top of page
Human Development & Aging
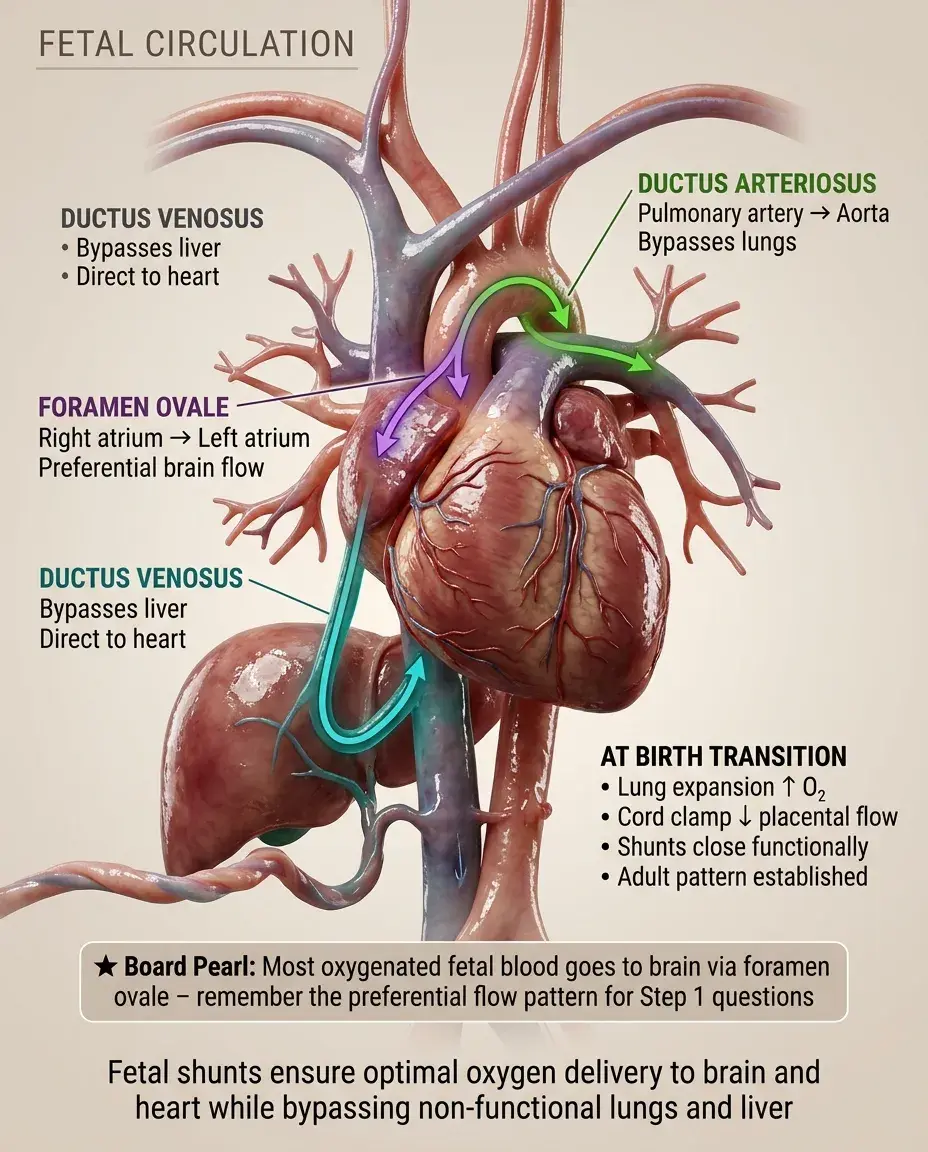
Fetal circulation
Core Principle of Fetal Circulation
🧷
Fetal circulation is fundamentally different from postnatal circulation because the placenta, not the lungs, serves as the organ of gas exchange.
🧷
The fetus exists in a state of relative hypoxia (PO₂ ~30 mmHg in umbilical vein) compared to postnatal life, yet this is physiologically normal and sufficient for fetal needs.
🧷
Three key shunts — the ductus venosus, foramen ovale, and ductus arteriosus — divert blood away from the non-functioning liver and lungs toward vital organs.
🧷
The right ventricle does most of the work in fetal circulation, pumping against high pulmonary vascular resistance, while the left ventricle supplies the brain and upper body with the most oxygenated blood available.

The Umbilical Circulation
📍
Two umbilical arteries carry deoxygenated blood from the fetus to the placenta, while one umbilical vein returns oxygenated blood (PO₂ ~30-35 mmHg) to the fetus.
📍
The umbilical vein enters at the umbilicus and travels through the falciform ligament toward the liver, where it divides.
📍
About 50% of umbilical venous blood perfuses the liver via the portal sinus, while the other 50% bypasses the liver through the ductus venosus to enter the inferior vena cava directly.
📍
Board pearl: Remember "2 arteries Away, 1 vein returning" — the umbilical arteries carry blood away from the fetus despite being called arteries.

The Ductus Venosus: First Shunt
🔹
The ductus venosus connects the umbilical vein to the inferior vena cava, allowing oxygenated placental blood to bypass the hepatic circulation.
🔹
This shunt ensures that the most oxygenated blood reaches the heart quickly rather than being diluted by deoxygenated hepatic venous blood.
🔹
The ductus venosus has a sphincter mechanism that regulates flow, increasing during hypoxia to maximize oxygen delivery to vital organs.
🔹
After birth, the ductus venosus constricts within minutes to hours and becomes the ligamentum venosum.
🔹
Board pearl: The ligamentum venosum is the adult remnant of the ductus venosus, found in the fissure between the caudate and left hepatic lobes.

Streaming in the Right Atrium
⭐
Blood entering the right atrium from the IVC does not mix uniformly but exhibits preferential streaming based on oxygen content.
⭐
The highly oxygenated blood from the ductus venosus preferentially streams across the foramen ovale into the left atrium due to the anatomical orientation of the IVC and the eustachian valve.
⭐
Meanwhile, deoxygenated blood from the SVC and coronary sinus preferentially flows through the tricuspid valve into the right ventricle.
⭐
This streaming phenomenon ensures that the brain and coronary circulation receive the most oxygenated blood available.
⭐
Board clue: This explains why the upper body is slightly pinker than the lower body in newborns with certain congenital heart defects.

The Foramen Ovale: Second Shunt
✅
The foramen ovale is a one-way flap valve between the right and left atria that allows right-to-left shunting of oxygenated blood.
✅
In utero, right atrial pressure exceeds left atrial pressure due to high pulmonary vascular resistance, keeping the foramen ovale open.
✅
Approximately one-third of IVC blood crosses the foramen ovale, bypassing the right ventricle and pulmonary circulation entirely.
✅
At birth, decreased pulmonary vascular resistance and increased systemic vascular resistance reverse the pressure gradient, functionally closing the foramen ovale.
✅
Board pearl: Patent foramen ovale (PFO) persists in 25-30% of adults and can cause paradoxical emboli (DVT → stroke).

The Ductus Arteriosus: Third Shunt
🧠
The ductus arteriosus connects the pulmonary artery to the descending aorta, allowing right ventricular output to bypass the high-resistance pulmonary circulation.
🧠
Approximately 90% of right ventricular output flows through the ductus arteriosus in utero, with only 10% perfusing the lungs.
🧠
The ductus remains open in utero due to high levels of prostaglandin E2 (PGE2) produced by the placenta and low oxygen tension.
🧠
Board pearl: Indomethacin (NSAID) closes a PDA by inhibiting PGE2 synthesis, while prostaglandin E1 keeps it open in duct-dependent congenital heart disease.
🧠
After birth, the ductus arteriosus becomes the ligamentum arteriosum.

Oxygen Saturation Geography in Fetal Circulation
⚡
Umbilical vein: ~80-85% saturation (highest in fetal circulation)
⚡
Left ventricle/ascending aorta: ~65% saturation (supplies brain and coronaries)
⚡
Right ventricle/pulmonary artery: ~55% saturation
⚡
Descending aorta: ~60% saturation (mixture of LV and ductus arteriosus blood)
⚡
Umbilical arteries: ~50% saturation (lowest, returning to placenta)
⚡
Board distinction: The brain receives more oxygenated blood than the lower body due to preferential streaming and the pre-ductal position of the carotid arteries.

Pulmonary Vascular Resistance in Utero
📌
Fetal lungs are fluid-filled and collapsed, creating extremely high pulmonary vascular resistance (PVR) that exceeds systemic vascular resistance (SVR).
📌
High PVR is maintained by hypoxic pulmonary vasoconstriction, mechanical compression of pulmonary vessels by fluid-filled alveoli, and low levels of vasodilators like nitric oxide.
📌
This high PVR is essential for shunting blood through the foramen ovale and ductus arteriosus.
📌
Only about 10% of combined ventricular output perfuses the lungs, just enough for lung development and metabolism.
📌
Board pearl: Maternal hyperoxia can decrease fetal PVR and increase pulmonary blood flow, a principle used in prenatal testing.

The Transitional Circulation at Birth
📣
The first breath expands the lungs, dramatically decreasing PVR by relieving mechanical compression and increasing alveolar oxygen tension.
📣
Clamping the umbilical cord removes the low-resistance placental circulation, suddenly increasing SVR.
📣
These pressure changes reverse the fetal pattern: left atrial pressure now exceeds right atrial pressure, closing the foramen ovale.
📣
Increased oxygen tension and decreased PGE2 cause the ductus arteriosus to constrict within 12-24 hours.
📣
Board concept: The transition is not instantaneous — it takes days to weeks for complete adaptation, explaining transitional murmurs and acrocyanosis in healthy newborns.

Persistent Fetal Circulation (PPHN)
🔸
Persistent pulmonary hypertension of the newborn occurs when PVR fails to decrease normally at birth, maintaining fetal circulatory patterns.
🔸
Right-to-left shunting continues through the foramen ovale and ductus arteriosus, causing profound hypoxemia unresponsive to oxygen.
🔸
Common causes include meconium aspiration, sepsis, congenital diaphragmatic hernia, and maternal SSRI use.
🔸
Board presentation: Term infant with respiratory distress, cyanosis, and a loud S2 with pre/post-ductal saturation difference >10%.
🔸
Treatment targets reducing PVR with oxygen, ventilation, inhaled nitric oxide, and in severe cases, ECMO.

Adult Remnants of Fetal Structures
🧷
Umbilical vein → ligamentum teres (round ligament of liver) in the falciform ligament
🧷
Ductus venosus → ligamentum venosum between caudate and left liver lobes
🧷
Foramen ovale → fossa ovalis (depression in interatrial septum)
🧷
Ductus arteriosus → ligamentum arteriosum connecting pulmonary artery to aortic arch
🧷
Umbilical arteries → medial umbilical ligaments and superior vesical arteries
🧷
Board pearl: These remnants are important surgical landmarks and can be sites of pathology (e.g., ligamentum teres recanalization in portal hypertension).

Maternal-Fetal Oxygen Exchange
📍
Despite the placenta being an efficient organ of gas exchange, fetal PO₂ remains low (~30 mmHg) due to limitations of placental diffusion and mixing of oxygenated and deoxygenated blood.
📍
The fetus compensates with fetal hemoglobin (HbF), which has higher oxygen affinity than adult hemoglobin due to poor binding of 2,3-BPG.
📍
The oxygen dissociation curve of HbF is left-shifted, allowing better oxygen loading at the low PO₂ of the placenta.
📍
Fetal blood also has higher hemoglobin concentration (15-20 g/dL) to maximize oxygen-carrying capacity.
📍
Board concept: The fetus thrives in "physiologic hypoxia" through multiple adaptations, not just circulatory shunts.

Regulation of Ductal Patency
🔹
The ductus arteriosus remains open in utero via high PGE2 (from placenta), low oxygen tension, and high levels of nitric oxide.
🔹
At birth, increased oxygen tension triggers smooth muscle constriction via oxygen-sensitive potassium channels and decreased PGE2 receptor sensitivity.
🔹
Initial functional closure occurs within 12-24 hours, but anatomical closure (fibrosis) takes 2-3 weeks.
🔹
Premature infants often have delayed ductal closure due to immature smooth muscle and persistent PGE2 sensitivity.
🔹
Board pearl: Duct-dependent lesions (e.g., critical coarctation, hypoplastic left heart) require PGE1 infusion to maintain ductal patency for survival.

Congenital Heart Disease and Fetal Circulation
⭐
Many congenital heart defects are well-tolerated in utero because of the parallel circulation and shunts that can compensate for anatomical abnormalities.
⭐
Lesions like transposition of the great arteries or hypoplastic left heart syndrome only become life-threatening after birth when shunts close.
⭐
Severe obstructions (critical aortic stenosis, pulmonary atresia) can be bypassed via the foramen ovale and ductus arteriosus.
⭐
Board distinction: "Duct-dependent" lesions require the ductus arteriosus for either pulmonary blood flow (right-sided lesions) or systemic blood flow (left-sided lesions).
⭐
Prenatal diagnosis allows for planned delivery at centers capable of immediate prostaglandin administration.

The Diving Reflex and Fetal Circulation
✅
The mammalian diving reflex shares similarities with fetal circulatory adaptations — both prioritize oxygen delivery to heart and brain during hypoxia.
✅
During fetal hypoxia, blood flow is redistributed away from the gut, kidneys, and periphery toward the brain, heart, and adrenals.
✅
This is mediated by selective vasoconstriction and the unique streaming patterns in fetal circulation.
✅
Chronic hypoxia leads to IUGR with asymmetric growth restriction (head-sparing) due to this preferential perfusion.
✅
Board application: Understanding this helps explain why placental insufficiency causes oligohydramnios (decreased renal perfusion) and increased middle cerebral artery flow.

Twin-Twin Transfusion Syndrome
🧠
In monochorionic twins, placental vascular anastomoses can create unbalanced blood flow between twins, essentially turning one twin into a parabiotic pump for the other.
🧠
The donor twin develops hypovolemia, oliguria, and oligohydramnios, while the recipient develops hypervolemia, polyuria, and polyhydramnios.
🧠
This represents a pathological extreme of fetal circulation where one fetus's cardiac output supplies both circulations.
🧠
Board presentation: Monochorionic twins with polyhydramnios in one sac and oligohydramnios ("stuck twin") in the other.
🧠
Without intervention, both twins are at risk — donor from hypovolemia and recipient from high-output heart failure.

Pharmacology and Fetal Circulation
⚡
Maternal medications cross the placenta and enter fetal circulation, but distribution differs from adults due to shunting patterns.
⚡
Drugs entering via the umbilical vein can bypass first-pass hepatic metabolism through the ductus venosus.
⚡
The blood-brain barrier is less developed in fetuses, increasing CNS exposure to drugs.
⚡
Board examples: Maternal labetalol can cause neonatal bradycardia; maternal magnesium causes neonatal hypotonia; maternal opioids cause neonatal abstinence syndrome.
⚡
Understanding fetal circulation helps predict drug effects and the timing of neonatal symptoms after delivery.

Circulatory Changes in Common Neonatal Conditions
📌
Sepsis: inflammatory mediators can reopen the ductus arteriosus and foramen ovale, reverting to fetal circulation patterns.
📌
Respiratory distress syndrome: hypoxia and acidosis increase PVR, maintaining right-to-left shunting.
📌
Congenital diaphragmatic hernia: lung hypoplasia causes fixed high PVR that cannot be overcome by normal transitional changes.
📌
Pneumothorax: can acutely increase PVR and reopen fetal shunts.
📌
Board pearl: Any condition causing hypoxia or acidosis in a newborn can trigger reversion to fetal circulation patterns with right-to-left shunting.

Board Question Stem Patterns
📣
Newborn with differential cyanosis (pink upper body, blue lower body) → right-to-left shunt through PDA with post-ductal coarctation.
📣
Premature infant with systolic murmur, wide pulse pressure, and bounding pulses → PDA with left-to-right shunting.
📣
Term infant with severe hypoxemia unresponsive to 100% oxygen → persistent pulmonary hypertension with fetal shunts.
📣
Echocardiogram shows right-to-left flow across atrial septum in newborn → elevated right-sided pressures, evaluate for PPHN.
📣
Adult with cryptogenic stroke and positive bubble study → paradoxical embolus through PFO.
📣
Fetal echo shows reversal of flow in ductus arteriosus → severe left heart obstruction.
📣
Newborn becomes cyanotic when PGE1 is stopped → duct-dependent congenital heart disease.

One-Line Recap
🔸
Fetal circulation uses three shunts (ductus venosus, foramen ovale, ductus arteriosus) to bypass the non-functioning lungs and liver, delivering the most oxygenated blood from the placenta preferentially to the brain and heart, with dramatic reversal at birth triggered by lung expansion and cord clamping that can be disrupted in various pathological states.
bottom of page

