top of page
Reproductive & Endocrine Systems
Glucagon, somatostatin, and pancreatic polypeptide
Core Principle of Pancreatic Islet Counter-Regulatory Hormones
🧷
The endocrine pancreas contains four major cell types: β cells (insulin), α cells (glucagon), δ cells (somatostatin), and PP cells (pancreatic polypeptide).
🧷
While insulin promotes anabolism and glucose storage, glucagon opposes insulin by promoting catabolism and glucose release — maintaining glucose homeostasis through reciprocal regulation.
🧷
Somatostatin acts as the universal brake, inhibiting secretion of both insulin and glucagon, while pancreatic polypeptide modulates digestive processes.
🧷
Understanding these hormones requires thinking in terms of metabolic states: fed versus fasted, and how the body coordinates fuel mobilization.

Glucagon Structure and Synthesis
📍
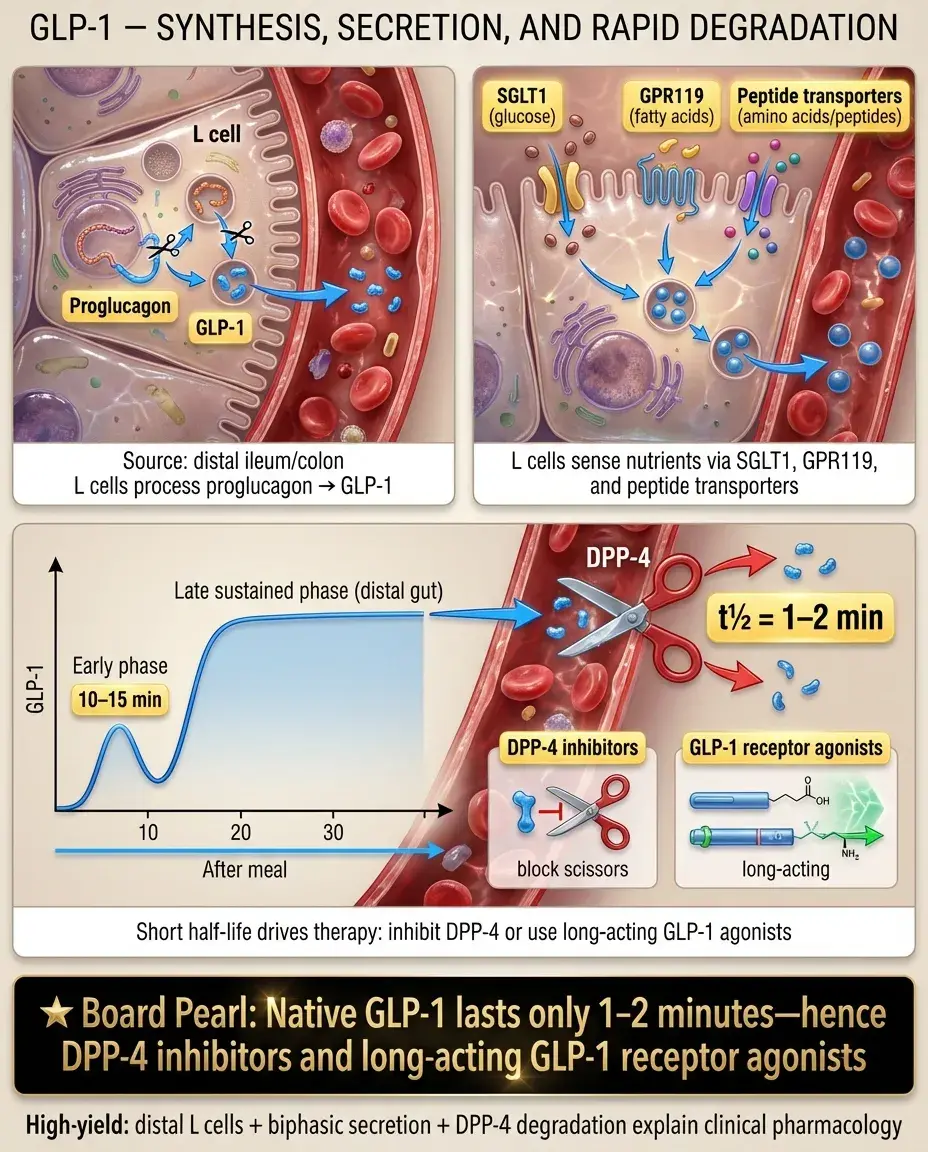
Glucagon is a 29-amino acid peptide hormone synthesized in pancreatic α cells from proglucagon.
📍
The proglucagon gene yields tissue-specific products: glucagon in α cells, but GLP-1 and GLP-2 in intestinal L cells — explaining why GLP-1 agonists don't cause hypoglycemia.
📍
Glucagon has a very short half-life (~5 minutes), allowing rapid on-off switching of its catabolic effects.
📍
Board pearl: Glucagon and GLP-1 come from the same precursor but have opposite effects on glucose — glucagon raises it, GLP-1 lowers it by enhancing insulin secretion.
Glucagon Secretion Triggers and Inhibitors
🔹
Primary stimuli: hypoglycemia (most potent), amino acids (especially arginine and alanine), sympathetic activation (β-adrenergic), cortisol.
🔹
Primary inhibitors: hyperglycemia, insulin, somatostatin, GLP-1, fatty acids.
🔹
The postprandial amino acid rise explains why protein meals stimulate both insulin AND glucagon — preventing hypoglycemia from protein-induced insulin secretion.
🔹
During prolonged fasting, falling insulin removes tonic suppression of α cells, allowing glucagon to rise and maintain euglycemia.
🔹
Board clue: Mixed meal → both insulin and glucagon rise; pure glucose load → insulin rises, glucagon falls.

Glucagon Mechanism of Action
⭐
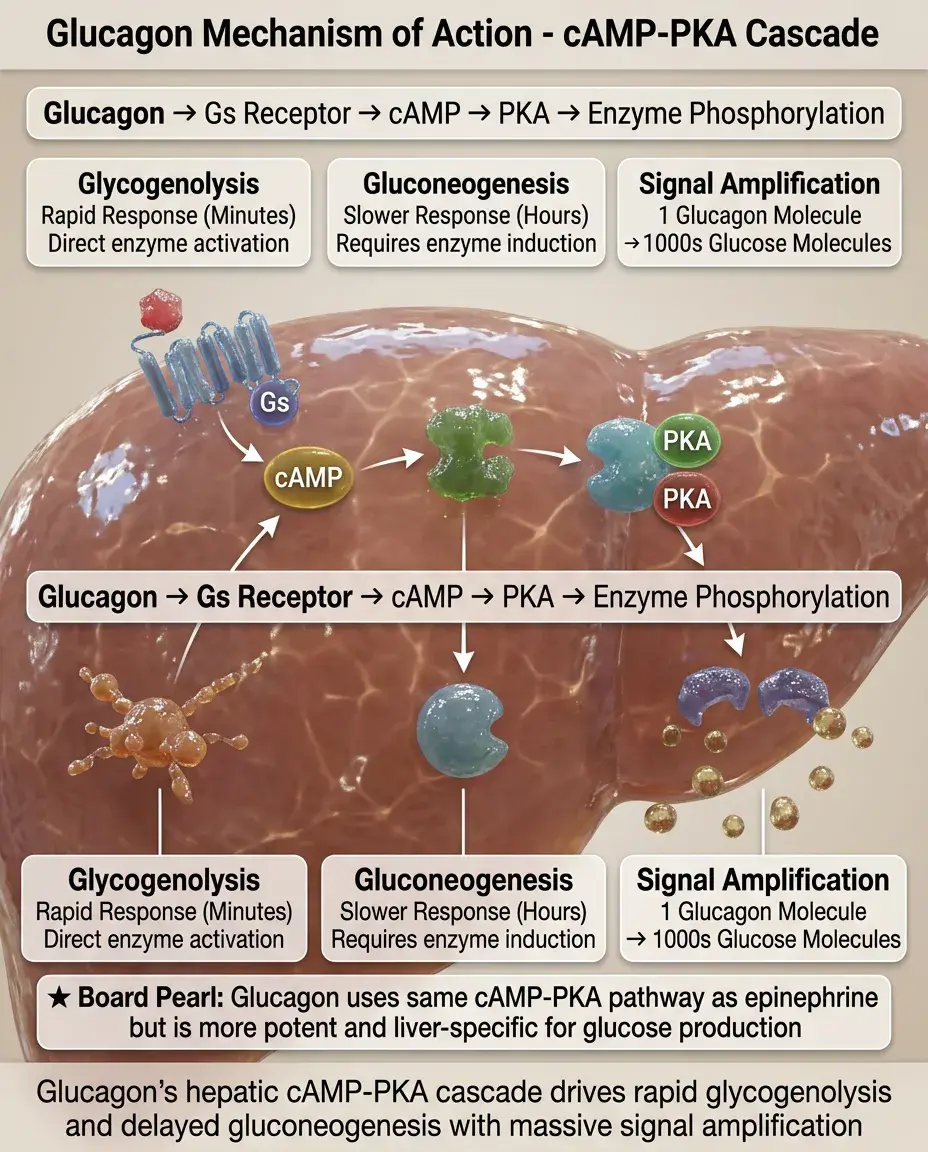
Glucagon binds Gs-coupled receptors primarily in liver (also kidney, heart, adipose tissue) → ↑cAMP → PKA activation.
⭐
In liver: PKA phosphorylates key enzymes, activating catabolic pathways and inhibiting anabolic ones.
⭐
The cAMP-PKA cascade amplifies the signal — one glucagon molecule can generate thousands of glucose molecules.
⭐
Glucagon effects are rapid (minutes) for glycogenolysis but slower (hours) for gluconeogenesis due to required enzyme induction.
⭐
Board pearl: Glucagon works through the same cAMP-PKA pathway as epinephrine in liver, but glucagon is more potent and specific for hepatic glucose production.
Glucagon's Hepatic Effects: The Catabolic Cascade
✅
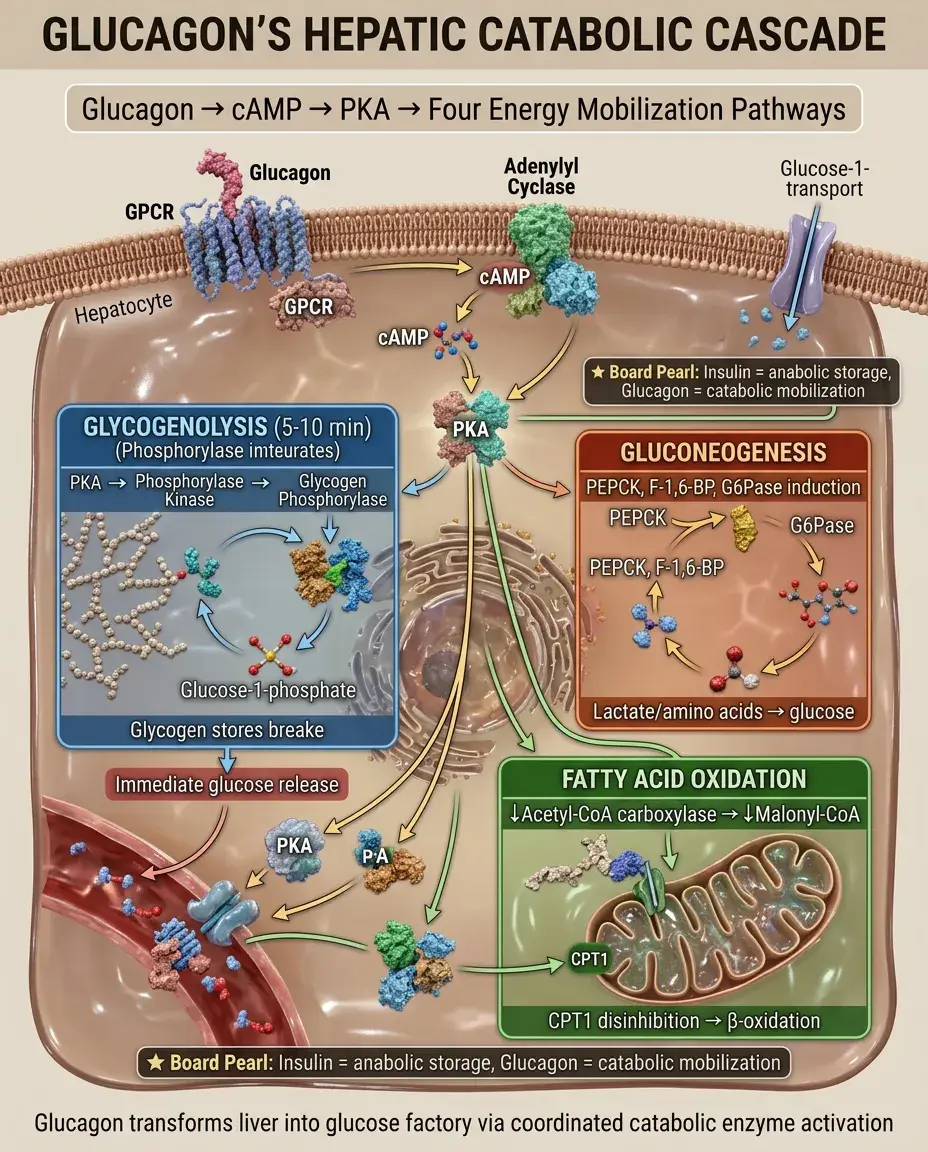
Glycogenolysis: PKA phosphorylates phosphorylase kinase → activates glycogen phosphorylase → glucose release within 5-10 minutes.
✅
Gluconeogenesis: induces PEPCK, fructose-1,6-bisphosphatase, glucose-6-phosphatase → glucose synthesis from lactate, amino acids, glycerol.
✅
Fatty acid oxidation: ↓acetyl-CoA carboxylase → ↓malonyl-CoA → disinhibition of CPT1 → fatty acids enter mitochondria for β-oxidation.
✅
Ketogenesis: acetyl-CoA from β-oxidation → HMG-CoA → ketone bodies (acetoacetate, β-hydroxybutyrate).
✅
Board distinction: Insulin promotes energy storage; glucagon promotes energy mobilization and oxidation.
Glucagon in Lipid and Protein Metabolism
🧠
Adipose tissue: promotes lipolysis → free fatty acids and glycerol released → substrate for hepatic gluconeogenesis and ketogenesis.
🧠
Protein metabolism: increases hepatic amino acid uptake and transamination → carbon skeletons for gluconeogenesis, nitrogen for urea cycle.
🧠
The combination of proteolysis and ureagenesis explains the negative nitrogen balance seen in uncontrolled diabetes or prolonged fasting.
🧠
Glucagon does NOT directly affect muscle protein breakdown — that's mediated by cortisol and lack of insulin.
🧠
Board pearl: Glucagon mobilizes all three macronutrients, but its primary board-tested effects are on glucose and ketone production.

Somatostatin: The Universal Inhibitor
⚡
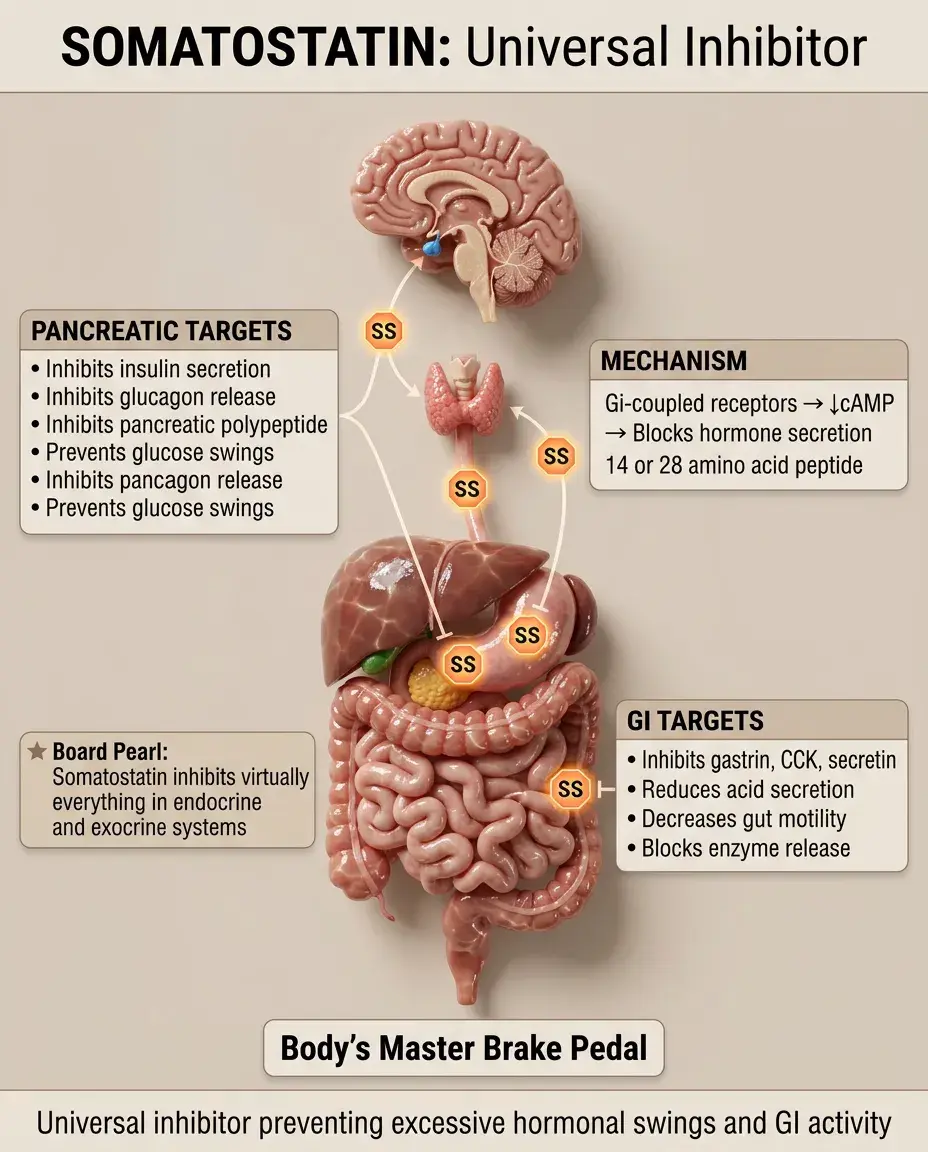
Somatostatin is a 14 or 28 amino acid peptide (two active forms) produced by pancreatic δ cells, hypothalamus, GI tract D cells.
⚡
Acts through Gi-coupled receptors → ↓cAMP → inhibits hormone secretion and reduces GI motility and secretion.
⚡
Pancreatic effects: inhibits insulin, glucagon, and pancreatic polypeptide — preventing excessive swings in glucose.
⚡
GI effects: inhibits gastrin, CCK, secretin, motilin, VIP, GIP → reduced acid secretion, enzyme secretion, and gut motility.
⚡
Board pearl: Somatostatin inhibits virtually everything — think of it as the body's brake pedal for both endocrine and exocrine secretion.
Somatostatin Regulation and Clinical Correlates
📌
Stimulated by: hyperglycemia, amino acids, fatty acids, CCK, glucagon — essentially all nutrients and gut hormones.
📌
Inhibited by: α-adrenergic stimulation, low glucose.
📌
Paracrine action is key — δ cells are strategically positioned to sense and modulate neighboring α and β cell secretion.
📌
Octreotide (somatostatin analog) clinical uses: acromegaly, carcinoid syndrome, VIPomas, variceal bleeding, persistent hypoglycemia from insulinoma.
📌
Board clue: Patient with secretory diarrhea from VIPoma or carcinoid → octreotide reduces hormone secretion and improves symptoms.

Pancreatic Polypeptide: The Satiety Signal
📣
PP is a 36-amino acid peptide from pancreatic PP cells (also called F cells), primarily in the pancreatic head.
📣
Secretion is under vagal control — rises with meals (especially protein), peaks at 10-20 minutes, remains elevated for 4-6 hours.
📣
Acts centrally to reduce appetite and food intake; peripherally to slow gastric emptying and reduce pancreatic enzyme secretion.
📣
PP levels are used clinically to assess vagal integrity and distinguish type 3c (pancreatogenic) from type 1/2 diabetes.
📣
Board pearl: PP is the only pancreatic hormone that requires intact vagal innervation for normal meal-stimulated secretion.

Integration of Islet Cell Paracrine Signaling
🔸
Islet architecture enables paracrine regulation: β cells in core, α and δ cells in mantle → insulin inhibits glucagon; somatostatin inhibits both.
🔸
Blood flows from β-cell core to α/δ-cell periphery, allowing insulin to suppress glucagon secretion directly.
🔸
Gap junctions between β cells synchronize insulin pulses; similar connections coordinate α cell responses.
🔸
Loss of paracrine regulation in diabetes: β cell destruction → loss of tonic glucagon suppression → inappropriate hyperglucagonemia → worsened hyperglycemia.
🔸
Board concept: Type 1 diabetes involves both insulin deficiency AND glucagon excess due to lost paracrine inhibition.

Glucagon in Hypoglycemia Defense
🧷
First defense (glucose ~80 mg/dL): ↓insulin secretion.
🧷
Second defense (glucose ~65-70 mg/dL): ↑glucagon and ↑epinephrine.
🧷
Third defense (glucose ~55-60 mg/dL): ↑cortisol and growth hormone.
🧷
Glucagon provides rapid glucose recovery via hepatic glycogenolysis; epinephrine provides backup when glucagon is deficient.
🧷
Board pearl: In type 1 diabetes with frequent hypoglycemia → impaired glucagon response (α cell dysfunction) → increased reliance on epinephrine → if autonomic neuropathy present → hypoglycemia unawareness and severe risk.

Clinical Glucagonoma Syndrome
📍
Rare α cell tumor secreting excessive glucagon → characteristic syndrome.
📍
Necrolytic migratory erythema: pathognomonic rash with erythematous patches, central clearing, bronze induration — affects intertriginous areas.
📍
Glucose intolerance/diabetes: from unopposed glucagon action.
📍
Weight loss, anemia, glossitis, cheilitis: from increased protein catabolism and amino acid depletion.
📍
Venous thrombosis risk increased.
📍
Board clue: Diabetic patient + characteristic migratory rash + weight loss → measure glucagon levels → if >1000 pg/mL → glucagonoma.

Somatostatinoma: The Inhibitory Syndrome
🔹
Rare δ cell tumor causing excessive somatostatin → inhibition of multiple hormones.
🔹
Triad: diabetes (inhibited insulin), cholelithiasis (inhibited CCK → gallbladder stasis), steatorrhea (inhibited pancreatic enzymes).
🔹
Often associated with neurofibromatosis type 1.
🔹
May present with dyspepsia, hypochlorhydria (inhibited gastrin), weight loss.
🔹
Diagnosis: elevated somatostatin levels + imaging showing pancreatic or duodenal mass.
🔹
Board distinction: Glucagonoma → too much stimulation; somatostatinoma → too much inhibition.

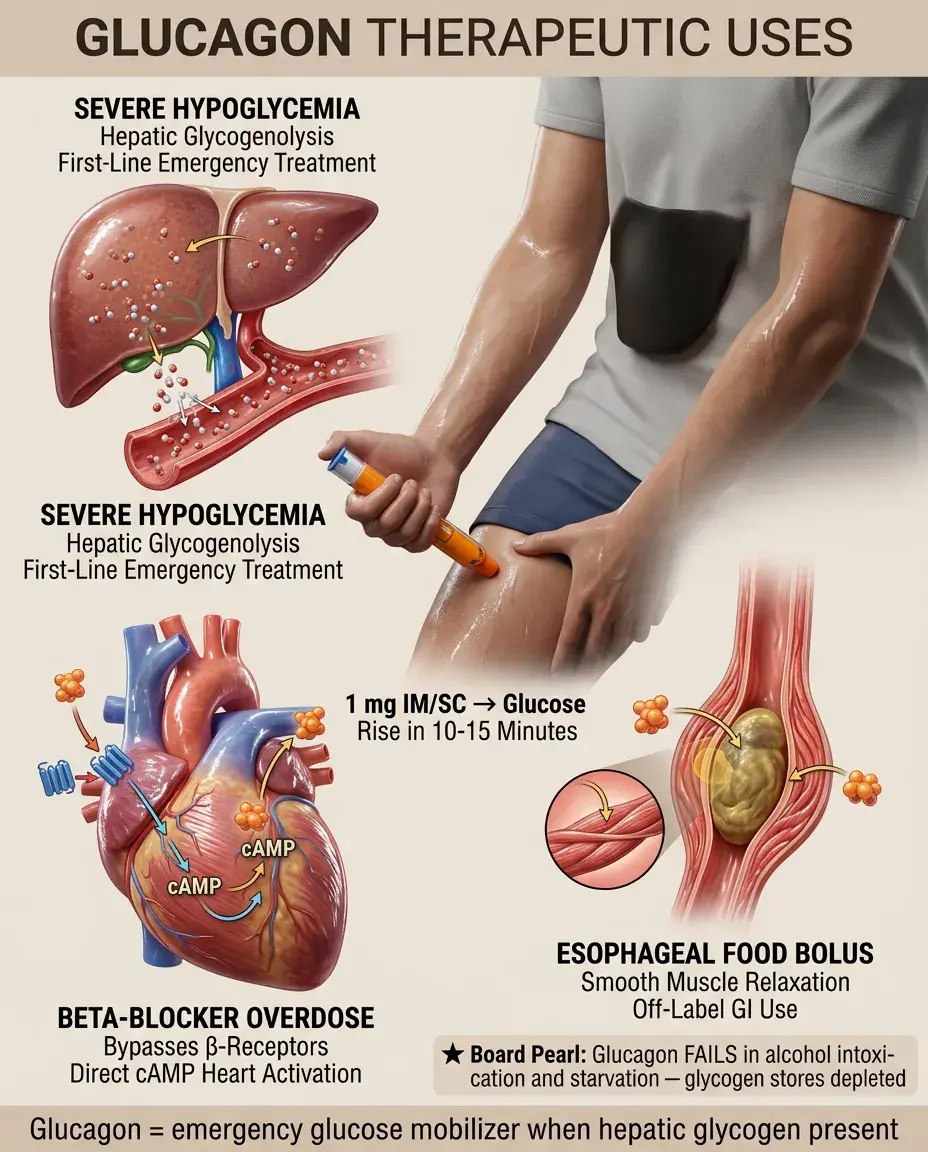
Therapeutic Uses of Glucagon
⭐
Severe hypoglycemia: 1 mg IM/SC → raises glucose within 10-15 minutes by hepatic glycogenolysis (ineffective if glycogen depleted).
⭐
Beta-blocker overdose: bypasses β-receptor blockade via direct cAMP activation in heart → positive inotropic/chronotropic effects.
⭐
Esophageal food bolus: smooth muscle relaxation (off-label use).
⭐
Diagnostic imaging: GI hypomotility for better visualization.
⭐
Board pearl: Glucagon won't work for hypoglycemia in alcohol intoxication or prolonged starvation — these states deplete hepatic glycogen stores.
Fasting Physiology and Hormonal Coordination
✅
Early fasting (0-4 hours): insulin falls, glucagon rises modestly → hepatic glucose output matches peripheral utilization.
✅
Prolonged fasting (>24 hours): glucagon remains elevated → glycogen exhausted → gluconeogenesis becomes primary glucose source.
✅
Starvation (>72 hours): glucagon drives ketogenesis → brain shifts to ketone utilization → glucose sparing for obligate users (RBCs, renal medulla).
✅
Counter-regulatory failure in diabetes: cannot suppress glucagon → excessive hepatic glucose output even when glucose is already elevated.
✅
Board concept: Fasting = ↓insulin/glucagon ratio; feeding = ↑insulin/glucagon ratio.

Islet Cell Tumors and MEN Syndromes
🧠
Insulinoma: most common islet tumor → hypoglycemia with ↑insulin, ↑C-peptide, ↑proinsulin.
🧠
Gastrinoma: second most common → Zollinger-Ellison syndrome (peptic ulcers, diarrhea, GERD).
🧠
VIPoma: watery diarrhea, hypokalemia, achlorhydria (WDHA syndrome).
🧠
Glucagonoma and somatostatinoma: rare but characteristic syndromes.
🧠
Board association: Multiple islet tumors + hyperparathyroidism + pituitary adenoma → MEN1; islet tumor + pheochromocytoma + medullary thyroid cancer → consider MEN2.

Laboratory Testing and Hormone Measurement
⚡
Glucagon normal range: 50-150 pg/mL; >1000 pg/mL suggests glucagonoma.
⚡
Somatostatin: not routinely measured due to short half-life; somatostatin-like immunoreactivity used instead.
⚡
Pancreatic polypeptide: elevated in islet tumors (non-specific marker), low in chronic pancreatitis and vagal dysfunction.
⚡
Dynamic testing: glucagon stimulation test evaluates pituitary and adrenal reserve; arginine stimulation assesses α cell function.
⚡
Board tip: Random hormone levels less useful than stimulation/suppression tests for functional assessment.

Integration with Incretin System
📌
GLP-1 and GIP are incretins — gut hormones that augment insulin secretion in response to oral nutrients.
📌
GLP-1 also suppresses glucagon, but only when glucose is elevated → why GLP-1 agonists rarely cause hypoglycemia.
📌
Somatostatin inhibits both GLP-1 and GIP secretion from intestinal cells.
📌
In type 2 diabetes: impaired incretin effect contributes to postprandial hyperglycemia and inappropriate glucagon secretion.
📌
Board connection: DPP-4 inhibitors prevent incretin breakdown → enhanced insulin, suppressed glucagon → improved glucose without hypoglycemia risk.

Board Question Stem Patterns
📣
Diabetic + migratory rash + weight loss → glucagonoma until proven otherwise.
📣
Diabetes + gallstones + steatorrhea → somatostatinoma.
📣
Hypoglycemic patient given glucagon with no response → consider alcohol intoxication or glycogen storage disease.
📣
Type 1 diabetic with recurrent severe hypoglycemia → impaired glucagon and epinephrine responses.
📣
Octreotide for GI bleeding → reduces splanchnic blood flow via inhibition of vasodilatory hormones.
📣
Post-gastrectomy patient with better glucose control → loss of incretin-stimulated insulin and glucagon.
📣
MEN1 patient with peptic ulcers → gastrinoma most likely, but screen for other islet tumors.

One-Line Recap
🔸
Glucagon (α cells) opposes insulin by promoting hepatic glucose output and ketogenesis via cAMP-PKA signaling, while somatostatin (δ cells) inhibits both insulin and glucagon to prevent metabolic extremes, and pancreatic polypeptide (PP cells) modulates satiety and digestive functions — together maintaining glucose homeostasis through intricate paracrine networks disrupted in diabetes and rare islet cell tumors.

bottom of page

