top of page
Hematology & Immunology
Hematopoietic stem cell hierarchy
Core Principle of Hematopoietic Stem Cell Hierarchy
🧷
Hematopoiesis is the highly regulated process of blood cell formation from a single pluripotent hematopoietic stem cell (HSC) through progressive lineage restriction.
🧷
The hierarchy follows a branching pattern: HSC → multipotent progenitors → lineage-committed progenitors → precursors → mature blood cells.
🧷
Each differentiation step involves loss of self-renewal capacity and gain of lineage-specific markers, driven by transcription factors and cytokine signals.
🧷
Board pearl: HSCs are defined by two key properties — self-renewal (ability to divide and maintain the stem cell pool) and multipotency (ability to differentiate into all blood cell lineages).

The Hematopoietic Stem Cell: Foundation of the Hierarchy
📍
HSCs reside primarily in the bone marrow niche, where they remain largely quiescent (G₀ phase) to preserve their self-renewal capacity.
📍
Surface markers: CD34⁺ CD38⁻ (human) or Lin⁻ Sca-1⁺ c-Kit⁺ (mouse). CD34 is the classic HSC marker tested on boards.
📍
HSCs comprise only 0.05% of bone marrow cells but can reconstitute the entire hematopoietic system after transplantation.
📍
Two types: long-term HSCs (LT-HSCs) maintain lifelong hematopoiesis; short-term HSCs (ST-HSCs) provide transient reconstitution.
📍
Board pearl: The gold standard test for HSC function is the ability to reconstitute lethally irradiated recipients — this defines true stem cell activity.

First Branch Point: Myeloid vs Lymphoid Commitment
🔹
HSCs first differentiate into multipotent progenitors (MPPs) that have lost self-renewal but retain multilineage potential.
🔹
MPPs then commit to either common myeloid progenitor (CMP) or common lymphoid progenitor (CLP) — the fundamental branch point of hematopoiesis.
🔹
CMP gives rise to all myeloid cells: erythrocytes, megakaryocytes, granulocytes (neutrophils, eosinophils, basophils), and monocytes.
🔹
CLP gives rise to all lymphoid cells: B cells, T cells, NK cells, and dendritic cells.
🔹
This commitment is driven by master transcription factors: PU.1 and C/EBPα promote myeloid fate; Notch1 and Ikaros promote lymphoid fate.

Common Myeloid Progenitor Differentiation
⭐
CMP further branches into two lineage-restricted progenitors:
⭐
Megakaryocyte-erythroid progenitor (MEP) → produces red blood cells and platelets
⭐
Granulocyte-monocyte progenitor (GMP) → produces neutrophils, eosinophils, basophils, and monocytes
⭐
MEP commitment requires GATA-1 and FOG-1 transcription factors; GMP commitment requires PU.1 and C/EBPα
⭐
Board pearl: Mutations in GATA-1 cause congenital dyserythropoietic anemia and thrombocytopenia, highlighting its role in the MEP lineage.

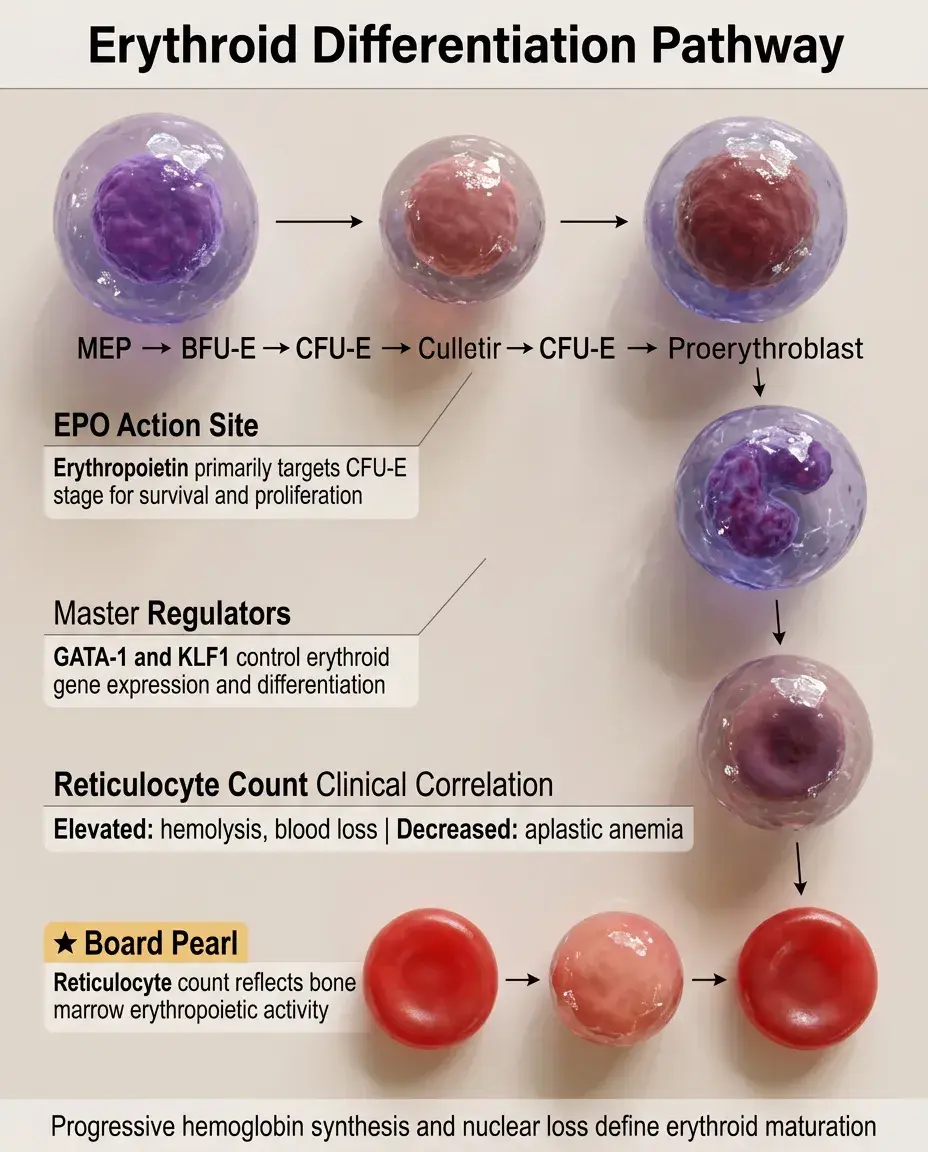
Erythroid Differentiation Pathway
✅
MEP → burst-forming unit-erythroid (BFU-E) → colony-forming unit-erythroid (CFU-E) → proerythroblast → basophilic erythroblast → polychromatophilic erythroblast → orthochromic erythroblast → reticulocyte → mature RBC
✅
Key features: progressive hemoglobin synthesis, nuclear condensation, and eventual enucleation
✅
Erythropoietin (EPO) acts primarily on CFU-E stage, promoting survival and proliferation
✅
GATA-1 and KLF1 are master regulators; mutations cause various congenital anemias
✅
Board pearl: The reticulocyte count reflects erythropoietic activity — elevated in hemolysis or blood loss, decreased in aplastic anemia.
Megakaryocyte Differentiation and Platelet Production
🧠
MEP → megakaryocyte progenitor → promegakaryoblast → megakaryoblast → megakaryocyte → platelets
🧠
Unique feature: endomitosis (DNA replication without cell division) creates polyploid cells up to 128N
🧠
Thrombopoietin (TPO) is the primary regulator, binding c-Mpl receptor throughout differentiation
🧠
Mature megakaryocytes extend proplatelets through marrow sinusoids, releasing 2000-3000 platelets per cell
🧠
Board pearl: Essential thrombocythemia involves JAK2, CALR, or MPL mutations affecting the TPO signaling pathway.

Granulocyte Differentiation: Neutrophil Pathway
⚡
GMP → myeloblast → promyelocyte → myelocyte → metamyelocyte → band → segmented neutrophil
⚡
Promyelocyte stage contains primary (azurophilic) granules with myeloperoxidase
⚡
Myelocyte stage develops secondary (specific) granules containing lactoferrin and neutrophil-specific proteins
⚡
G-CSF drives neutrophil production and release from marrow; stress causes "left shift" with increased bands
⚡
Board pearl: Acute promyelocytic leukemia (APL) arrests at promyelocyte stage due to PML-RARA fusion, causing bleeding from DIC.

Eosinophil and Basophil Differentiation
📌
Both arise from GMP through distinct transcriptional programs
📌
Eosinophils: IL-5 is the primary driver; GATA-1 and C/EBP transcription factors required; contain major basic protein and eosinophil peroxidase
📌
Basophils: IL-3 drives differentiation; contain histamine and heparin; least common granulocyte (<1% of WBCs)
📌
Eosinophilia suggests parasitic infection, allergy, drug reaction, or malignancy (especially Hodgkin lymphoma)
📌
Board pearl: Hypereosinophilic syndrome requires >1500 eosinophils/μL for >6 months with organ damage — can involve FIP1L1-PDGFRA fusion responsive to imatinib.

Monocyte-Macrophage-Dendritic Cell Axis
📣
GMP → monoblast → promonocyte → monocyte → tissue macrophage or dendritic cell
📣
M-CSF (CSF1) drives monocyte production; monocytes circulate 1-3 days before entering tissues
📣
Tissue signals determine final differentiation: microglia (brain), Kupffer cells (liver), alveolar macrophages (lung), osteoclasts (bone)
📣
Dendritic cells can arise from both myeloid (mDC) and lymphoid (pDC) pathways
📣
Board pearl: Langerhans cell histiocytosis shows CD1a⁺ Langerin⁺ cells with "tennis racket" Birbeck granules on EM.

Common Lymphoid Progenitor Differentiation
🔸
CLP gives rise to all adaptive immune cells through distinct niches and signals
🔸
B cell pathway: CLP → pro-B → pre-B → immature B → mature B cell (bone marrow development)
🔸
T cell pathway: CLP → thymic progenitor → double negative → double positive → single positive T cell (thymic development)
🔸
NK cell pathway: CLP → NK progenitor → immature NK → mature NK cell (bone marrow and secondary lymphoid organs)
🔸
IL-7 is essential for both B and T cell development; IL-15 drives NK cell development
🔸
Board pearl: X-linked SCID results from IL-2Rγ mutations, affecting T and NK cells but sparing B cells (T⁻B⁺NK⁻ phenotype).

B Cell Development in Bone Marrow
🧷
Pro-B cell: DJ rearrangement of heavy chain; expresses CD19, CD10, TdT
🧷
Pre-B cell: VDJ rearrangement complete; μ heavy chain pairs with surrogate light chain to form pre-BCR
🧷
Immature B cell: light chain rearrangement; expresses surface IgM; undergoes negative selection
🧷
Key transcription factors: E2A, EBF1, and PAX5 drive B cell commitment
🧷
Board pearl: Acute lymphoblastic leukemia (ALL) markers reflect arrested development — TdT⁺ indicates lymphoblast; CD10⁺ suggests B-ALL.

T Cell Development in Thymus
📍
Double negative (DN) stages: DN1-DN4, characterized by CD44/CD25 expression and TCR β-chain rearrangement
📍
Double positive (DP): CD4⁺CD8⁺, undergoes positive and negative selection based on MHC interaction
📍
Single positive (SP): CD4⁺ or CD8⁺, ready for peripheral circulation
📍
Notch signaling is essential for T cell commitment; GATA-3 drives CD4⁺ fate; Runx3 drives CD8⁺ fate
📍
Board pearl: DiGeorge syndrome (22q11.2 deletion) causes thymic hypoplasia → absent T cells but normal B cells → recurrent viral/fungal infections.

Cytokine Control of Hematopoiesis
🔹
Stem cell factor (SCF/c-Kit ligand): maintains HSCs and early progenitors
🔹
Flt3 ligand: expands early lymphoid and myeloid progenitors
🔹
IL-3: multilineage growth factor supporting various progenitors
🔹
Lineage-specific cytokines: EPO (erythroid), TPO (megakaryocyte), G-CSF (neutrophil), M-CSF (monocyte), IL-5 (eosinophil)
🔹
Board pearl: Polycythemia vera shows EPO-independent erythrocytosis due to JAK2 V617F mutation causing constitutive activation.

Transcription Factor Networks in Hematopoiesis
⭐
Master regulators determine lineage fate through cross-antagonism
⭐
GATA-1 vs PU.1: the paradigm of lineage decision — GATA-1 promotes erythroid/megakaryocyte; PU.1 promotes myeloid/lymphoid
⭐
C/EBPα drives granulocyte differentiation; mutations cause specific AML subtypes
⭐
RUNX1 (AML1) is essential for definitive hematopoiesis; t(8;21) RUNX1-ETO fusion causes AML
⭐
Board pearl: Core binding factor AMLs [t(8;21) and inv(16)] have relatively good prognosis despite being AML.

Hematopoietic Stem Cell Niche
✅
HSCs reside in specialized bone marrow microenvironments that regulate quiescence and self-renewal
✅
Endosteal niche: near bone surface, maintains HSC quiescence through low oxygen and specific signals
✅
Vascular niche: near sinusoids, supports HSC proliferation and differentiation
✅
Key niche cells: osteoblasts, mesenchymal stromal cells, endothelial cells, sympathetic neurons
✅
CXCL12-CXCR4 axis anchors HSCs; AMD3100 blocks this interaction for stem cell mobilization
✅
Board pearl: G-CSF mobilizes HSCs by disrupting CXCL12-CXCR4 binding, allowing harvest for transplantation.

Clinical Markers of Hematopoietic Stages
🧠
CD34: HSCs and early progenitors; used for stem cell enumeration
🧠
CD117 (c-Kit): HSCs and early progenitors; also on mast cells and germ cells
🧠
CD33: myeloid lineage from myeloblast onward; target for AML therapy (gemtuzumab)
🧠
CD15: mature myeloid cells; positive in Hodgkin lymphoma Reed-Sternberg cells
🧠
TdT: lymphoblasts only; distinguishes ALL from mature lymphomas
🧠
Board pearl: Flow cytometry immunophenotyping is essential for leukemia classification — know CD34/CD117 (blasts), CD13/CD33 (myeloid), CD19/CD20 (B cell), CD3/CD5 (T cell).

Disorders of Hematopoietic Hierarchy
⚡
Aplastic anemia: HSC failure → pancytopenia with empty marrow
⚡
Myelodysplastic syndromes: ineffective hematopoiesis with dysplastic changes
⚡
Leukemias: arrested differentiation at specific stages (AML at myeloblast, ALL at lymphoblast)
⚡
Myeloproliferative neoplasms: increased production of one or more mature lineages
⚡
Board pearl: The level of differentiation arrest determines leukemia phenotype — earlier arrest (blast) is acute; later arrest allows maturation (chronic).

Hematopoietic Growth Factor Therapeutics
📌
Erythropoiesis-stimulating agents (ESAs): recombinant EPO for anemia of chronic kidney disease
📌
G-CSF (filgrastim): reduces neutropenia duration after chemotherapy
📌
GM-CSF (sargramostim): broader myeloid stimulation, used less frequently
📌
Thrombopoietin receptor agonists (romiplostim, eltrombopag): for immune thrombocytopenia
📌
IL-11 (oprelvekin): thrombocytopenia treatment (limited use due to side effects)
📌
Board pearl: ESAs increase thrombosis risk and are contraindicated in cancer patients not receiving chemotherapy due to tumor progression concerns.

Board Question Stem Patterns
📣
Pancytopenia with hypocellular marrow and no blasts → aplastic anemia affecting HSCs
📣
Auer rods in myeloblasts → acute myeloid leukemia with arrested granulocyte development
📣
Philadelphia chromosome with maturation to neutrophils → chronic myeloid leukemia
📣
TdT⁺ CD19⁺ CD10⁺ blasts → B-cell acute lymphoblastic leukemia
📣
Teardrop cells and dry tap → myelofibrosis disrupting marrow architecture
📣
Hypersegmented neutrophils → megaloblastic anemia affecting DNA synthesis
📣
CD34⁺ cell collection for transplant → hematopoietic stem cell harvest

One-Line Recap
🔸
Hematopoiesis follows a strict hierarchy from self-renewing CD34⁺ HSCs through multipotent progenitors that branch at the CMP/CLP decision point into all blood lineages, regulated by cytokine-transcription factor networks (EPO-GATA1 for erythroid, G-CSF-C/EBPα for granulocyte, IL-7-PAX5 for B cells), with differentiation arrests causing leukemias and regulatory failures causing cytopenias or myeloproliferative disorders.

bottom of page

