomyoma or rhabdomyosarcoma.

Board Question Stem Patterns
📣
Branched cells with central nuclei and intercalated discs → cardiac muscle identification.
📣
Pale cells with peripheral myofibrils in the subendocardium → Purkinje fibers of conduction system.
📣
Golden-brown perinuclear pigment in cardiomyocytes → lipofuscin accumulation with aging.
📣
Eosinophilic interstitial deposits with apple-green birefringence → cardiac amyloidosis.
📣
Lymphocytes and myocyte necrosis in a young patient with viral prodrome → viral myocarditis.
📣
Hypercontracted sarcomeres with transverse bands → contraction band necrosis from reperfusion.
📣
Fibrin-platelet vegetation without bacteria in cancer patient → marantic endocarditis.
📣
Bread-and-butter pericardium in uremic patient → fibrinous pericarditis.

One-Line Recap
🔸
Cardiac histology centers on three layers — endocardium (endothelial lining), myocardium (branched striated cardiomyocytes connected by intercalated discs), and epicardium (mesothelial covering) — with pathology patterns including myocyte necrosis in ischemia, inflammatory infiltrates in myocarditis, vegetations in endocarditis, and fibrin deposition in pericarditis that correlate with clinical presentations tested on boards.

bottom of page

