top of page
Reproductive & Endocrine Systems
Endometrial Changes: Proliferative vs Secretory
Core Principle of Endometrial Cycling
🧷
The endometrium undergoes predictable, hormone-driven changes throughout the menstrual cycle to prepare for potential implantation of an embryo.
🧷
The cycle is divided into two major phases: proliferative (follicular) phase driven by estrogen, and secretory (luteal) phase driven by progesterone.
🧷
Understanding these phases is essential for interpreting endometrial biopsies, diagnosing causes of abnormal uterine bleeding, and recognizing pathologic states like hyperplasia and carcinoma.
🧷
Board pearl: The endometrium is the only tissue in the body that undergoes complete shedding and regeneration every month in response to hormonal signals.

The Proliferative Phase: Estrogen-Driven Growth
📍
The proliferative phase spans from menstruation to ovulation (days 1–14 in a 28-day cycle), though its length is variable.
📍
Rising estrogen from developing ovarian follicles stimulates endometrial regeneration after menstrual shedding.
📍
Histologic features: straight to slightly coiled glands, mitotic figures in glandular and stromal cells, thin compact stroma, and increasing endometrial thickness from 1–2 mm to 8–12 mm.
📍
The functional layer regenerates from the basal layer, which is preserved during menstruation.
📍
Board pearl: Proliferative endometrium on biopsy in a postmenopausal woman suggests unopposed estrogen exposure.

The Secretory Phase: Progesterone-Driven Differentiation
🔹
The secretory phase extends from ovulation to menstruation (days 15–28), with a consistent 14-day duration.
🔹
Progesterone from the corpus luteum halts proliferation and induces secretory transformation to support potential implantation.
🔹
Histologic features: tortuous "corkscrew" glands, subnuclear vacuoles (early secretory), luminal secretions, stromal edema, and prominent spiral arteries.
🔹
The endometrium reaches maximal thickness (10–16 mm) and becomes highly vascularized.
🔹
Board pearl: Dating secretory endometrium allows precise determination of cycle day, unlike the variable proliferative phase.

Early Secretory Phase Hallmarks (Days 16–19)
⭐
Day 16: First appearance of subnuclear vacuoles in glandular epithelium — the earliest sign of ovulation having occurred.
⭐
Day 17: Subnuclear vacuoles become uniform throughout the glands, creating a characteristic "piano key" appearance.
⭐
Day 18: Vacuoles migrate to the supranuclear position as secretions begin.
⭐
Day 19: Vacuoles disappear as secretory products are released into glandular lumens.
⭐
Board pearl: Subnuclear vacuoles are pathognomonic for post-ovulatory endometrium and confirm that ovulation has occurred.

Mid to Late Secretory Phase Features (Days 20–28)
✅
Day 20–21: Peak secretory activity with eosinophilic secretions filling dilated gland lumens.
✅
Day 22–23: Stromal edema becomes prominent, separating the glands and creating a "fluffy" appearance.
✅
Day 24–25: Spiral arteries become prominent; predecidual change begins around arterioles.
✅
Day 26–28: Extensive predecidual transformation, neutrophil infiltration, glandular exhaustion, and apoptosis herald impending menstruation.
✅
Board pearl: Predecidual change (stromal cells becoming large and polygonal) indicates the late secretory phase.

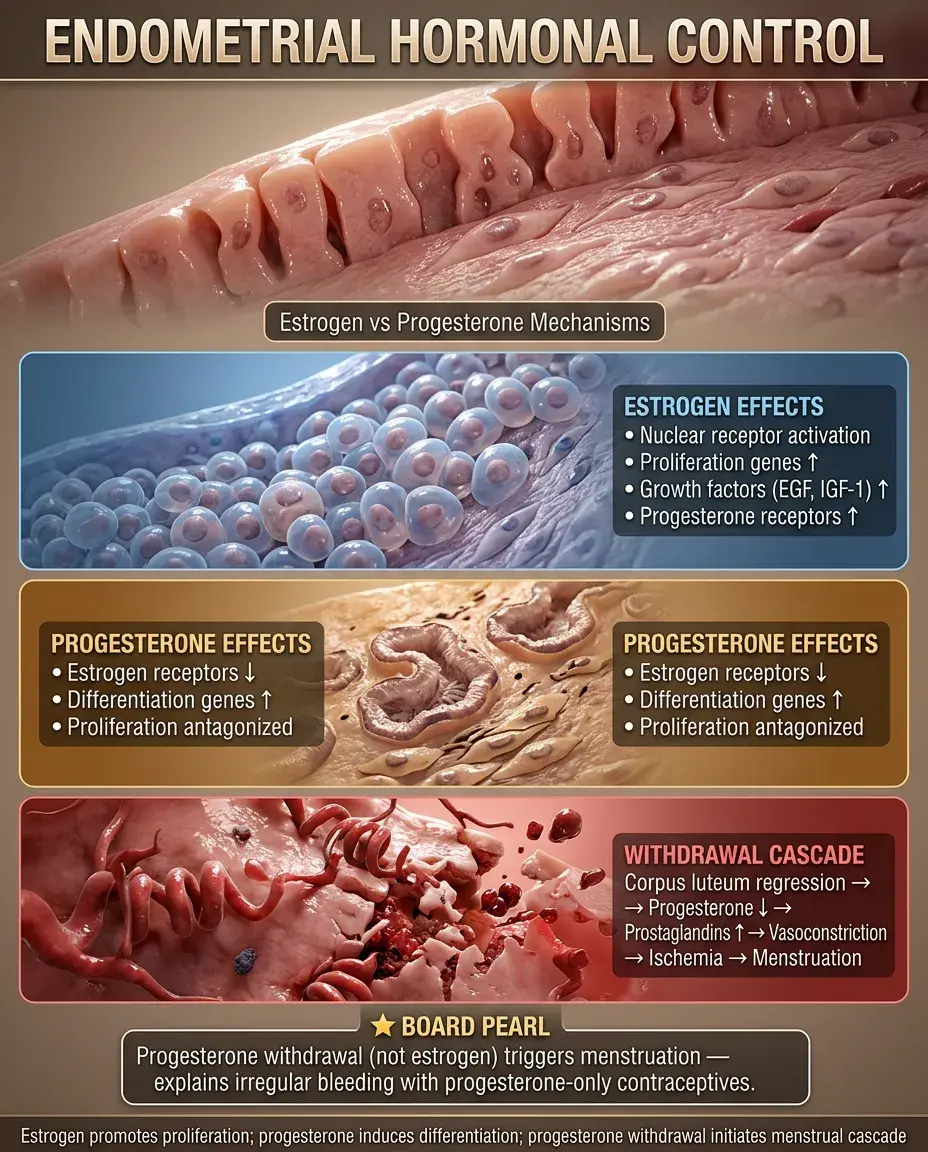
Hormonal Control Mechanisms
🧠
Estrogen acts through nuclear receptors to induce proliferation genes, growth factors (EGF, IGF-1), and progesterone receptor expression.
🧠
Progesterone antagonizes estrogen's proliferative effects by downregulating estrogen receptors and inducing differentiation genes.
🧠
Without pregnancy, corpus luteum regression → progesterone withdrawal → prostaglandin release → vasoconstriction → ischemia → menstruation.
🧠
Board pearl: Progesterone withdrawal, not estrogen withdrawal, triggers menstruation — explaining why progesterone-only contraceptives often cause irregular bleeding.
Clinical Correlation: Endometrial Dating
⚡
Endometrial dating determines whether the histologic appearance matches the expected cycle day based on last menstrual period.
⚡
"In-phase" endometrium: histology matches the cycle day (±2 days).
⚡
"Out-of-phase" endometrium: >2-day discrepancy suggests luteal phase defect, anovulation, or hormonal abnormality.
⚡
Dating is most accurate in the early secretory phase (days 16–19) due to the precise timing of subnuclear vacuole changes.
⚡
Board pearl: Luteal phase defect shows secretory endometrium that lags behind the expected date, suggesting inadequate progesterone.

Anovulatory Cycles and Persistent Proliferative Phase
📌
Without ovulation, no corpus luteum forms → no progesterone → continuous estrogen stimulation → persistent proliferative endometrium.
📌
Common causes: PCOS, perimenopause, hypothalamic dysfunction, obesity (peripheral aromatization), thyroid disorders.
📌
Clinical presentation: irregular, unpredictable bleeding as the thickened endometrium outgrows its blood supply and sheds irregularly.
📌
Histology shows proliferative features regardless of cycle day, often with breakdown and bleeding.
📌
Board pearl: Anovulatory bleeding is typically painless (no prostaglandins from secretory endometrium) and irregular.

Simple Hyperplasia Without Atypia
📣
Results from prolonged unopposed estrogen exposure → exaggerated proliferative response.
📣
Histology: increased gland-to-stroma ratio, crowded proliferative glands, cystic dilation ("Swiss cheese" pattern), no cytologic atypia.
📣
Risk factors: anovulation, obesity, estrogen therapy without progesterone, tamoxifen, granulosa cell tumors.
📣
Low malignant potential (<5% progression to carcinoma over 20 years).
📣
Board pearl: Simple hyperplasia often presents as heavy menstrual bleeding in obese perimenopausal women with PCOS.

Complex Hyperplasia and Atypical Hyperplasia
🔸
Complex hyperplasia: crowded glands with complex architecture (branching, budding) but no cytologic atypia. Intermediate cancer risk.
🔸
Atypical hyperplasia: cytologic atypia (nuclear enlargement, rounding, prominent nucleoli, loss of polarity) regardless of architecture.
🔸
Atypical hyperplasia has 25–40% risk of concurrent or future endometrial carcinoma.
🔸
EIN (endometrial intraepithelial neoplasia) is the preferred terminology, emphasizing its precancerous nature.
🔸
Board pearl: Atypical hyperplasia in postmenopausal women warrants hysterectomy due to high cancer risk.

Menstrual Phase Histology
🧷
Days 1–4: Endometrial shedding with fragmented glands and stroma, hemorrhage, neutrophils, and fibrin thrombi.
🧷
Prostaglandin F₂α causes spiral artery vasospasm → ischemia → tissue breakdown.
🧷
Only the functional layer sheds; the basal layer (supplied by straight arteries) remains intact.
🧷
Histologic features: fragmented tissue, collapsed glands, stromal breakdown, inflammatory infiltrate.
🧷
Board pearl: Menstrual endometrium is the only normal phase showing neutrophils — their presence elsewhere suggests infection or impending menstruation.

Endometrial Receptivity and Implantation Window
📍
The implantation window occurs days 20–24 (6–10 days post-ovulation) when the endometrium is optimally prepared.
📍
Key features: pinopodes (apical epithelial protrusions), optimal stromal edema, peak expression of adhesion molecules (integrins), and growth factors.
📍
Progesterone induces expression of LIF (leukemia inhibitory factor) and HOX genes essential for implantation.
📍
Asynchronous endometrium (out-of-phase) may contribute to implantation failure and recurrent pregnancy loss.
📍
Board pearl: The "window of implantation" corresponds to the mid-secretory phase when progesterone effects are maximal.

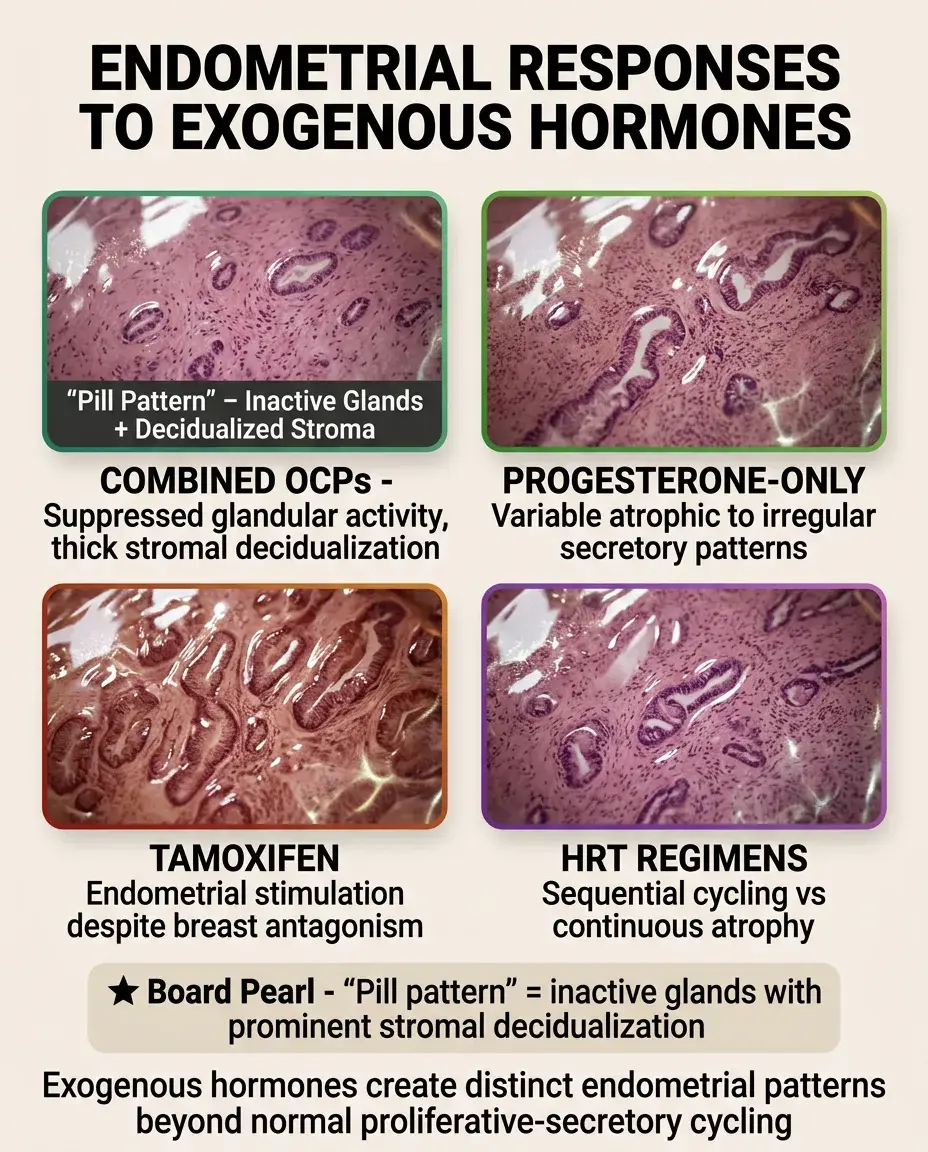
Effects of Exogenous Hormones
🔹
Combined oral contraceptives: inactive glands with decidualized stroma ("pill effect"), preventing normal cycling.
🔹
Progesterone-only methods: variable effects from atrophic to irregular secretory patterns, often with breakthrough bleeding.
🔹
Tamoxifen: anti-estrogenic in breast but agonist in endometrium → proliferative changes, polyps, hyperplasia, increased cancer risk.
🔹
HRT in menopause: sequential regimens mimic normal cycling; continuous combined therapy produces atrophic endometrium.
🔹
Board pearl: "Pill pattern" endometrium shows inactive glands with prominent stromal decidualization.
Postmenopausal Endometrium
⭐
Normal postmenopausal endometrium is atrophic: inactive glands, compact stroma, thickness <4–5 mm on ultrasound.
⭐
Any proliferative activity suggests abnormal estrogen exposure (obesity, HRT, ovarian tumor).
⭐
Postmenopausal bleeding always requires evaluation — even with atrophic endometrium, as early carcinomas may be focal.
⭐
Cystic atrophy: dilated inactive glands that can mimic hyperplasia but lack proliferative activity.
⭐
Board pearl: Endometrial thickness >5 mm in a postmenopausal woman not on hormones warrants biopsy.

Chronic Endometritis
✅
Defined by presence of plasma cells in endometrial stroma — the only reliable diagnostic criterion.
✅
Causes: retained products of conception, IUD, PID, tuberculosis (in endemic areas), actinomyces.
✅
Clinical features: abnormal bleeding, pelvic pain, infertility, recurrent pregnancy loss.
✅
Histology: plasma cells, lymphoid aggregates, stromal breakdown, disrupted normal cycling pattern.
✅
Board pearl: Special stains (CD138) may be needed to identify plasma cells, as they can be obscured by lymphocytes.

Endometrial Polyps
🧠
Benign overgrowths of endometrium containing glands, stroma, and thick-walled vessels.
🧠
More common in perimenopause; associated with tamoxifen use, obesity, hypertension.
🧠
Histology: irregular gland distribution, fibrous stroma, thick-walled vessels, may show various phases or hyperplasia.
🧠
Usually benign but 0.5–3% harbor malignancy, especially in postmenopausal women.
🧠
Board pearl: Polyps often show irregular cycling — proliferative glands may persist in secretory phase background.

Metaplasias and Epithelial Changes
⚡
Squamous metaplasia: benign response to chronic irritation, often associated with chronic endometritis or IUD.
⚡
Tubal metaplasia: ciliated cells resembling fallopian tube epithelium — common, benign, increases with age.
⚡
Eosinophilic metaplasia: cells with abundant eosinophilic cytoplasm, may mimic atypia but benign.
⚡
Arias-Stella reaction: pregnancy-related change with enlarged nuclei and clear cytoplasm — can mimic clear cell carcinoma.
⚡
Board pearl: Metaplasias are benign adaptive changes that should not be confused with atypia or malignancy.

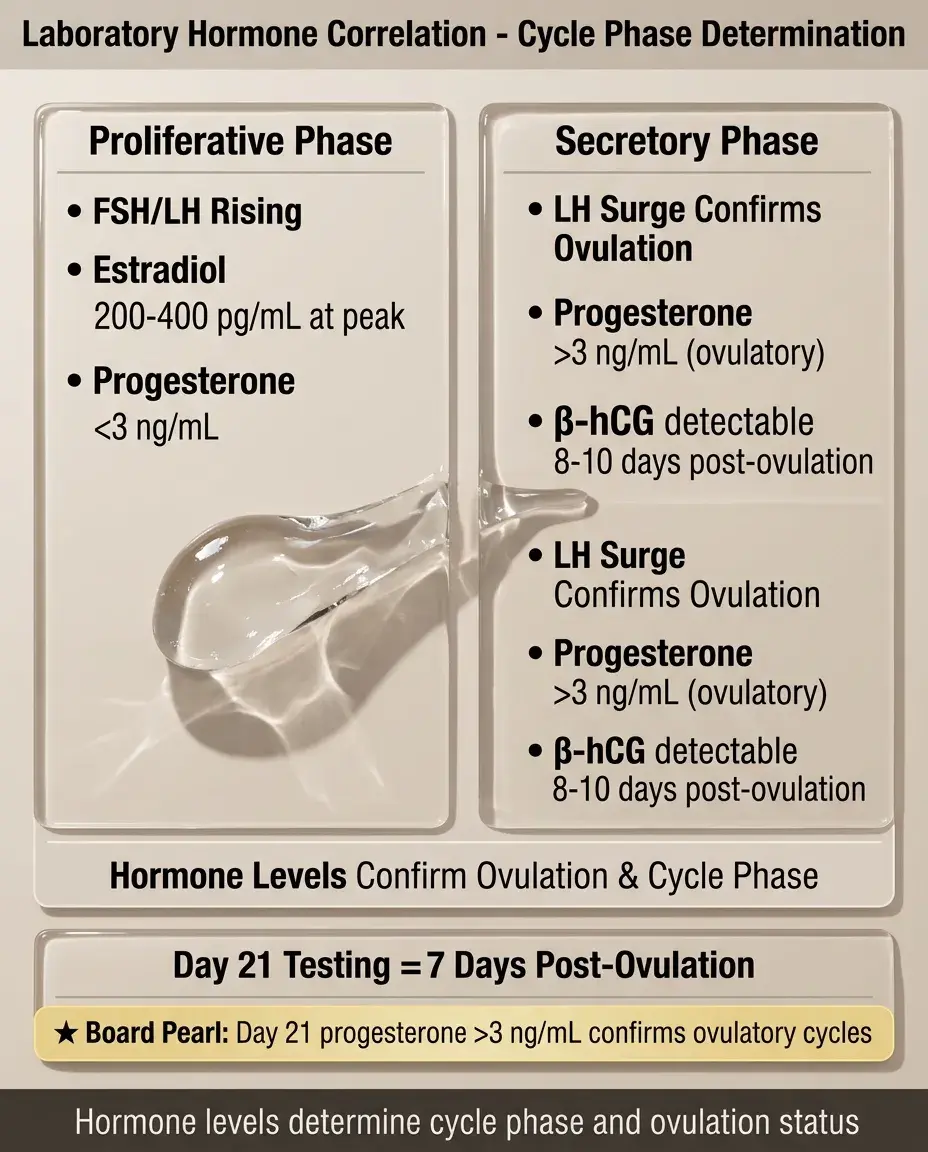
Laboratory Correlation
📌
FSH and LH levels help determine cycle phase and identify anovulation (LH surge absent).
📌
Progesterone >3 ng/mL in luteal phase confirms ovulation; <3 ng/mL suggests anovulation.
📌
Estradiol levels peak just before ovulation (200–400 pg/mL) then plateau in luteal phase.
📌
β-hCG becomes detectable 8–10 days after ovulation if pregnancy occurs.
📌
Board pearl: Day 21 progesterone level (7 days post-ovulation) is the standard test to confirm ovulatory cycles.
Board Question Stem Patterns
📣
Straight glands with mitoses in a 35-year-old on day 10 → normal proliferative phase.
📣
Subnuclear vacuoles in a woman trying to conceive → confirms ovulation has occurred.
📣
Persistent proliferative pattern in an obese 45-year-old with irregular bleeding → anovulation, consider hyperplasia.
📣
Inactive glands with decidualized stroma in a woman on OCPs → pill effect, expected finding.
📣
Plasma cells in endometrium of a woman with recurrent pregnancy loss → chronic endometritis.
📣
Postmenopausal woman with 8 mm endometrial thickness → biopsy indicated regardless of symptoms.
📣
Corkscrew glands with stromal edema on day 22 → normal secretory phase.

One-Line Recap
🔸
The endometrium cycles between estrogen-driven proliferative phase (straight glands, mitoses) and progesterone-driven secretory phase (tortuous glands, subnuclear vacuoles, stromal edema), with disruptions causing anovulatory bleeding, hyperplasia from unopposed estrogen, or out-of-phase endometrium affecting fertility — recognizable patterns essential for diagnosing abnormal uterine bleeding and infertility.

bottom of page

