top of page
Biostatistics & Epidemiology
Drug Safety Monitoring
Core Principle of Drug Safety Monitoring
🧷
Drug safety monitoring is the systematic surveillance of adverse drug reactions (ADRs) and medication errors throughout a drug's lifecycle — from preclinical studies through post-marketing surveillance.
🧷
The goal is to identify, characterize, and prevent harm to patients by detecting safety signals that may not have been apparent in pre-approval clinical trials.
🧷
This process involves multiple stakeholders: FDA, pharmaceutical companies, healthcare providers, and patients themselves.
🧷
Board pearl: Safety monitoring continues indefinitely after drug approval because rare adverse events may only become apparent after millions of patients are exposed.

Phases of Clinical Trials and Safety Assessment
📍
Phase I: First-in-human studies in healthy volunteers or patients to establish safety, tolerability, and pharmacokinetics. Primary goal is determining the maximum tolerated dose.
📍
Phase II: Efficacy and dose-finding studies in patients with the target condition. Safety monitoring intensifies as patient numbers increase.
📍
Phase III: Large randomized controlled trials comparing the new drug to standard therapy or placebo. Powered to detect efficacy but may miss rare adverse events.
📍
Phase IV: Post-marketing surveillance after FDA approval. Only now are sample sizes large enough to detect rare but serious adverse events.
📍
Board pearl: A drug withdrawn from market due to hepatotoxicity discovered after approval represents a Phase IV finding.

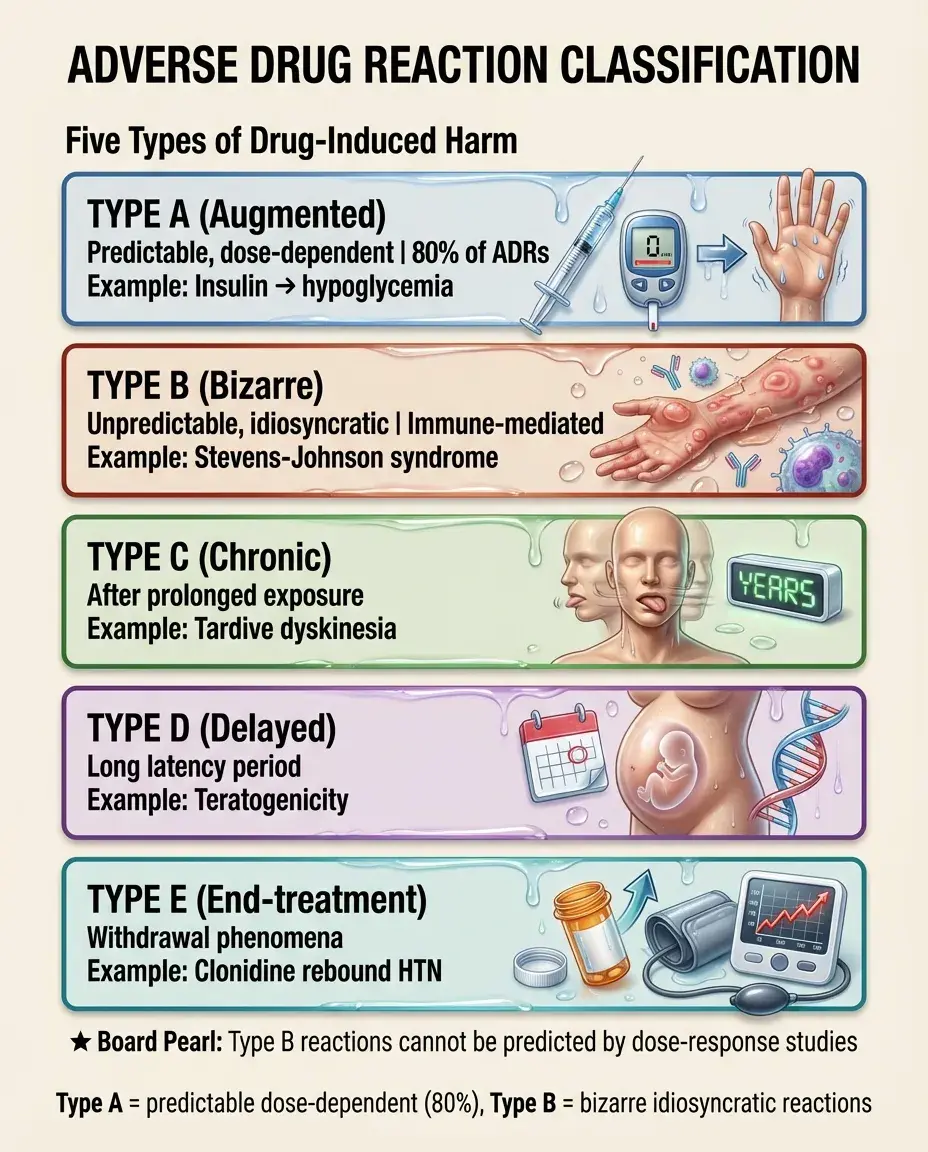
Types of Adverse Drug Reactions
🔹
Type A (Augmented): Predictable, dose-dependent extensions of the drug's pharmacologic effect. Account for ~80% of ADRs. Examples: hypoglycemia with insulin, bleeding with warfarin.
🔹
Type B (Bizarre): Unpredictable, idiosyncratic reactions unrelated to pharmacologic action. Often immune-mediated. Examples: Stevens-Johnson syndrome, drug-induced lupus.
🔹
Type C (Chronic): Reactions appearing after prolonged exposure. Example: tardive dyskinesia with antipsychotics.
🔹
Type D (Delayed): Reactions with long latency. Examples: teratogenicity, carcinogenicity.
🔹
Type E (End-of-treatment): Withdrawal phenomena. Example: rebound hypertension after clonidine discontinuation.
🔹
Board pearl: Type B reactions cannot be predicted by dose-response studies.
MedWatch and Spontaneous Reporting Systems
⭐
MedWatch is the FDA's voluntary reporting system for adverse events and medication errors involving drugs, biologics, medical devices, and dietary supplements.
⭐
Healthcare professionals and consumers can submit reports directly to FDA via Form 3500 (voluntary) or Form 3500A (mandatory for manufacturers).
⭐
Spontaneous reporting creates a database of potential safety signals but cannot establish causality or calculate true incidence rates due to underreporting and lack of denominator data.
⭐
Board pearl: If a physician suspects a serious adverse event from a newly approved drug, the appropriate action is to report it to MedWatch, not wait for more cases to accumulate.

Risk Evaluation and Mitigation Strategies (REMS)
✅
REMS are FDA-mandated risk management plans for drugs with serious safety concerns that would otherwise limit approval or continued marketing.
✅
Components may include: Medication Guides, patient package inserts, communication plans to healthcare providers, Elements to Assure Safe Use (ETASU).
✅
ETASU requirements can include: prescriber certification, pharmacy certification, patient enrollment in registry, documentation of safe-use conditions, monitoring requirements.
✅
Examples: isotretinoin (iPLEDGE program for teratogenicity), clozapine (monitoring for agranulocytosis), opioids (prescriber training).
✅
Board pearl: A drug requiring monthly pregnancy tests and two forms of contraception represents a REMS program addressing teratogenic risk.

Black Box Warnings
🧠
Black box warnings are the FDA's strongest safety warning, appearing in a black border at the beginning of the prescribing information.
🧠
Reserved for serious or life-threatening risks that can be mitigated through appropriate use.
🧠
Common categories: increased mortality risk (antipsychotics in dementia), suicidality (antidepressants in adolescents), hepatotoxicity, cardiovascular events.
🧠
Presence of a black box warning does not contraindicate use but mandates careful patient selection and monitoring.
🧠
Board pearl: Antidepressants carry a black box warning for increased suicidality in patients <24 years old, requiring close monitoring in the first months of treatment.

Signal Detection and Data Mining
⚡
A safety signal is information suggesting a new potentially causal association between a drug and an adverse event.
⚡
Disproportionality analysis compares the frequency of drug-event pairs in adverse event databases to expected background rates.
⚡
Data mining algorithms scan large databases for statistical associations that warrant further investigation.
⚡
Signals require clinical review to assess biological plausibility, temporality, and potential confounders before regulatory action.
⚡
Board pearl: Detection of a statistical signal in post-marketing data triggers further investigation, not immediate drug withdrawal.

Pharmacovigilance in Special Populations
📌
Pregnant women: Usually excluded from clinical trials. Safety data comes from pregnancy registries, case reports, and epidemiologic studies.
📌
Pediatric patients: Require specific safety studies due to developmental differences in drug metabolism and organ sensitivity.
📌
Elderly: Increased risk due to polypharmacy, altered pharmacokinetics, and comorbidities often excluded from trials.
📌
Patients with organ dysfunction: Hepatic or renal impairment alters drug clearance and increases toxicity risk.
📌
Board pearl: A drug causing phocomelia in exposed fetuses would likely have been missed in pre-approval trials since pregnant women are systematically excluded.

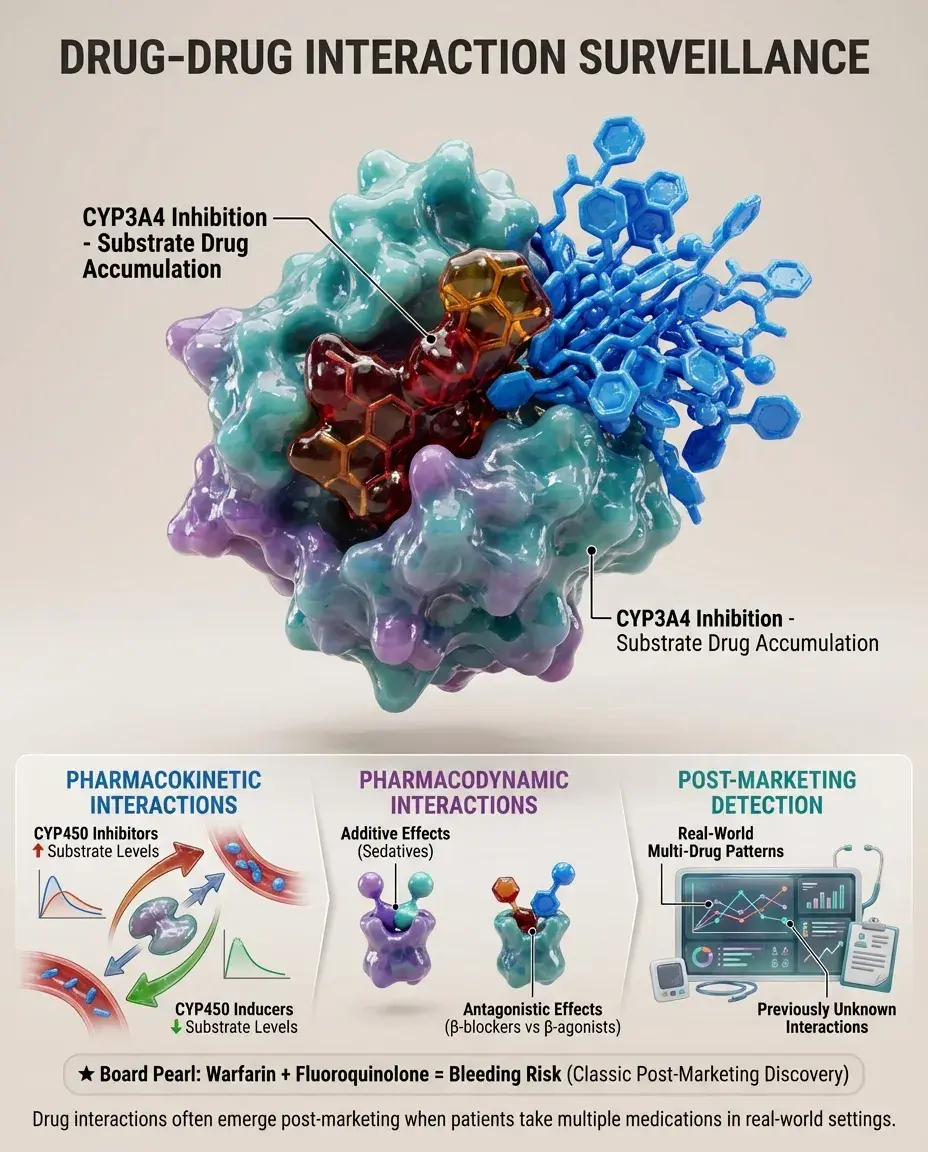
Drug-Drug Interaction Surveillance
📣
Many serious adverse events result from drug interactions not fully characterized during development.
📣
Cytochrome P450 interactions: Inhibitors increase substrate drug levels; inducers decrease them. CYP3A4 interactions are most common.
📣
Pharmacodynamic interactions: Additive effects (sedatives), antagonistic effects (β-blockers and β-agonists), or unpredictable synergies.
📣
Post-marketing surveillance reveals real-world interaction patterns in patients taking multiple medications.
📣
Board pearl: A patient on warfarin who develops bleeding after starting a fluoroquinolone represents a post-marketing drug interaction discovery.
Medication Error Reporting and Prevention
🔸
Medication errors include prescribing errors, transcription errors, dispensing errors, administration errors, and monitoring failures.
🔸
Institute for Safe Medication Practices (ISMP) maintains databases of error-prone abbreviations, look-alike/sound-alike drugs, and high-alert medications.
🔸
System-based interventions (computerized order entry, bar coding, standardized protocols) are more effective than targeting individual providers.
🔸
Root cause analysis identifies system failures contributing to errors rather than assigning individual blame.
🔸
Board pearl: Implementing bar-code medication administration reduces errors more effectively than education alone.

Vaccine Safety Monitoring Systems
🧷
Vaccine Adverse Event Reporting System (VAERS): Passive surveillance system co-managed by FDA and CDC for post-licensure monitoring.
🧷
Vaccine Safety Datalink (VSD): Active surveillance using electronic health records from large healthcare organizations to conduct epidemiologic studies.
🧷
Clinical Immunization Safety Assessment (CISA) project: Provides expert consultation for complex vaccine safety questions.
🧷
Special considerations: temporal clustering of events, background rates of conditions, and vaccine-preventable disease risks.
🧷
Board pearl: Intussusception risk with rotavirus vaccine was detected through post-marketing surveillance after the first vaccine was withdrawn.

International Drug Safety Collaboration
📍
World Health Organization (WHO) Programme for International Drug Monitoring coordinates global pharmacovigilance through Uppsala Monitoring Centre.
📍
International Conference on Harmonisation (ICH) creates standardized safety reporting guidelines used across regulatory agencies.
📍
MedDRA (Medical Dictionary for Regulatory Activities) provides standardized terminology for adverse event coding internationally.
📍
Safety signals detected in one country trigger investigations globally, enabling faster recognition of rare events.
📍
Board pearl: A drug withdrawn in Europe for hepatotoxicity would prompt immediate FDA review of U.S. safety data.

Benefit-Risk Assessment
🔹
Drug approval and continued marketing require ongoing assessment that benefits outweigh risks in the intended population.
🔹
Factors considered: severity of the condition treated, availability of alternatives, magnitude of benefit, frequency and severity of adverse events.
🔹
Risk tolerance varies by indication: higher risks accepted for cancer drugs than for symptomatic relief medications.
🔹
Patient preferences and quality of life impacts factor into individual benefit-risk decisions.
🔹
Board pearl: A drug causing rare but fatal hepatotoxicity might remain available for treating life-threatening conditions lacking alternatives.

Drug Safety Communication
⭐
FDA Drug Safety Communications alert healthcare providers and patients to new safety information requiring action.
⭐
Dear Healthcare Provider letters notify prescribers directly of important safety updates or label changes.
⭐
Medication Guides provide FDA-approved patient information for drugs with serious risks requiring patient awareness for safe use.
⭐
Risk communication must balance providing adequate warning without causing unwarranted alarm or medication non-adherence.
⭐
Board pearl: Discovery of QT prolongation risk with a marketed drug triggers immediate safety communication while formal label revision is pending.

Post-Marketing Study Requirements
✅
FDA can require post-marketing studies (Phase IV commitments) as a condition of drug approval when questions remain about safety or efficacy.
✅
Post-marketing requirements (PMRs) are studies required by statute; post-marketing commitments (PMCs) are agreed to by manufacturers.
✅
Common reasons: long-term safety, pediatric studies, drug-drug interactions, evaluation in patients with organ impairment.
✅
Failure to complete required studies can result in regulatory action including drug withdrawal.
✅
Board pearl: A drug approved based on surrogate endpoints typically has PMR to confirm clinical benefit.

Teratogenicity Surveillance
🧠
Pregnancy exposure registries prospectively collect outcomes data on women inadvertently exposed to medications during pregnancy.
🧠
Retrospective studies using birth defect registries and administrative databases identify potential teratogenic signals.
🧠
Animal reproductive toxicity studies have limited predictive value for human teratogenicity.
🧠
Thalidomide tragedy led to current requirements for reproductive toxicity testing and pregnancy prevention programs.
🧠
Board pearl: A new antiepileptic drug would require a pregnancy registry given the class teratogenicity risk.

Hepatotoxicity Monitoring
⚡
Drug-induced liver injury (DILI) is a leading cause of drug withdrawal and black box warnings.
⚡
Hy's Law: ALT/AST >3× ULN plus bilirubin >2× ULN without alkaline phosphatase elevation indicates severe hepatocellular injury with 10% mortality.
⚡
Regular monitoring may detect injury before clinical symptoms but cannot prevent idiosyncratic reactions.
⚡
Genetic markers (HLA alleles) increasingly used to identify at-risk patients for certain drugs.
⚡
Board pearl: A patient with ALT 5× ULN and bilirubin 3× ULN on a new drug meets Hy's Law criteria, predicting potential for fatal hepatotoxicity.

Cardiovascular Safety Assessment
📌
ICH E14 guideline requires thorough QT studies for all new drugs to assess torsades de pointes risk.
📌
QTc prolongation >20 ms or absolute QTc >500 ms indicates significant arrhythmia risk requiring risk mitigation.
📌
Major adverse cardiovascular events (MACE): composite endpoint of cardiovascular death, myocardial infarction, and stroke.
📌
Post-marketing cardiovascular outcome trials required for diabetes drugs after rosiglitazone controversy.
📌
Board pearl: A drug causing QTc prolongation would contraindicate concurrent use with other QT-prolonging medications.

Board Question Stem Patterns
📣
New drug withdrawn 2 years after approval due to rare liver failure → Phase IV surveillance finding.
📣
Pregnant patient inadvertently exposed to Category X drug → report to pregnancy exposure registry.
📣
Patient develops Stevens-Johnson syndrome on new antibiotic → Type B adverse reaction, report to MedWatch.
📣
Drug requires monthly laboratory monitoring and restricted distribution → REMS with ETASU program.
📣
Clinical trial stopped early due to increased deaths in treatment arm → Data Safety Monitoring Board action.
📣
Medication error due to similar drug names → system-based intervention more effective than education.

One-Line Recap
🔸
Drug safety monitoring encompasses the continuous surveillance of adverse events from preclinical development through post-marketing, utilizing spontaneous reporting systems, mandated REMS programs, black box warnings, and international collaboration to detect, communicate, and mitigate risks that threaten the benefit-risk balance of marketed medications.

bottom of page

