top of page
Cardiovascular System
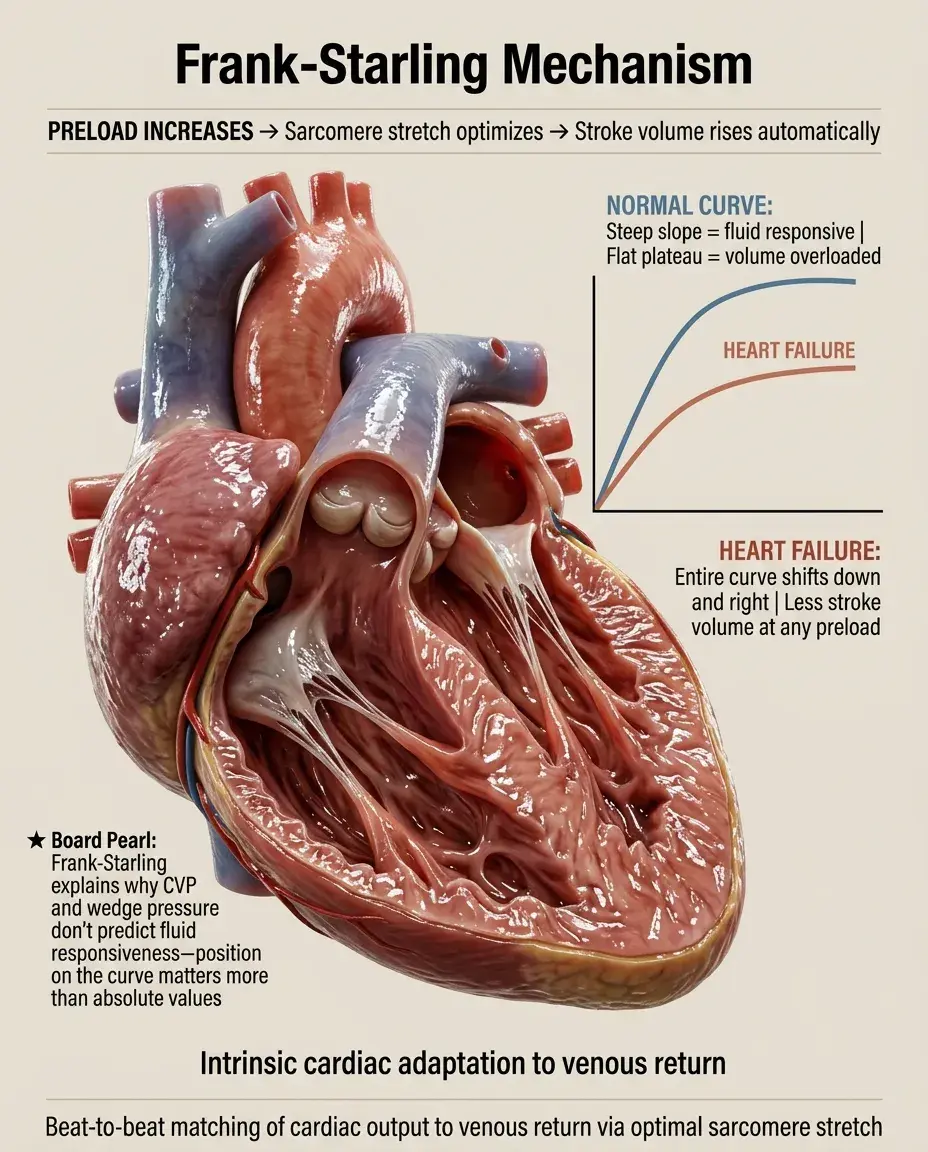
Frank–Starling mechanism
Core Principle of the Frank-Starling Mechanism
🧷
The Frank-Starling mechanism states that the heart automatically adjusts its contractile force based on the volume of blood returning to it — within physiological limits, increased venous return leads to increased stroke volume.
🧷
This intrinsic property of cardiac muscle occurs at the sarcomere level: optimal overlap between actin and myosin filaments produces maximal force generation when the muscle is stretched to its ideal length.
🧷
The mechanism ensures that cardiac output from the right and left ventricles remains balanced over time, preventing blood accumulation in either the pulmonary or systemic circulation.
🧷
This autoregulatory mechanism operates independently of neural or hormonal control, making it the heart's first-line response to changes in preload.

The Molecular Basis: Length-Tension Relationship
📍
At the sarcomere level, increased ventricular filling stretches cardiac myocytes, optimizing actin-myosin overlap for cross-bridge formation.
📍
Unlike skeletal muscle, cardiac muscle operates on the ascending limb of the length-tension curve under normal conditions — meaning physiological stretching always increases contractile force.
📍
Stretched cardiac muscle also increases calcium sensitivity of troponin C, enhancing the contractile response without requiring more calcium release.
📍
Beyond optimal stretch (seen in severe heart failure), the descending limb is reached where further stretching reduces contractile force — this is rarely achieved in vivo due to pericardial constraint.
📍
Board pearl: The Frank-Starling mechanism is why increased preload leads to increased stroke volume in a normal heart.

Ventricular Function Curves
🔹
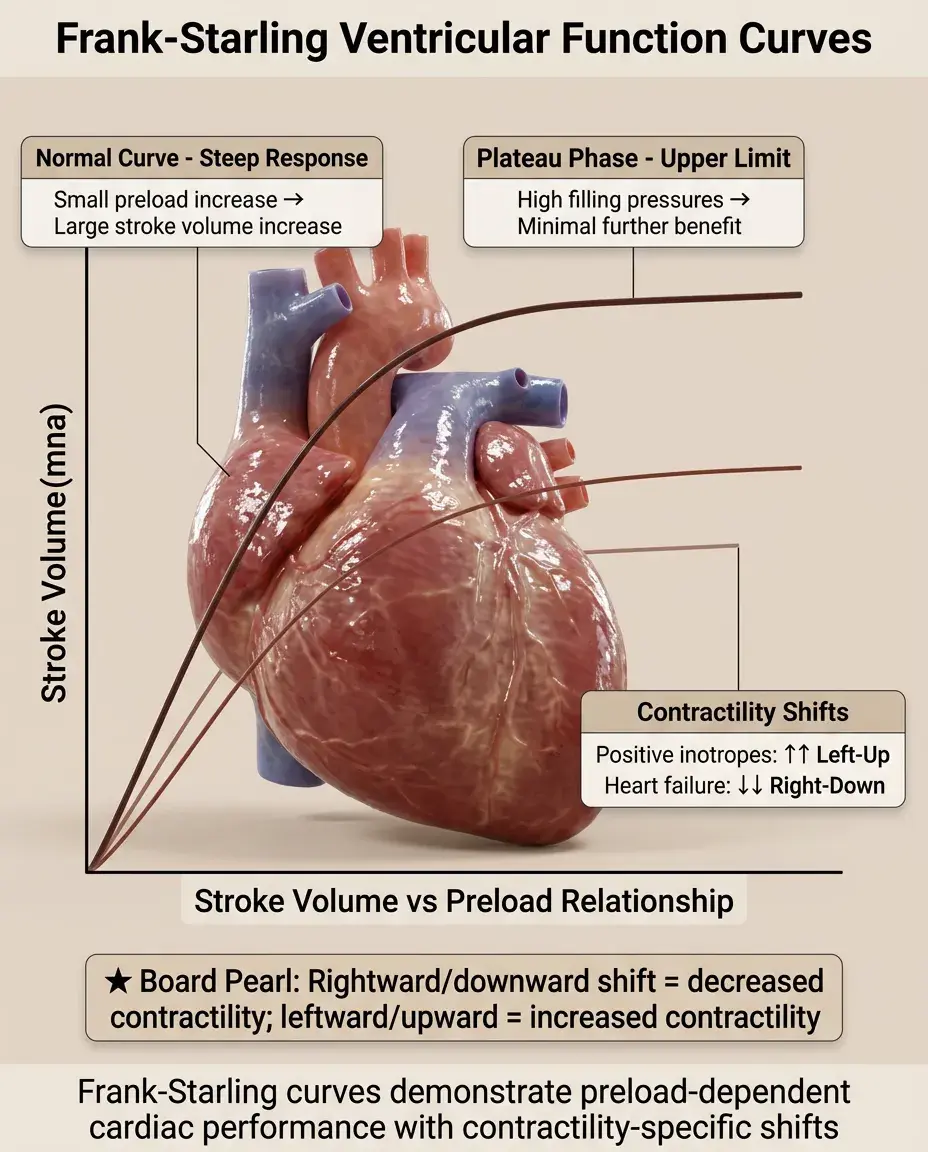
The Frank-Starling curve plots stroke volume (or cardiac output) against end-diastolic volume (or pressure), showing the positive relationship between preload and cardiac performance.
🔹
In a normal heart, the curve is steep — small increases in preload produce substantial increases in stroke volume.
🔹
The curve plateaus at high filling pressures, representing the upper limit of the Frank-Starling mechanism where further stretch provides minimal benefit.
🔹
Different curves exist for different contractile states: positive inotropes shift the curve upward and leftward, while heart failure shifts it downward and rightward.
🔹
Board pearl: On exam diagrams, a rightward/downward shift indicates decreased contractility; leftward/upward indicates increased contractility.
Clinical Determinants of Preload
⭐
Preload is determined by venous return, which depends on: total blood volume, venous tone, body position, intrathoracic pressure, atrial contraction, and ventricular compliance.
⭐
Increased preload occurs with: IV fluid administration, blood transfusion, leg elevation, decreased venous compliance (sympathetic activation), and negative-pressure breathing.
⭐
Decreased preload occurs with: hemorrhage, dehydration, venodilation (nitrates), standing upright, positive-pressure ventilation, and loss of atrial kick (atrial fibrillation).
⭐
End-diastolic volume is the best measure of preload, though clinically we often use surrogates like central venous pressure or pulmonary capillary wedge pressure.
⭐
Board distinction: Preload affects stroke volume via the Frank-Starling mechanism; afterload affects stroke volume via the force-velocity relationship.

The Relationship Between EDV, EDP, and Compliance
✅
End-diastolic volume (EDV) directly determines sarcomere length and thus contractile force via Frank-Starling.
✅
End-diastolic pressure (EDP) is what we measure clinically but is only a surrogate for EDV — the relationship between them depends on ventricular compliance.
✅
In a compliant ventricle, large volume changes produce small pressure changes; in a stiff ventricle, small volume changes produce large pressure changes.
✅
Decreased compliance (diastolic dysfunction, ventricular hypertrophy, restrictive cardiomyopathy) means higher filling pressures are needed to achieve the same EDV and stroke volume.
✅
Board pearl: A stiff ventricle operates on a steeper pressure-volume curve, requiring higher filling pressures to achieve normal stroke volumes.

Frank-Starling in Exercise Physiology
🧠
During exercise, venous return increases due to muscle pump action, sympathetic venoconstriction, and increased respiratory pump — all increasing preload.
🧠
The Frank-Starling mechanism provides the immediate response to increased venous return, increasing stroke volume within beats.
🧠
Sympathetic activation then provides additional inotropy, shifting the entire Frank-Starling curve upward to further augment stroke volume.
🧠
Trained athletes have larger end-diastolic volumes and operate on a more favorable portion of the Frank-Starling curve, contributing to their increased stroke volume.
🧠
The combination of Frank-Starling (preload-mediated) and sympathetic (inotropy-mediated) mechanisms can increase cardiac output 4-6 fold during maximal exercise.

Heart Failure and the Frank-Starling Curve
⚡
In systolic heart failure, the Frank-Starling curve is depressed and flattened — the same preload produces less stroke volume, and the curve plateaus at lower outputs.
⚡
Compensatory mechanisms (neurohormonal activation, fluid retention) increase preload to maintain cardiac output by moving rightward along the depressed curve.
⚡
This compensation comes at the cost of elevated filling pressures, leading to pulmonary and systemic congestion — the clinical syndrome of congestive heart failure.
⚡
Decompensation occurs when the curve is so flat that further increases in preload provide minimal benefit while dramatically worsening congestion.
⚡
Board pearl: Heart failure patients operate on the flat portion of a depressed Frank-Starling curve — high filling pressures with low cardiac output.

Therapeutic Manipulation of the Frank-Starling Relationship
📌
Diuretics reduce preload, moving leftward on the curve — in heart failure, this reduces congestion with minimal impact on cardiac output if operating on the flat portion.
📌
Inotropes (digoxin, dobutamine, milrinone) shift the entire curve upward, improving cardiac output at any given preload.
📌
Vasodilators reduce afterload, indirectly improving the Frank-Starling relationship by allowing more complete emptying and thus lower end-systolic volumes.
📌
ACE inhibitors and ARBs provide combined preload and afterload reduction while preventing maladaptive remodeling that would further depress the curve.
📌
Board pearl: The goal in heart failure is to optimize position on the Frank-Starling curve — reducing congestion without compromising cardiac output.

Right Ventricular Frank-Starling Dynamics
📣
The right ventricle follows the same Frank-Starling principles but operates at lower pressures and is more sensitive to afterload changes.
📣
RV preload is determined by systemic venous return and is measured as central venous pressure (CVP) or right atrial pressure.
📣
The RV Frank-Starling curve is shifted downward in RV failure, pulmonary hypertension, or RV infarction.
📣
Because the ventricles are in series, RV and LV outputs must match over time — the Frank-Starling mechanism ensures this balance beat-to-beat.
📣
Board pearl: Acute pulmonary embolism causes RV dilation as the ventricle uses Frank-Starling to maintain output against increased afterload.

Pericardial Constraint and Ventricular Interdependence
🔸
The pericardium limits total cardiac volume, creating ventricular interdependence — expansion of one ventricle restricts filling of the other.
🔸
In cardiac tamponade, pericardial fluid prevents normal ventricular filling, shifting both ventricles to the steep portion of their pressure-volume curves.
🔸
Constrictive pericarditis creates a rigid shell that abruptly limits filling at higher volumes, causing a "square root sign" on pressure tracings.
🔸
Ventricular interdependence is exaggerated in these conditions, leading to pulsus paradoxus — inspiratory decrease in LV stroke volume as RV filling increases.
🔸
Board pearl: Pericardial disease limits the Frank-Starling mechanism by preventing normal ventricular filling and stretch.

Atrial Contribution to the Frank-Starling Mechanism
🧷
Atrial contraction contributes 15-25% of ventricular filling in normal hearts, providing a "atrial kick" that optimizes end-diastolic volume.
🧷
Loss of atrial contraction (atrial fibrillation) reduces preload and shifts the operating point leftward on the Frank-Starling curve.
🧷
In stiff ventricles (LVH, elderly), atrial contribution increases to 30-40% of filling — these patients are particularly sensitive to loss of atrial kick.
🧷
AV synchrony ensures atrial contraction occurs during ventricular diastole; AV dissociation (complete heart block) eliminates this contribution.
🧷
Board pearl: New-onset atrial fibrillation can precipitate heart failure in patients with diastolic dysfunction by eliminating atrial contribution to preload.

Myocardial Stretch and Immediate vs Slow Force Response
📍
The immediate force response (Frank-Starling) occurs within the same beat — stretched muscle immediately generates more force.
📍
The slow force response (Anrep effect) develops over minutes — sustained stretch gradually increases calcium transients and contractility.
📍
Acute volume loading demonstrates pure Frank-Starling; chronic volume overload invokes both mechanisms plus eventual hypertrophy.
📍
Chronic stretch activates growth pathways leading to eccentric hypertrophy — adding sarcomeres in series to normalize wall stress.
📍
Board distinction: Frank-Starling is immediate and intrinsic; the Anrep effect is gradual and involves altered calcium handling.

Integration with Autonomic Regulation
🔹
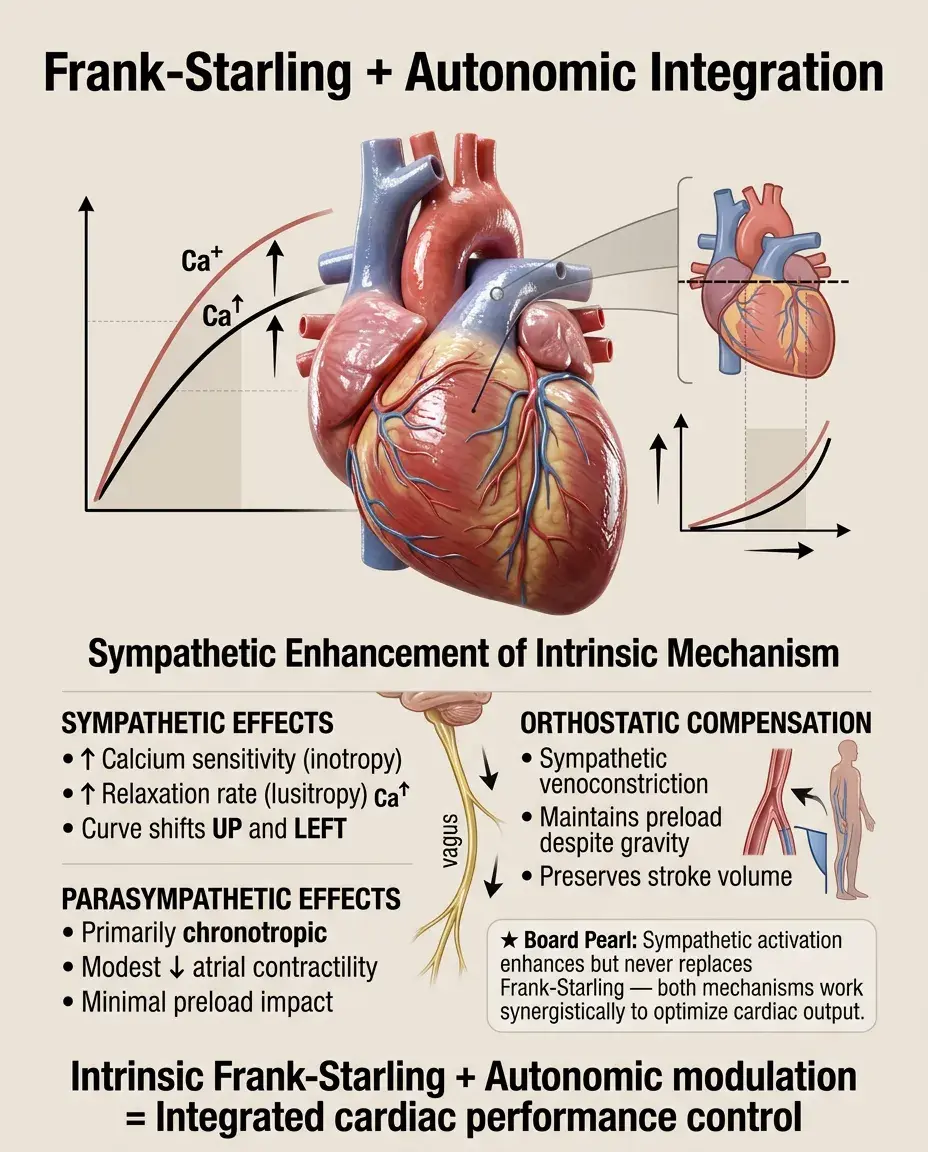
While Frank-Starling is intrinsic, sympathetic stimulation enhances it by increasing calcium sensitivity and lusitropy (relaxation).
🔹
β-adrenergic stimulation shifts the entire Frank-Starling curve upward (inotropy) and leftward (lusitropy improves filling).
🔹
Parasympathetic effects are primarily chronotropic but vagal tone can modestly decrease atrial contractility, reducing preload contribution.
🔹
During orthostatic stress, sympathetic venoconstriction maintains preload to preserve stroke volume via Frank-Starling despite gravitational pooling.
🔹
Board pearl: Sympathetic activation enhances but does not replace the Frank-Starling mechanism — both work synergistically to increase cardiac output.
Measurement and Clinical Assessment
⭐
Swan-Ganz catheterization directly measures the Frank-Starling relationship by plotting cardiac output against pulmonary capillary wedge pressure.
⭐
Echocardiography estimates preload using end-diastolic volume and assesses the Frank-Starling relationship through stroke volume variation.
⭐
Passive leg raise transiently increases preload — a 10% increase in cardiac output suggests the patient is on the steep portion of the curve (preload-responsive).
⭐
Central venous pressure variation with respiration indicates volume status and position on the Frank-Starling curve.
⭐
Board pearl: Preload responsiveness means the patient is on the steep part of the Frank-Starling curve and will benefit from volume administration.

Pathological Limits and Decompensation
✅
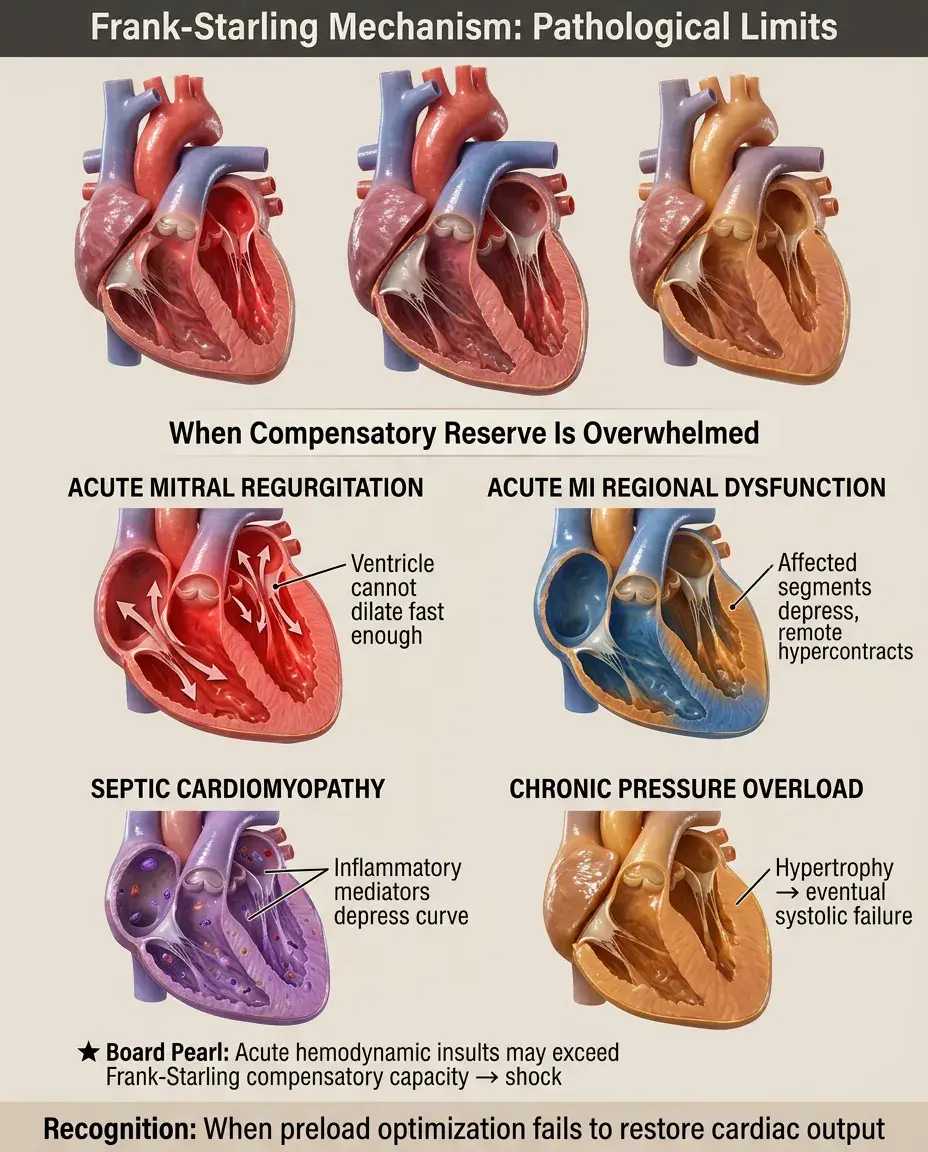
Acute severe mitral regurgitation overwhelms the Frank-Starling mechanism — the ventricle cannot dilate fast enough to maintain forward stroke volume.
✅
Acute myocardial infarction depresses regional Frank-Starling function in the affected segments while remote myocardium hypercontracts.
✅
Septic cardiomyopathy depresses the Frank-Starling curve through inflammatory mediators despite often normal or increased preload.
✅
Chronic pressure overload (aortic stenosis, hypertension) eventually transitions from concentric hypertrophy to systolic failure with depressed Frank-Starling function.
✅
Board pearl: Acute hemodynamic insults may exceed the compensatory capacity of the Frank-Starling mechanism, leading to shock.
Species Differences and Evolutionary Perspective
🧠
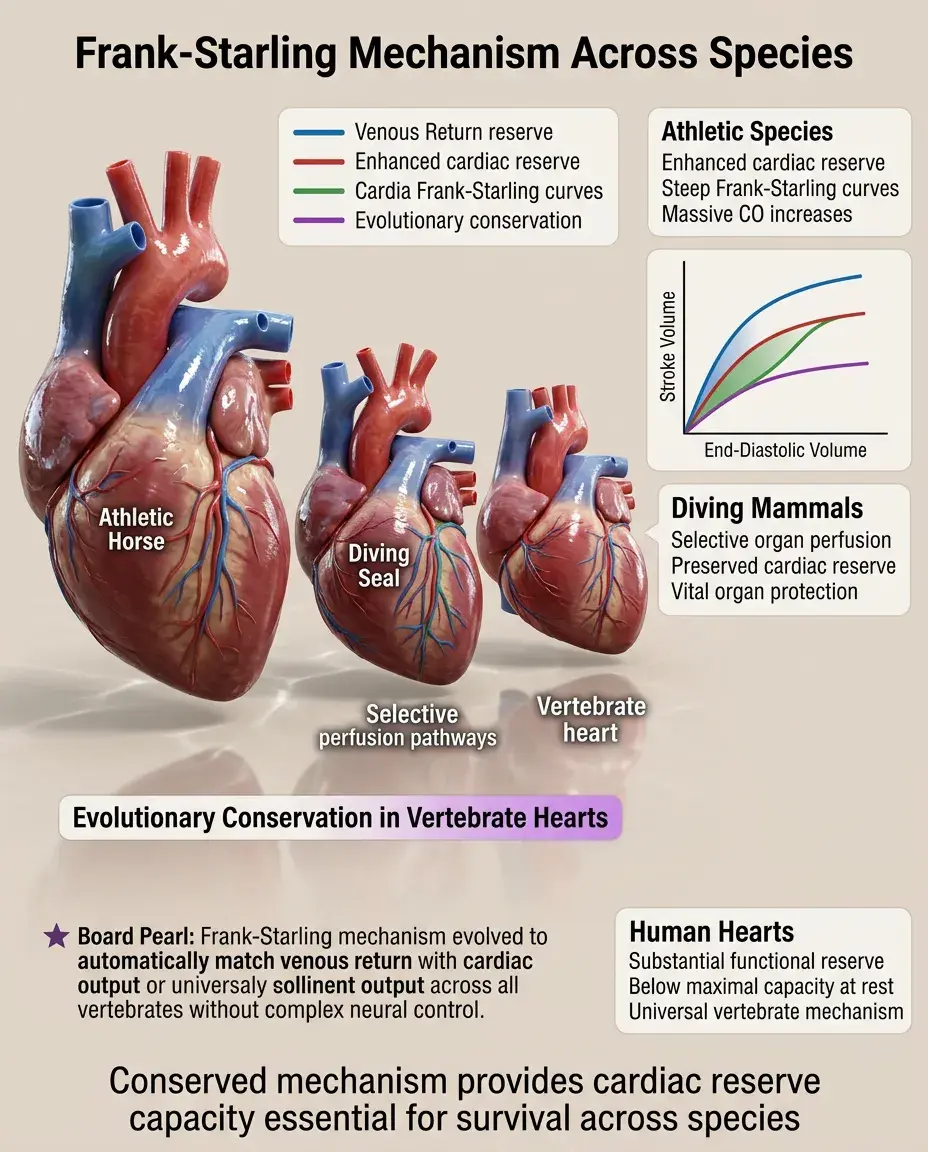
The Frank-Starling mechanism is conserved across all vertebrates, highlighting its fundamental importance to circulatory function.
🧠
Athletic species (horses, dogs) have particularly steep Frank-Starling curves allowing massive increases in cardiac output with exercise.
🧠
Diving mammals can selectively reduce peripheral perfusion while maintaining cardiac Frank-Starling reserve for vital organs.
🧠
The mechanism likely evolved to match venous return with cardiac output without requiring complex neural control.
🧠
Human hearts operate well below maximal Frank-Starling reserve under resting conditions, providing substantial functional reserve.
Mathematical Relationships and Board Calculations
⚡
Stroke volume = End-diastolic volume − End-systolic volume; Frank-Starling primarily affects SV by increasing EDV.
⚡
Ejection fraction = SV/EDV; in pure Frank-Starling, both numerator and denominator increase proportionally.
⚡
Cardiac output = SV × HR; Frank-Starling affects the SV component while autonomic control primarily affects HR.
⚡
LaPlace law: Wall tension = (Pressure × Radius) / (2 × Wall thickness); explains why dilated ventricles require more wall tension for the same pressure.
⚡
Board pearl: When calculating changes in cardiac output, consider both Frank-Starling effects on SV and autonomic effects on HR.

Clinical Scenarios and Frank-Starling Applications
📌
Hypovolemic shock: Operating on steep portion of curve, very preload-responsive, fluid resuscitation immediately improves cardiac output.
📌
Cardiogenic shock: Operating on flat portion of depressed curve, minimally preload-responsive, fluids worsen congestion without helping output.
📌
Distributive shock: Normal Frank-Starling curve but inadequate preload due to vasodilation, responds to fluids and vasopressors.
📌
Obstructive shock: Normal myocardial function but impaired filling (tamponade, tension pneumothorax), relieving obstruction restores Frank-Starling.
📌
Board distinction: Position on the Frank-Starling curve determines fluid responsiveness — steep portion benefits from fluids, flat portion does not.

Board Question Stem Patterns
📣
Increased venous return leading to increased stroke volume → Frank-Starling mechanism.
📣
Heart failure patient with high filling pressures but low cardiac output → operating on flat portion of depressed curve.
📣
Athlete with large stroke volume at rest → favorable Frank-Starling due to increased EDV and compliant ventricle.
📣
Loss of atrial kick causing decreased exercise tolerance → reduced preload contribution to Frank-Starling.
📣
Positive inotrope improving cardiac output at same filling pressure → upward shift of Frank-Starling curve.
📣
Leg elevation improving blood pressure in hypovolemic patient → increased preload via Frank-Starling.
📣
Diuresis improving dyspnea without decreasing cardiac output → moving leftward on flat portion of curve.

One-Line Recap
🔸
The Frank-Starling mechanism — the heart's intrinsic ability to increase stroke volume in response to increased venous return through optimal sarcomere stretch — provides beat-to-beat matching of cardiac output to venous return, operates on a curve that is depressed in heart failure and enhanced by inotropes, and determines fluid responsiveness based on whether the ventricle is operating on the steep (responsive) or flat (unresponsive) portion of its function curve.
bottom of page

