top of page
Reproductive & Endocrine Systems
Disorders of spermatogenesis
Core Principle of Spermatogenesis Disorders
🧷
Spermatogenesis is the 74-day process of transforming diploid spermatogonial stem cells into haploid spermatozoa within seminiferous tubules, requiring precise hormonal regulation (FSH, LH, testosterone) and optimal testicular temperature (2–3°C below core body temperature).
🧷
Disorders manifest as oligospermia (<15 million sperm/mL), azoospermia (no sperm), or abnormal morphology/motility, accounting for 40–50% of couple infertility.
🧷
The key diagnostic framework separates pre-testicular (hypothalamic-pituitary), testicular (primary), and post-testicular (obstructive) causes.
🧷
Board pearl: Low testosterone with low FSH/LH = secondary hypogonadism; low testosterone with elevated FSH/LH = primary hypogonadism.

Hormonal Regulation of Spermatogenesis
📍
GnRH from hypothalamus → LH and FSH from anterior pituitary → LH stimulates Leydig cells to produce testosterone; FSH acts on Sertoli cells to support spermatogenesis.
📍
Testosterone concentrations within seminiferous tubules must be 50–100× higher than serum levels for normal spermatogenesis.
📍
Negative feedback: testosterone inhibits GnRH/LH; inhibin B from Sertoli cells selectively inhibits FSH.
📍
Board pearl: Exogenous testosterone suppresses LH → decreased intratesticular testosterone → impaired spermatogenesis and testicular atrophy.
📍
This explains why anabolic steroid users develop small, firm testes despite high serum testosterone levels.

Klinefelter Syndrome (47,XXY)
🔹
Most common genetic cause of male infertility (1:500–1,000 males), presenting with tall stature, gynecomastia, small firm testes, and azoospermia.
🔹
Seminiferous tubule dysgenesis → fibrosis and hyalinization → elevated FSH/LH, low-normal testosterone, undetectable inhibin B.
🔹
Associated features: learning disabilities, increased risk of breast cancer, osteoporosis, diabetes, and autoimmune disorders.
🔹
Diagnosis: karyotype analysis showing 47,XXY (or mosaic variants).
🔹
Board pearl: The combination of small firm testes + gynecomastia + elevated FSH in a tall male = Klinefelter syndrome until proven otherwise.

Y Chromosome Microdeletions
⭐
Second most common genetic cause of severe oligospermia/azoospermia, affecting the AZF (azoospermia factor) regions on Yq11.
⭐
AZFa deletion → Sertoli cell-only syndrome (complete absence of germ cells).
⭐
AZFb deletion → maturation arrest at primary spermatocyte stage.
⭐
AZFc deletion → hypospermatogenesis with variable phenotype, most common deletion (60–70% of cases).
⭐
Board pearl: Y microdeletions are transmitted to male offspring through assisted reproduction, so genetic counseling is mandatory before ICSI.
⭐
Testing indicated for severe oligospermia (<5 million/mL) or azoospermia with normal karyotype.

Varicocele Pathophysiology
✅
Dilated pampiniform plexus veins (90% left-sided due to perpendicular insertion into renal vein) present in 15% of men, 40% of infertile men.
✅
Mechanisms of impaired spermatogenesis: increased scrotal temperature, testicular hypoxia, reflux of renal/adrenal metabolites, oxidative stress.
✅
Physical exam: "bag of worms" that increases with Valsalva, decreases when supine; graded I (palpable with Valsalva), II (palpable without Valsalva), III (visible).
✅
Board pearl: New-onset right varicocele or varicocele that doesn't decompress when supine → evaluate for retroperitoneal mass obstructing venous drainage.
✅
Varicocele repair improves sperm parameters in 60–70% but pregnancy rates in only 30–40%.

Cryptorchidism and Temperature Effects
🧠
Undescended testis affects 3% of term infants, 30% of premature infants; 80% descend spontaneously by 1 year.
🧠
Optimal spermatogenesis requires 34–35°C; each 1°C increase → 14% decline in sperm production.
🧠
Germ cell damage begins after age 1–2 years if undescended; bilateral cryptorchidism → 90% infertility rate if untreated.
🧠
Orchiopexy before age 2 improves fertility potential but doesn't normalize it; cancer risk remains elevated (3–5× general population).
🧠
Board pearl: Retractile testis (can be brought into scrotum, stays briefly) has normal fertility; true cryptorchidism requires surgical correction.

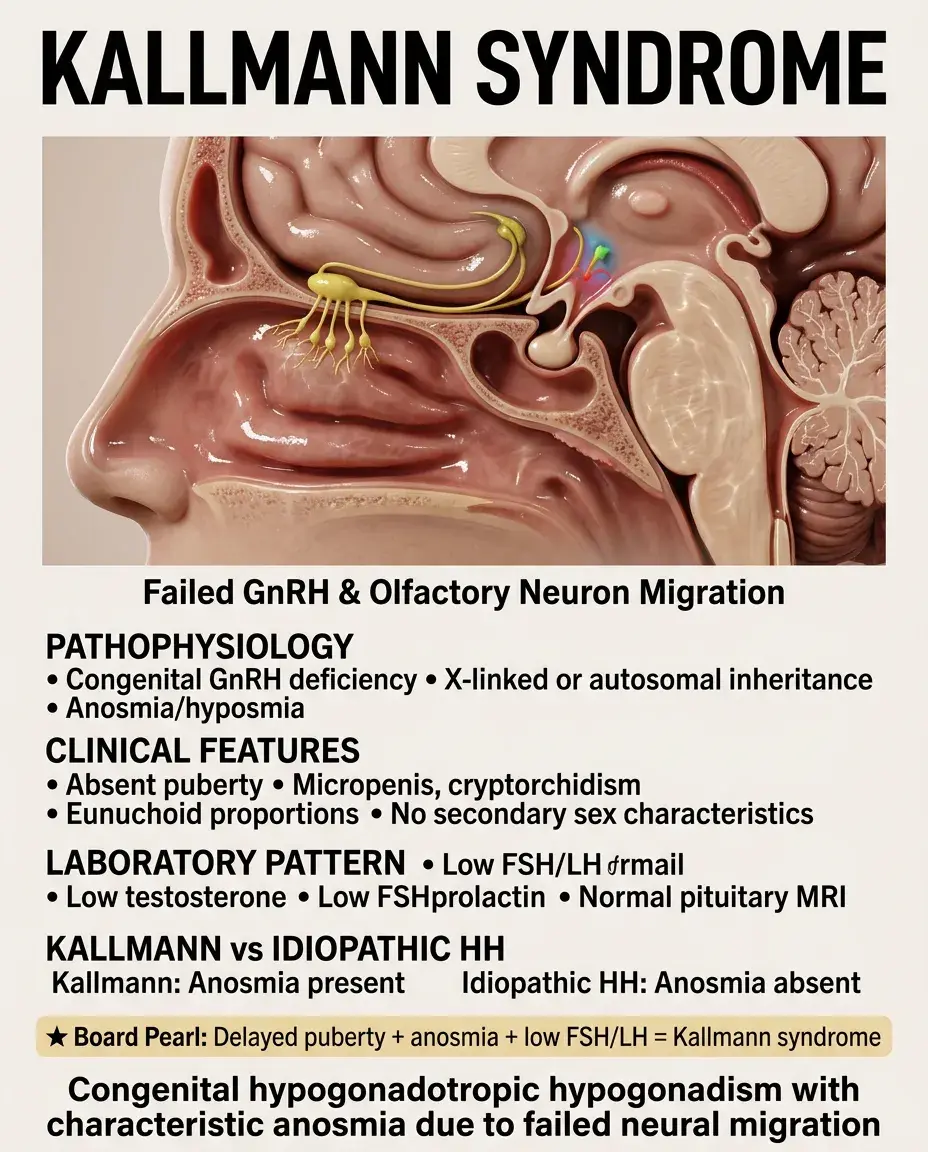
Kallmann Syndrome
⚡
Congenital GnRH deficiency + anosmia/hyposmia due to failed migration of GnRH neurons and olfactory neurons during development.
⚡
X-linked (KAL1), autosomal dominant (FGFR1, PROKR2), or autosomal recessive inheritance patterns.
⚡
Clinical features: absent puberty, micropenis, cryptorchidism, eunuchoid proportions (arm span > height), absent secondary sexual characteristics.
⚡
Laboratory: low testosterone, low FSH/LH (distinguishes from primary hypogonadism), normal prolactin, normal MRI pituitary.
⚡
Board pearl: Delayed puberty + anosmia + low FSH/LH = Kallmann syndrome; without anosmia = idiopathic hypogonadotropic hypogonadism.
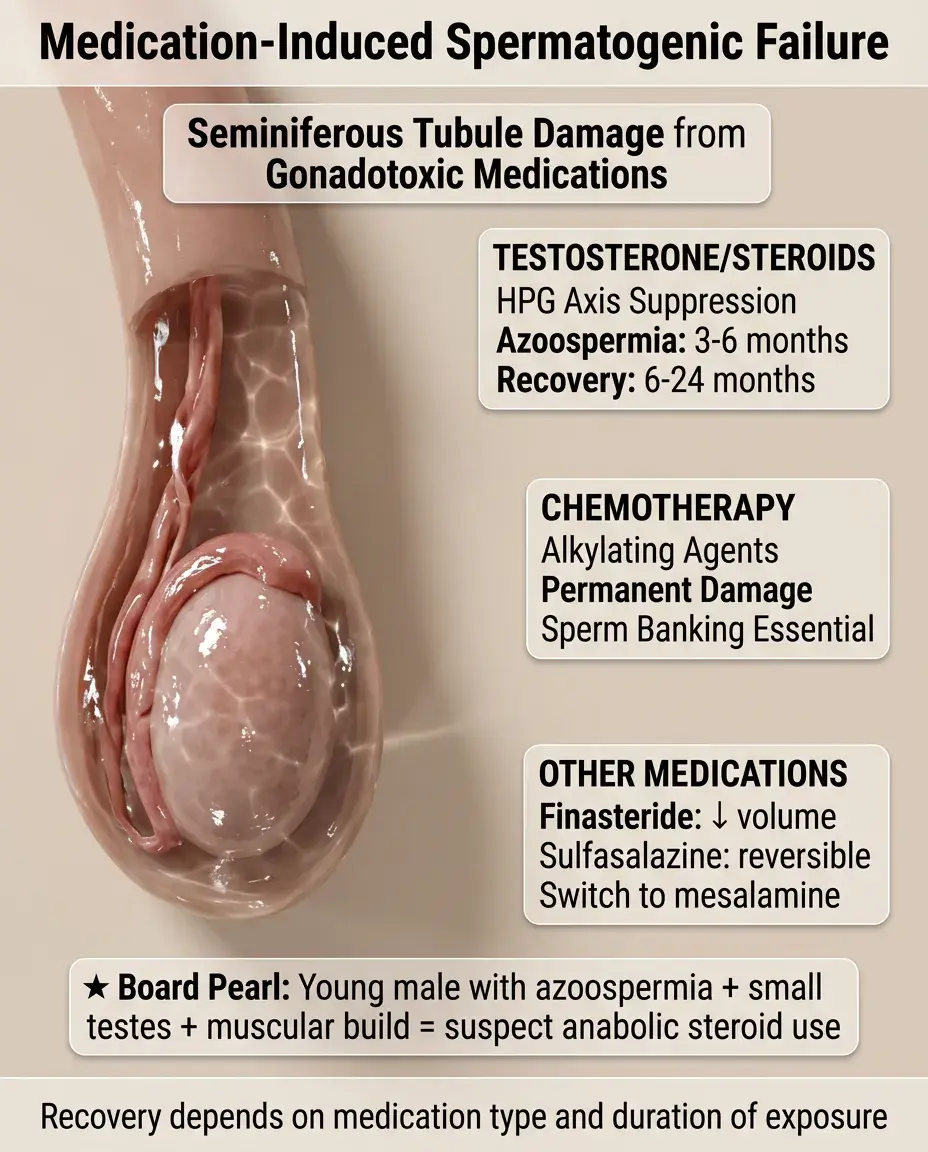
Medication-Induced Spermatogenic Failure
📌
Testosterone/anabolic steroids: suppress HPG axis → azoospermia in 3–6 months, recovery takes 6–24 months after cessation.
📌
Chemotherapy: alkylating agents (cyclophosphamide, busulfan) cause dose-dependent, often permanent damage; always offer sperm banking.
📌
5α-reductase inhibitors (finasteride): reduce ejaculate volume, may decrease sperm concentration.
📌
Sulfasalazine: reversible oligospermia through unknown mechanism; switch to mesalamine if possible.
📌
Board pearl: Young male with azoospermia, small testes, and muscular physique → suspect anabolic steroid use even if denied.
Sertoli Cell-Only Syndrome
📣
Complete absence of germ cells with only Sertoli cells lining seminiferous tubules → azoospermia with normal-sized testes.
📣
Causes: Y chromosome deletions (AZFa), chemotherapy, radiation, viral orchitis, idiopathic (most common).
📣
FSH markedly elevated (no inhibin B production), LH elevated, testosterone low-normal.
📣
Testicular biopsy shows tubules containing only Sertoli cells, no spermatogenesis.
📣
Board pearl: Azoospermia + normal testicular volume + very high FSH (>15–20 IU/L) suggests Sertoli cell-only syndrome or maturation arrest.

Maturation Arrest
🔸
Spermatogenesis halts at a specific stage: spermatogonial, primary spermatocyte (most common), or spermatid level.
🔸
Causes include genetic defects, heat exposure, toxins, idiopathic; presents as severe oligospermia or azoospermia.
🔸
FSH elevated but less than Sertoli cell-only syndrome; testicular volume usually reduced but not severely.
🔸
Testicular histology shows normal early stages up to arrest point, then absence of more mature forms.
🔸
Board distinction: Sertoli cell-only = no germ cells at all; maturation arrest = germ cells present but incomplete development.

Obstructive Azoospermia
🧷
Normal spermatogenesis but physical blockage prevents sperm from reaching ejaculate; accounts for 40% of azoospermia.
🧷
Causes: congenital bilateral absence of vas deferens (CBAVD), ejaculatory duct obstruction, vasectomy, epididymal obstruction from infection.
🧷
Normal FSH and testicular volume (unlike non-obstructive azoospermia); fructose absent if seminal vesicles obstructed.
🧷
CBAVD associated with CFTR mutations — test patient and partner for CF carrier status.
🧷
Board pearl: Azoospermia + normal FSH + normal testicular volume + low ejaculate volume (<1.5 mL) = obstructive azoospermia.

Retrograde Ejaculation
📍
Semen flows backward into bladder instead of urethra due to incompetent bladder neck closure during ejaculation.
📍
Causes: diabetes (autonomic neuropathy), TURP, retroperitoneal lymph node dissection, α-blockers, spinal cord injury.
📍
Presents as "dry orgasm" or low-volume ejaculate (<1 mL) with absent or few sperm.
📍
Diagnosis: post-ejaculatory urinalysis shows >10–15 sperm per high-power field.
📍
Board pearl: Diabetic male with low ejaculate volume and infertility → check post-ejaculatory urine for sperm before extensive workup.

Primary Ciliary Dyskinesia (Kartagener Syndrome)
🔹
Defective ciliary structure/function → immotile sperm flagella + chronic sinusitis + bronchiectasis ± situs inversus (50%).
🔹
Autosomal recessive mutations in dynein arm proteins; incidence 1:15,000–30,000.
🔹
Sperm count normal but motility near zero; electron microscopy shows absent or defective dynein arms.
🔹
Associated with recurrent respiratory infections from birth due to impaired mucociliary clearance.
🔹
Board pearl: Male infertility + chronic sinusitis + bronchiectasis = primary ciliary dyskinesia; add situs inversus = Kartagener syndrome.

Endocrine Disruptors and Environmental Factors
⭐
Occupational exposures: pesticides (DBCP), heavy metals (lead, cadmium), organic solvents, heat exposure in bakers/welders.
⭐
Endocrine disruptors: phthalates, BPA, organochlorines → anti-androgenic effects, decreased sperm quality.
⭐
Lifestyle factors: obesity (aromatization of testosterone to estrogen), smoking (oxidative damage), marijuana (decreased sperm concentration).
⭐
Anabolic steroids: most severe impact, complete azoospermia possible within months.
⭐
Board pearl: History of pesticide exposure + oligospermia + peripheral neuropathy suggests DBCP (dibromochloropropane) toxicity.

Immunologic Infertility
✅
Antisperm antibodies (ASA) develop after blood-testis barrier disruption: vasectomy, trauma, infection, torsion.
✅
ASA coat sperm surface → impaired motility, acrosome reaction, sperm-egg binding.
✅
Direct mixed antiglobulin reaction (MAR) test or immunobead test detects antibody-bound sperm.
✅
>50% sperm with adherent antibodies considered significant; treatment options limited.
✅
Board pearl: Previous vasectomy reversal + normal sperm count but poor motility → suspect antisperm antibodies.

Genetic Testing Indications
🧠
Karyotype: severe oligospermia (<5 million/mL), azoospermia, suspected Klinefelter syndrome.
🧠
Y chromosome microdeletion: non-obstructive azoospermia or severe oligospermia with normal karyotype.
🧠
CFTR mutations: congenital bilateral absence of vas deferens, obstructive azoospermia with absent vas.
🧠
Androgen receptor gene: suspected androgen insensitivity with undervirilization.
🧠
Board pearl: The threshold for genetic testing is sperm concentration <5 million/mL, not the normal reference of 15 million/mL.

Semen Analysis Parameters
⚡
WHO 2010 criteria: volume ≥1.5 mL, concentration ≥15 million/mL, total motility ≥40%, progressive motility ≥32%, morphology ≥4% normal forms.
⚡
Abstinence 2–7 days before collection; analyze within 1 hour; repeat abnormal results after 3 months (one full spermatogenic cycle).
⚡
Oligospermia: <15 million/mL; severe: <5 million/mL; azoospermia: no sperm after centrifugation.
⚡
Board pearl: Single abnormal semen analysis never diagnostic — always repeat due to high intra-individual variability.
⚡
Fever can suppress spermatogenesis for up to 3 months.

Testicular Biopsy and TESE
📌
Indicated to differentiate obstructive from non-obstructive azoospermia when non-invasive tests equivocal.
📌
Testicular sperm extraction (TESE): retrieve sperm for ICSI in non-obstructive azoospermia; success rate 30–60%.
📌
Microsurgical TESE (micro-TESE): uses operating microscope to identify seminiferous tubules likely to contain sperm.
📌
Finding sperm in non-obstructive azoospermia allows biological paternity through ICSI.
📌
Board pearl: Even in Sertoli cell-only syndrome, focal areas of spermatogenesis may exist, making micro-TESE worthwhile.

Board Question Stem Patterns
📣
Young male + gynecomastia + small firm testes + tall stature → Klinefelter syndrome.
📣
Infertility + chronic sinusitis + bronchiectasis → primary ciliary dyskinesia/Kartagener syndrome.
📣
Delayed puberty + anosmia + low FSH/LH → Kallmann syndrome.
📣
Azoospermia + normal testicular volume + normal FSH → obstructive azoospermia.
📣
Low ejaculate volume + diabetes → retrograde ejaculation, check post-ejaculatory urine.
📣
Muscular male + small testes + azoospermia → anabolic steroid use.
📣
Right varicocele that doesn't decompress supine → evaluate for retroperitoneal mass.
📣
Previous chemotherapy + azoospermia + elevated FSH → chemotherapy-induced testicular failure.

One-Line Recap
🔸
Spermatogenesis disorders divide into pre-testicular (low FSH/LH), testicular (high FSH/LH), and post-testicular (normal FSH) causes, with genetic evaluation indicated for severe oligospermia (<5 million/mL), recognizing that exogenous testosterone paradoxically causes azoospermia, varicoceles impair sperm through heat/hypoxia, and even severe cases like Klinefelter syndrome may have focal spermatogenesis retrievable by micro-TESE for assisted reproduction.

bottom of page

