top of page
Reproductive & Endocrine Systems
Hypoglycemia mechanisms
Core Principle of Hypoglycemia
🧷
Hypoglycemia occurs when glucose supply fails to meet tissue demand, threatening the obligate glucose-consuming brain.
🧷
Normal glucose homeostasis requires balance between hepatic glucose production (glycogenolysis and gluconeogenesis), peripheral glucose utilization, and hormonal regulation.
🧷
The body's defense against hypoglycemia operates through a hierarchical counter-regulatory response: decreased insulin secretion at ~80 mg/dL, glucagon release at ~70 mg/dL, epinephrine surge at ~70 mg/dL, and cortisol/growth hormone at ~65 mg/dL.
🧷
When glucose falls below 55 mg/dL, neuroglycopenic symptoms emerge as the brain exhausts its minimal glycogen reserves.
🧷
Board pearl: The threshold for symptoms varies with glycemic control — diabetics with chronic hyperglycemia may experience symptoms at higher glucose levels.

The Counter-Regulatory Hormone Cascade
📍
As glucose falls, pancreatic α-cells sense the decline and release glucagon — the primary defense against hypoglycemia in healthy individuals.
📍
Glucagon stimulates hepatic glycogenolysis within minutes and gluconeogenesis within hours, while inhibiting glycolysis.
📍
Epinephrine provides backup defense through β₂-adrenergic stimulation of hepatic glucose production and α-adrenergic inhibition of insulin secretion.
📍
Cortisol and growth hormone act more slowly (hours) to increase gluconeogenesis and decrease peripheral glucose utilization.
📍
Board pearl: In type 1 diabetes, both glucagon and epinephrine responses to hypoglycemia become impaired over time, creating "hypoglycemia unawareness.

Insulin-Mediated Hypoglycemia Mechanisms
🔹
Exogenous insulin excess (most common cause): mismatched insulin dose to carbohydrate intake, exercise, or renal function.
🔹
Endogenous hyperinsulinism: insulinoma (β-cell tumor), nesidioblastosis (β-cell hyperplasia), autoimmune hypoglycemia (insulin receptor antibodies), or sulfonylurea-induced.
🔹
Insulin drives glucose into cells via GLUT4 translocation in muscle/adipose tissue, while simultaneously suppressing hepatic glucose output.
🔹
The diagnostic hallmark is inappropriately elevated insulin and C-peptide during hypoglycemia (except in exogenous insulin where C-peptide is suppressed).
🔹
Board distinction: Elevated insulin with low C-peptide = exogenous insulin; elevated insulin with elevated C-peptide = endogenous source.

Non-Insulin-Mediated Hypoglycemia: Substrate Deficiency
⭐
Critical illness hypoglycemia results from depleted glycogen stores plus impaired gluconeogenesis — seen in sepsis, liver failure, renal failure.
⭐
Alcohol-induced hypoglycemia occurs when ethanol metabolism (ethanol → acetaldehyde → acetate) depletes NAD⁺, shifting the NAD⁺/NADH ratio and blocking gluconeogenesis at multiple steps.
⭐
Starvation hypoglycemia develops after glycogen depletion (~24 hours fasting) when gluconeogenesis cannot meet demands — accelerated in children due to higher brain:body mass ratio.
⭐
Ketotic hypoglycemia of childhood: most common cause of hypoglycemia in children 1–5 years, triggered by prolonged fasting during illness.
⭐
Board pearl: Alcohol specifically blocks gluconeogenesis but not glycogenolysis — hypoglycemia occurs only after glycogen depletion.

Inborn Errors of Metabolism Causing Hypoglycemia
✅
Glycogen storage diseases: defective glycogenolysis → severe fasting hypoglycemia with hepatomegaly (GSD I, III, VI, IX).
✅
Gluconeogenesis defects: fructose-1,6-bisphosphatase deficiency → hypoglycemia with lactic acidosis during fasting.
✅
Fatty acid oxidation defects: impaired ketogenesis → hypoketotic hypoglycemia during fasting (MCAD deficiency most common).
✅
Hereditary fructose intolerance: fructose-1-phosphate accumulation inhibits gluconeogenesis → postprandial hypoglycemia after fructose ingestion.
✅
Board pearl: Hypoglycemia with hepatomegaly in an infant = glycogen storage disease until proven otherwise.

Drug-Induced Hypoglycemia Beyond Insulin
🧠
Sulfonylureas and meglitinides: stimulate endogenous insulin release independent of glucose level → prolonged hypoglycemia risk, especially with renal impairment.
🧠
Pentamidine: direct β-cell toxicity causes acute insulin release → hypoglycemia, followed by diabetes from β-cell destruction.
🧠
Quinolones: rare but serious hypoglycemia through unclear mechanism, particularly in elderly or those with renal dysfunction.
🧠
β-blockers: mask adrenergic symptoms and impair glycogenolysis/gluconeogenesis, especially non-selective agents.
🧠
Salicylates: uncouple oxidative phosphorylation → increased glucose utilization.
🧠
Board pearl: Sulfonylurea-induced hypoglycemia may last >24 hours due to long half-life — requires prolonged monitoring and glucose support.

Tumor-Associated Hypoglycemia
⚡
Insulinoma: pancreatic β-cell tumor secreting insulin autonomously → episodic hypoglycemia, especially fasting, with relief after eating.
⚡
Non-islet cell tumors: large mesenchymal tumors (fibrosarcomas, mesotheliomas) produce IGF-2 → binds insulin receptors → hypoglycemia.
⚡
The IGF-2 mechanism involves production of "big IGF-2" that escapes normal binding protein regulation, increasing bioavailability.
⚡
Diagnostic approach: 72-hour supervised fast with serial glucose, insulin, C-peptide, proinsulin, β-hydroxybutyrate measurements.
⚡
Board clue: Whipple's triad (symptoms during fasting, glucose <55 mg/dL, relief with glucose) plus elevated insulin/C-peptide = insulinoma.

Hepatic Failure and Hypoglycemia
📌
The liver produces 80% of endogenous glucose through glycogenolysis (first 12–24 hours fasting) and gluconeogenesis (beyond 24 hours).
📌
Acute fulminant hepatic failure → rapid glycogen depletion plus failed gluconeogenesis → severe refractory hypoglycemia.
📌
Chronic liver disease rarely causes hypoglycemia until >80% hepatic function is lost — the liver has tremendous reserve capacity.
📌
Contributing factors: decreased glycogen stores, impaired gluconeogenesis, reduced insulin clearance, malnutrition, concurrent alcohol use.
📌
Board pearl: Hypoglycemia in chronic liver disease indicates end-stage disease and portends poor prognosis.

Adrenal Insufficiency and Hypoglycemia
📣
Cortisol deficiency impairs gluconeogenesis by reducing hepatic gluconeogenic enzyme transcription (PEPCK, glucose-6-phosphatase).
📣
Primary adrenal insufficiency: concurrent aldosterone deficiency → hyponatremia, hyperkalemia, hypotension plus hypoglycemia.
📣
Secondary/tertiary insufficiency: isolated cortisol deficiency → hypoglycemia may be the only metabolic abnormality.
📣
Hypoglycemia is more common in children with adrenal insufficiency due to lower glycogen stores and higher metabolic demands.
📣
Board pearl: Morning cortisol <3 μg/dL during hypoglycemia suggests adrenal insufficiency; >20 μg/dL excludes it.

Reactive (Postprandial) Hypoglycemia
🔸
True reactive hypoglycemia is rare — most "hypoglycemic symptoms" after meals occur without documented low glucose.
🔸
Post-gastric surgery hypoglycemia: rapid gastric emptying → exaggerated incretin response → excessive insulin release → hypoglycemia 1–3 hours postprandially.
🔸
Hereditary fructose intolerance: fructose-1-phosphate accumulation after fructose ingestion → acute inhibition of gluconeogenesis/glycogenolysis.
🔸
Insulin autoimmune syndrome: anti-insulin antibodies bind and release insulin unpredictably → postprandial hypoglycemia.
🔸
Board distinction: Fasting hypoglycemia suggests insulin excess or impaired glucose production; postprandial hypoglycemia suggests excessive insulin response to meals.

Neonatal and Infantile Hypoglycemia
🧷
Transitional neonatal hypoglycemia: normal in first 24–48 hours as neonate adapts from constant maternal glucose supply to intermittent feeding.
🧷
Persistent hyperinsulinemic hypoglycemia of infancy (PHHI): genetic defects in K-ATP channel → unregulated insulin secretion → severe hypoglycemia requiring high glucose infusion rates.
🧷
Small for gestational age (SGA) and premature infants: limited glycogen/fat stores → increased hypoglycemia risk.
🧷
Infant of diabetic mother: fetal hyperinsulinemia from maternal hyperglycemia persists briefly after birth → transient hypoglycemia.
🧷
Board pearl: Glucose requirement >10 mg/kg/min to maintain normoglycemia suggests hyperinsulinism.

Exercise-Induced Hypoglycemia
📍
Exercise increases glucose uptake by muscle through insulin-independent GLUT4 translocation via AMPK activation.
📍
During exercise: increased muscle glucose consumption, decreased hepatic glucose production if insulin present, enhanced insulin sensitivity lasting up to 48 hours.
📍
Type 1 diabetics are particularly vulnerable: cannot decrease insulin levels during exercise, impaired counter-regulatory responses.
📍
Late-onset hypoglycemia can occur 6–15 hours post-exercise due to muscle glycogen replenishment and sustained insulin sensitivity.
📍
Board pearl: Exercise-induced hypoglycemia in diabetics requires both insulin dose reduction and carbohydrate supplementation.

Sepsis and Critical Illness Hypoglycemia
🔹
Sepsis creates a hypermetabolic state with increased peripheral glucose consumption by immune cells and infected tissues.
🔹
Cytokines (TNF-α, IL-1, IL-6) impair hepatic gluconeogenesis while increasing peripheral glucose uptake.
🔹
Depletion of glycogen stores, substrate limitation (muscle proteolysis cannot meet amino acid demands), and mitochondrial dysfunction contribute.
🔹
Hypoglycemia in sepsis is an independent predictor of mortality — reflects severity of metabolic derangement.
🔹
Board pearl: Hypoglycemia in sepsis often indicates gram-negative bacteremia and correlates with poor outcomes.

Autoimmune Hypoglycemia Syndromes
⭐
Insulin autoimmune syndrome (Hirata disease): anti-insulin antibodies bind secreted insulin → delayed, unpredictable insulin release → hypoglycemia hours after meals.
⭐
Type B insulin resistance syndrome: antibodies against insulin receptor → initial receptor blockade (hyperglycemia) followed by agonist activity (hypoglycemia).
⭐
Associated with other autoimmune conditions: Graves' disease, SLE, multiple myeloma.
⭐
Triggered by sulfhydryl-containing drugs (methimazole, captopril, penicillamine) in genetically susceptible individuals (HLA-DR4).
⭐
Board clue: Hypoglycemia with markedly elevated total insulin but normal free insulin suggests insulin antibodies.

Factitious and Malicious Hypoglycemia
✅
Factitious hypoglycemia: self-administration of insulin or sulfonylureas — most common in healthcare workers or diabetic patients/relatives.
✅
Münchausen by proxy: caregiver-induced hypoglycemia in children — high mortality if unrecognized.
✅
Diagnostic approach: during hypoglycemia, check insulin, C-peptide, proinsulin, sulfonylurea screen.
✅
Exogenous insulin: high insulin, low C-peptide, low proinsulin.
✅
Sulfonylurea abuse: high insulin, high C-peptide, positive drug screen.
✅
Board pearl: The presence of insulin antibodies in a non-diabetic patient suggests surreptitious insulin use.

Renal Failure and Hypoglycemia
🧠
Kidney contributes 20% of gluconeogenesis — loss in renal failure reduces glucose production capacity.
🧠
Decreased insulin clearance (kidney clears 30–50% of insulin) → prolonged insulin action → increased hypoglycemia risk in diabetics.
🧠
Uremia impairs hepatic gluconeogenesis and causes anorexia → reduced substrate availability.
🧠
Dialysis-related hypoglycemia: glucose-free dialysate, poor nutritional status, removal of gluconeogenic precursors.
🧠
Board pearl: Insulin dose typically needs 25–50% reduction when GFR falls below 30 mL/min.

Hormone Deficiency Syndromes
⚡
Panhypopituitarism: combined deficiency of GH, ACTH (→ cortisol), TSH → impaired counter-regulation and gluconeogenesis.
⚡
Isolated GH deficiency: hypoglycemia mainly in children due to lack of lipolysis → decreased gluconeogenic substrates.
⚡
Glucagon deficiency: rare, seen after total pancreatectomy → loss of primary counter-regulatory hormone.
⚡
Combined deficiencies create more severe hypoglycemia than isolated deficiencies due to loss of backup mechanisms.
⚡
Board pearl: Adults with isolated GH deficiency rarely develop hypoglycemia; children commonly do.

Diagnostic Approach to Hypoglycemia
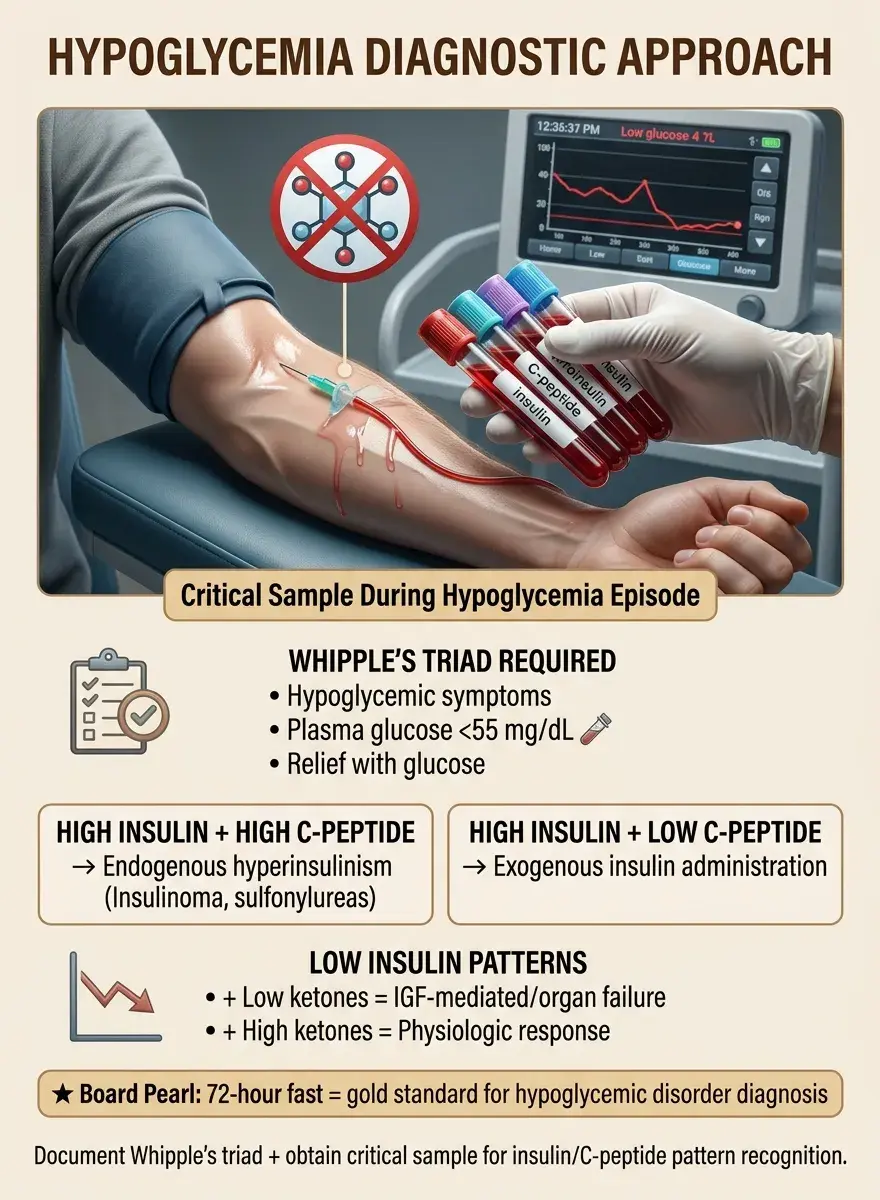
📌
Whipple's triad must be documented: symptoms consistent with hypoglycemia, low plasma glucose (<55 mg/dL), relief with glucose administration.
📌
Critical sample during hypoglycemia: glucose, insulin, C-peptide, proinsulin, β-hydroxybutyrate, cortisol, GH, IGF-2.
📌
High insulin + high C-peptide = endogenous hyperinsulinism (insulinoma, sulfonylurea).
📌
High insulin + low C-peptide = exogenous insulin.
📌
Low insulin + low ketones = IGF-mediated or severe hepatic/renal failure.
📌
Low insulin + high ketones = appropriate physiologic response (alcohol, starvation, hormone deficiency).
📌
Board pearl: The 72-hour fast remains the gold standard for diagnosing hypoglycemic disorders.
Board Question Stem Patterns
📣
Intoxicated patient with hypoglycemia and high anion gap acidosis → alcoholic ketoacidosis.
📣
Hypoglycemia + hepatomegaly in infant → glycogen storage disease.
📣
Hypoglycemia after fructose-containing meal + vomiting → hereditary fructose intolerance.
📣
Large mesenchymal tumor + fasting hypoglycemia → IGF-2 producing tumor.
📣
Hypoglycemia with high insulin and low C-peptide → factitious (exogenous insulin).
📣
Recurrent hypoglycemia + hyperpigmentation + hyperkalemia → primary adrenal insufficiency.
📣
Neonate requiring >10 mg/kg/min glucose infusion → congenital hyperinsulinism.

One-Line Recap
🔸
Hypoglycemia results from either excess insulin action (endogenous or exogenous) or failure of glucose production (hepatic, renal, hormonal, substrate deficiency), with diagnosis requiring Whipple's triad documentation and critical sampling of insulin, C-peptide, and ketones to distinguish insulin-mediated from non-insulin-mediated causes.

bottom of page

