top of page
Reproductive & Endocrine Systems
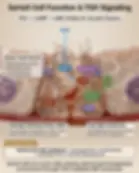
Male Reproductive Hormone Regulation: Leydig and Sertoli Cells
Core Principle of Male Reproductive Hormone Regulation
🧷
The hypothalamic-pituitary-testicular axis orchestrates male reproductive function through a negative feedback loop involving GnRH, LH, FSH, testosterone, and inhibin.
🧷
Two distinct testicular cell populations respond to pituitary signals: Leydig cells (in the interstitium) produce testosterone in response to LH, while Sertoli cells (within seminiferous tubules) support spermatogenesis in response to FSH.
🧷
This dual-cell system separates hormone production from gamete production, allowing independent regulation of masculinization and fertility.
🧷
Board pearl: LH acts on Leydig cells → testosterone; FSH acts on Sertoli cells → spermatogenesis support.

Leydig Cell Function and LH Signaling
📍
Leydig cells are the testosterone factories of the testes, located in the interstitial space between seminiferous tubules.
📍
LH binds to G-protein coupled receptors on Leydig cells → cAMP activation → StAR protein expression → cholesterol transport into mitochondria → testosterone synthesis.
📍
Testosterone production follows a pulsatile pattern matching LH pulses, with highest levels in early morning (circadian rhythm).
📍
Leydig cells express all enzymes needed for testosterone synthesis: cholesterol → pregnenolone → 17-hydroxypregnenolone → DHEA → androstenedione → testosterone.
📍
Board pearl: Leydig cell tumors present with precocious puberty in boys or feminization in adults due to excess testosterone/estrogen production.

Sertoli Cell Function and FSH Signaling
🔹
Sertoli cells are the "nurse cells" of spermatogenesis, forming the blood-testis barrier and creating the specialized microenvironment for developing sperm.
🔹
FSH binds to receptors on basolateral Sertoli cell membrane → cAMP signaling → production of androgen-binding protein (ABP), inhibin B, and growth factors.
🔹
ABP concentrates testosterone within seminiferous tubules to levels 100× higher than serum — essential for spermatogenesis.
🔹
Sertoli cells also produce anti-Müllerian hormone (AMH) during fetal development, causing regression of Müllerian ducts.
🔹
Board pearl: Sertoli cell-only syndrome presents with azoospermia, small testes, elevated FSH, but normal testosterone and virilization.
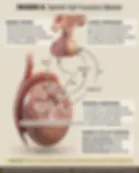
The Blood-Testis Barrier
⭐
Tight junctions between adjacent Sertoli cells create an immunologically privileged site protecting developing sperm from autoimmune attack.
⭐
The barrier divides the seminiferous epithelium into basal (spermatogonia) and adluminal (meiotic and post-meiotic cells) compartments.
⭐
Spermatocytes must traverse the barrier during development — Sertoli cells form new junctions before breaking old ones, maintaining barrier integrity.
⭐
Barrier disruption (trauma, infection, vasectomy reversal) can expose sperm antigens → antisperm antibodies → infertility.
⭐
Board pearl: Mumps orchitis in postpubertal males can breach the blood-testis barrier → autoimmune orchitis → infertility.

Testosterone Synthesis Pathway
✅
Cholesterol → pregnenolone (rate-limiting step via CYP11A1/side chain cleavage enzyme).
✅
Pregnenolone → 17-hydroxypregnenolone → DHEA (via CYP17A1 with 17α-hydroxylase and 17,20-lyase activity).
✅
DHEA → androstenedione → testosterone (via 3β-HSD and 17β-HSD).
✅
Testosterone → dihydrotestosterone (DHT) via 5α-reductase in peripheral tissues — DHT is the more potent androgen.
✅
Testosterone → estradiol via aromatase in adipose tissue, bone, and brain.
✅
Board pearl: 5α-reductase deficiency presents with ambiguous genitalia at birth but virilization at puberty when testosterone levels surge.

Negative Feedback Mechanisms
🧠
Testosterone inhibits GnRH release at the hypothalamus and LH release at the pituitary → classic negative feedback.
🧠
Inhibin B (produced by Sertoli cells) selectively inhibits FSH release without affecting LH — allows independent control of spermatogenesis.
🧠
Estradiol (aromatized from testosterone) provides additional negative feedback, particularly important in obesity where increased aromatase activity suppresses gonadotropins.
🧠
Feedback operates at both tonic (basal) and surge (not applicable in males) levels.
🧠
Board pearl: Exogenous testosterone suppresses LH/FSH → testicular atrophy and azoospermia (mechanism of male hormonal contraception).

Paracrine Interactions Between Leydig and Sertoli Cells
⚡
Testosterone from Leydig cells diffuses to adjacent Sertoli cells where it binds androgen receptors → essential for spermatogenesis.
⚡
Sertoli cells cannot produce testosterone but require high local concentrations (via ABP) to support germ cell development.
⚡
Sertoli cells produce growth factors (IGF-1, TGF-β) that enhance Leydig cell steroidogenesis — bidirectional communication.
⚡
This paracrine system ensures coordinated function: hormone production supports spermatogenesis, while spermatogenesis signals modulate hormone production.
⚡
Board pearl: Androgen insensitivity syndrome affects Sertoli cells → impaired spermatogenesis despite normal Leydig cell testosterone production.

Inhibin B as a Marker of Sertoli Cell Function
📌
Inhibin B is a glycoprotein hormone produced exclusively by Sertoli cells in response to FSH stimulation.
📌
Serum inhibin B levels correlate with Sertoli cell mass and spermatogenic activity — low levels indicate Sertoli cell dysfunction.
📌
Inhibin B provides more specific feedback than testosterone: selectively suppresses FSH without affecting LH.
📌
In infertility workup: low inhibin B with high FSH suggests primary testicular failure affecting Sertoli cells.
📌
Board pearl: Inhibin B is undetectable in Sertoli cell-only syndrome but may be normal in maturation arrest or hypospermatogenesis.
Hormonal Changes Through Male Development
📣
Fetal: testosterone surge (weeks 8-12) drives male differentiation; AMH from Sertoli cells causes Müllerian duct regression.
📣
Mini-puberty (2-6 months): transient gonadotropin rise → testosterone surge → penile growth and Leydig cell proliferation.
📣
Childhood: quiescent HPT axis with low gonadotropins and testosterone ("juvenile pause").
📣
Puberty: nocturnal GnRH pulses → LH/FSH rise → Leydig cell proliferation → testosterone surge → virilization and spermatogenesis initiation.
📣
Aging: gradual testosterone decline (1-2% per year after 30), but no true "andropause" — unlike abrupt menopause.

Clinical Assessment of the HPT Axis
🔸
Morning testosterone (8-10 AM) — accounts for diurnal variation; repeat if low.
🔸
LH and FSH levels distinguish primary (high gonadotropins) from secondary (low/normal gonadotropins) hypogonadism.
🔸
Semen analysis assesses Sertoli cell function indirectly through sperm production.
🔸
GnRH stimulation test evaluates pituitary reserve; hCG stimulation test evaluates Leydig cell reserve.
🔸
Board pearl: Young man with low testosterone, low LH/FSH, and anosmia → Kallmann syndrome (GnRH deficiency with olfactory bulb agenesis).

Primary vs Secondary Hypogonadism
🧷
Primary (hypergonadotropic): testicular failure → low testosterone, high LH/FSH. Causes include Klinefelter syndrome, chemotherapy, radiation, trauma, infections.
🧷
Secondary (hypogonadotropic): hypothalamic/pituitary failure → low testosterone, low/normal LH/FSH. Causes include Kallmann syndrome, pituitary tumors, hyperprolactinemia, severe obesity.
🧷
Primary affects both Leydig and Sertoli cells → low testosterone and impaired spermatogenesis.
🧷
Secondary can be treated with gonadotropins or pulsatile GnRH to restore fertility; primary requires testosterone replacement only.
🧷
Board pearl: Klinefelter syndrome (47,XXY) is the most common cause of primary hypogonadism — small firm testes, gynecomastia, elevated gonadotropins.

Androgen Receptor Distribution and Function
📍
Androgen receptors are nuclear receptors present in Sertoli cells, Leydig cells, and throughout the body.
📍
In Sertoli cells: essential for spermatogenesis, blood-testis barrier maintenance, and germ cell survival.
📍
In Leydig cells: provides intratesticular feedback to modulate testosterone production.
📍
Testosterone must be converted to DHT by 5α-reductase for full activity in external genitalia and prostate.
📍
Board pearl: Complete androgen insensitivity (testicular feminization) — 46,XY with female external genitalia, absent uterus/fallopian tubes (AMH effect preserved), cryptorchid testes.

Cryptorchidism and Temperature Regulation
🔹
Normal spermatogenesis requires scrotal temperature 2-3°C below core body temperature.
🔹
Cryptorchid testes remain at body temperature → Leydig cells function normally (testosterone production preserved) but Sertoli cells/spermatogenesis severely impaired.
🔹
Prolonged cryptorchidism → progressive seminiferous tubule damage, reduced fertility even after orchiopexy.
🔹
Increased risk of testicular cancer persists even after surgical correction.
🔹
Board pearl: Unilateral cryptorchidism → normal puberty/virilization (contralateral testis compensates) but increased cancer risk in both testes.
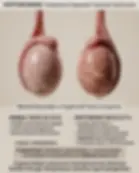
Varicocele Effects on Testicular Function
⭐
Varicocele (dilated pampiniform plexus) impairs testicular temperature regulation → bilateral testicular dysfunction despite being usually left-sided.
⭐
Proposed mechanisms: increased scrotal temperature, reflux of adrenal/renal metabolites, hypoxia.
⭐
Progressive Leydig cell dysfunction → suboptimal testosterone; Sertoli cell dysfunction → impaired spermatogenesis.
⭐
Most common correctable cause of male infertility; repair improves semen parameters in 70% of cases.
⭐
Board pearl: "Bag of worms" on standing examination that decreases when supine, more common on left due to venous anatomy.

Hormonal Control of Sexual Function
✅
Testosterone is essential for libido but not for erection — men with very low testosterone can still achieve erections with adequate stimulation.
✅
Testosterone maintains nitric oxide synthase expression in penile tissue → facilitates erectile function.
✅
FSH/LH have no direct role in sexual function — only indirect effects through testosterone production.
✅
Prolactin elevation inhibits GnRH → low testosterone → decreased libido and erectile dysfunction.
✅
Board pearl: First-line erectile dysfunction treatment (PDE5 inhibitors) works independently of testosterone levels — don't need to check testosterone before prescribing.

Gynecomastia and Estrogen/Androgen Balance
🧠
Gynecomastia results from increased estrogen/androgen ratio — either increased estrogen or decreased androgen action.
🧠
Physiologic: neonatal (maternal estrogens), pubertal (transient E/T imbalance), elderly (decreased T, increased aromatase).
🧠
Pathologic causes: hypogonadism (Klinefelter), estrogen excess (obesity, aromatase upregulation), androgen resistance, drugs.
🧠
Leydig cell tumors can produce excess estrogen → rapid-onset gynecomastia in adults.
🧠
Board pearl: Unilateral firm breast mass in adolescent male → pubertal gynecomastia (not breast cancer); bilateral in obese adult → increased peripheral aromatization.

Anabolic Steroid Effects on HPT Axis
⚡
Exogenous androgens suppress GnRH/LH/FSH through negative feedback → testicular atrophy.
⚡
Leydig cells atrophy from lack of LH stimulation → loss of intratesticular testosterone production.
⚡
Sertoli cells dysfunction from lack of FSH and low intratesticular testosterone → azoospermia.
⚡
Recovery after cessation is variable — can take 6-12 months; some have permanent dysfunction.
⚡
Board pearl: Bodybuilder with small soft testes, low LH/FSH, low endogenous testosterone but muscular appearance → exogenous androgen use.

Endocrine Disruptors and Testicular Function
📌
Environmental chemicals (phthalates, BPA, pesticides) can interfere with testicular development and function.
📌
Mechanisms include androgen receptor antagonism, aromatase modulation, and direct Leydig/Sertoli cell toxicity.
📌
Fetal/neonatal exposure has greater impact than adult exposure — critical windows for reproductive programming.
📌
Associated with cryptorchidism, hypospadias, reduced sperm counts, and testicular dysgenesis syndrome.
📌
Board pearl: Declining sperm counts in industrialized nations over past 50 years may reflect cumulative endocrine disruptor exposure.

Board Question Stem Patterns
📣
Small firm testes + gynecomastia + elevated LH/FSH → Klinefelter syndrome (47,XXY).
📣
Low testosterone + low LH/FSH + anosmia → Kallmann syndrome.
📣
Ambiguous genitalia at birth + virilization at puberty → 5α-reductase deficiency.
📣
Azoospermia + normal testosterone + elevated FSH → Sertoli cell-only syndrome or maturation arrest.
📣
Precocious puberty + testicular mass → Leydig cell tumor.
📣
Female phenotype + 46,XY + absent uterus → androgen insensitivity syndrome.
📣
Infertility + varicocele + oligospermia → varicocele-induced testicular dysfunction.

One-Line Recap
🔸
The male HPT axis coordinates testosterone production by LH-stimulated Leydig cells with spermatogenesis support by FSH-stimulated Sertoli cells, using testosterone and inhibin B negative feedback to maintain hormonal homeostasis and fertility, with disruptions causing distinct patterns of hypogonadism, infertility, and developmental anomalies recognizable by specific hormone profiles.

bottom of page

