top of page
Reproductive & Endocrine Systems
Cushing syndrome (ACTH-dependent vs independent)
Core Principle of Cushing Syndrome
🧷
Cushing syndrome is a state of pathologic hypercortisolism — excess cortisol from any source leading to a characteristic clinical syndrome.
🧷
The fundamental diagnostic branch point is determining whether the hypercortisolism is ACTH-dependent (driven by ACTH) or ACTH-independent (autonomous cortisol production).
🧷
ACTH-dependent causes account for ~80% of cases: pituitary adenoma (Cushing disease) and ectopic ACTH production.
🧷
ACTH-independent causes account for ~20%: adrenal adenoma, carcinoma, or bilateral hyperplasia.
🧷
This distinction drives all subsequent workup because it localizes the problem to either ACTH overproduction or direct adrenal pathology.

Clinical Features of Hypercortisolism
📍
Central obesity with supraclavicular and dorsocervical fat pads ("buffalo hump"), moon facies, and abdominal striae — purple striae are particularly specific.
📍
Proximal muscle weakness from protein catabolism, easy bruising from capillary fragility, and poor wound healing.
📍
Hypertension (cortisol has mineralocorticoid activity), glucose intolerance (cortisol opposes insulin), and osteoporosis (inhibits osteoblasts).
📍
Neuropsychiatric symptoms: depression, emotional lability, cognitive impairment, and frank psychosis in severe cases.
📍
Board pearl: Purple striae >1 cm wide are highly specific for Cushing syndrome, unlike the pink striae of simple obesity.

Confirming Hypercortisolism: The First Step
🔹
Before localizing the source, you must first confirm pathologic hypercortisolism exists.
🔹
Three screening tests have equivalent sensitivity: 24-hour urine free cortisol, overnight 1-mg dexamethasone suppression test, and late-night salivary cortisol.
🔹
24-hour UFC: measures unbound cortisol excretion; values >3× upper limit of normal are diagnostic.
🔹
Overnight DST: 1 mg dexamethasone at 11 PM should suppress morning cortisol to <1.8 μg/dL; failure indicates hypercortisolism.
🔹
Late-night salivary cortisol: loss of diurnal rhythm with elevated nighttime cortisol.
🔹
Board clue: At least two abnormal tests are required for diagnosis.

The Physiology Behind ACTH Dependence
⭐
In ACTH-dependent Cushing syndrome, excess ACTH drives bilateral adrenal hyperplasia and cortisol overproduction.
⭐
Pituitary source (Cushing disease): ACTH-secreting adenoma partially responsive to negative feedback — high-dose dexamethasone can suppress.
⭐
Ectopic ACTH: typically from small cell lung cancer or bronchial carcinoid; completely autonomous — no suppression with dexamethasone.
⭐
In ACTH-independent disease, the adrenals produce cortisol autonomously while ACTH is suppressed by negative feedback.
⭐
Key concept: ACTH levels differentiate these pathways — elevated in ACTH-dependent, suppressed in ACTH-independent.

ACTH Measurement: The Critical Branch Point
✅
Once hypercortisolism is confirmed, measure morning ACTH to determine dependence.
✅
ACTH <5 pg/mL → ACTH-independent (adrenal source); proceed to adrenal imaging.
✅
ACTH >20 pg/mL → ACTH-dependent; proceed to high-dose dexamethasone suppression test.
✅
ACTH 5-20 pg/mL → indeterminate; consider CRH stimulation test.
✅
The ACTH assay is sensitive to sample handling — must be collected in EDTA tube, kept on ice, and processed quickly to prevent degradation.
✅
Board pearl: Low ACTH with hypercortisolism = adrenal source; high ACTH = pituitary or ectopic source.

High-Dose Dexamethasone Suppression Test
🧠
Used to distinguish pituitary from ectopic ACTH sources in ACTH-dependent disease.
🧠
Protocol: 8 mg dexamethasone at 11 PM or 2 mg every 6 hours for 48 hours.
🧠
Pituitary adenomas retain partial feedback sensitivity → cortisol suppresses to <50% of baseline.
🧠
Ectopic ACTH sources are completely autonomous → no suppression.
🧠
Sensitivity ~80% for pituitary source, but some pituitary macroadenomas and carcinoid tumors may not follow expected patterns.
🧠
Board distinction: Suppression with high-dose dex = pituitary; no suppression = ectopic.

Inferior Petrosal Sinus Sampling (IPSS)
⚡
The gold standard for differentiating pituitary from ectopic ACTH when biochemical tests are equivocal.
⚡
Catheters sample ACTH from bilateral inferior petrosal sinuses (draining the pituitary) and peripheral blood simultaneously.
⚡
Central-to-peripheral ACTH ratio >2.0 at baseline or >3.0 after CRH stimulation confirms pituitary source.
⚡
Lateralization (>1.4 ratio between sides) can guide surgical approach but is less reliable.
⚡
Invasive procedure with small risk of stroke; reserved for cases where noninvasive testing is inconclusive.
⚡
Board pearl: IPSS is the most accurate test to localize ACTH source when imaging is negative.

Imaging in ACTH-Dependent Disease
📌
Pituitary MRI: indicated when biochemical testing suggests Cushing disease; ~50% of ACTH adenomas are microadenomas <10 mm.
📌
Problem: 10% of normal people have incidental pituitary lesions — imaging alone cannot diagnose Cushing disease.
📌
If MRI shows adenoma >6 mm concordant with biochemical testing → proceed to transsphenoidal surgery.
📌
If MRI negative or shows microadenoma <6 mm → consider IPSS for confirmation.
📌
For suspected ectopic ACTH: CT chest/abdomen/pelvis to find source (lung, thymus, pancreas, adrenal medulla).
📌
Board concept: Never rely on imaging alone — biochemical confirmation is mandatory.

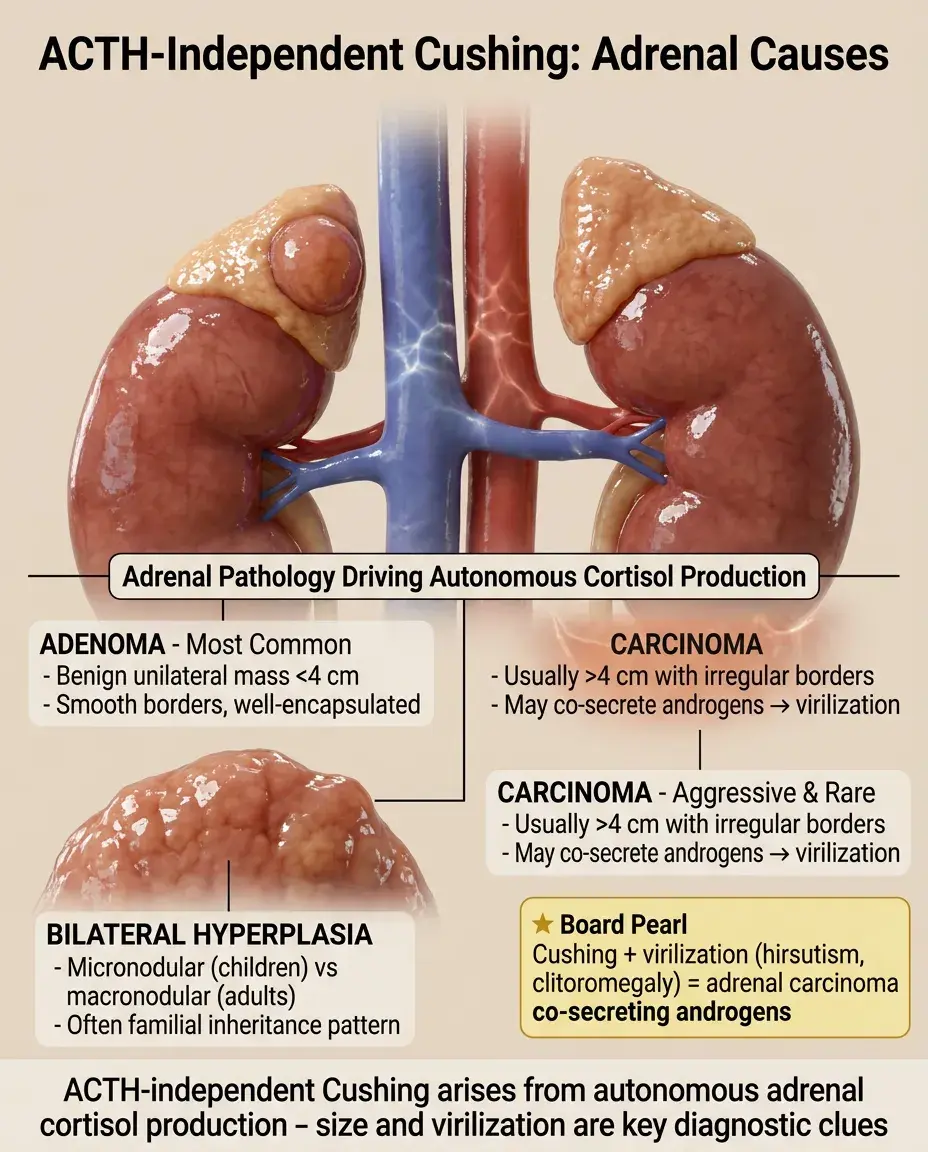
ACTH-Independent Cushing: Adrenal Causes
📣
Adrenal adenoma: most common cause of ACTH-independent Cushing; benign, usually unilateral, <4 cm.
📣
Adrenal carcinoma: rare, aggressive; typically >4 cm with irregular borders, may co-secrete androgens causing virilization.
📣
Bilateral adrenal hyperplasia: rare; can be micronodular (children) or macronodular (adults), often familial.
📣
Primary pigmented nodular adrenal disease (PPNAD): associated with Carney complex; paradoxical rise in cortisol with dexamethasone.
📣
Board clue: Cushing syndrome with virilization (hirsutism, clitoromegaly) → think adrenal carcinoma co-secreting androgens.
Adrenal Imaging and Characterization
🔸
CT or MRI adrenals for all ACTH-independent cases.
🔸
Adenoma features: <4 cm, homogeneous, low attenuation (<10 HU), >50% washout on delayed imaging.
🔸
Carcinoma features: >4 cm, heterogeneous, irregular borders, invasion of adjacent structures, delayed washout.
🔸
Bilateral disease may require adrenal vein sampling to determine if one or both glands are hyperfunctioning.
🔸
FDG-PET can help distinguish benign from malignant lesions — carcinomas are hypermetabolic.
🔸
Board distinction: Size >4 cm, heterogeneity, and poor washout suggest carcinoma over adenoma.

Special Situations: Cyclic and Subclinical Cushing
🧷
Cyclic Cushing: episodic cortisol secretion with periods of normal levels; requires multiple measurements over time to catch peaks.
🧷
Subclinical Cushing: autonomous cortisol secretion without classic clinical features; often discovered incidentally on adrenal imaging.
🧷
Pregnancy: increases cortisol-binding globulin → elevated total cortisol but normal free cortisol; use 24-hour UFC for screening.
🧷
Pseudo-Cushing: depression, alcoholism, severe illness can cause mild hypercortisolism that resolves with treatment of underlying condition.
🧷
Board pearl: Repeated normal tests don't exclude cyclic Cushing if clinical suspicion remains high.

Ectopic ACTH Syndrome: Clinical Clues
📍
Rapid onset with severe symptoms: profound weakness, hypokalemia (from mineralocorticoid excess), metabolic alkalosis.
📍
Less likely to have classic cushingoid body habitus due to rapid course — may present primarily with hypertension and hyperglycemia.
📍
Small cell lung cancer: most common source, very poor prognosis, extremely high ACTH and cortisol levels.
📍
Bronchial carcinoid: slower onset, may have more typical Cushing features, better prognosis.
📍
Occult sources: may require serial imaging over years; somatostatin receptor imaging can help localize neuroendocrine tumors.
📍
Board clue: Severe hypokalemia with metabolic alkalosis in Cushing → think ectopic ACTH.

Laboratory Patterns by Etiology
🔹
Pituitary adenoma: ACTH elevated (20-200 pg/mL), suppresses with high-dose dex, IPSS shows central gradient.
🔹
Ectopic ACTH: ACTH often very high (>200 pg/mL), no suppression with dex, no central gradient on IPSS.
🔹
Adrenal adenoma: ACTH suppressed (<5 pg/mL), contralateral adrenal atrophy on imaging.
🔹
Adrenal carcinoma: ACTH suppressed, may have elevated DHEAS and other adrenal androgens.
🔹
Board pattern: The combination of ACTH level + dexamethasone response usually identifies the source.

Complications of Chronic Hypercortisolism
⭐
Cardiovascular: hypertension (80% of patients), left ventricular hypertrophy, increased thrombotic risk.
⭐
Metabolic: diabetes/glucose intolerance, dyslipidemia, central obesity with increased visceral adiposity.
⭐
Bone: osteoporosis with vertebral fractures, avascular necrosis (especially femoral head).
⭐
Infectious: immunosuppression → opportunistic infections including PCP, cryptococcus, nocardia.
⭐
Psychiatric: depression (most common), cognitive impairment, psychosis in severe cases.
⭐
Board pearl: Unexplained osteoporosis in a young patient → screen for Cushing syndrome.

Nelson Syndrome: A Specific Complication
✅
Occurs after bilateral adrenalectomy for Cushing disease when the underlying pituitary adenoma is not removed.
✅
Loss of cortisol feedback → unchecked ACTH adenoma growth → mass effect and extreme ACTH elevation.
✅
Classic triad: hyperpigmentation (ACTH cross-reacts with melanocyte receptors), visual field defects, elevated ACTH.
✅
Prevention: pituitary radiation after adrenalectomy if adenoma not resected.
✅
Treatment: transsphenoidal surgery, radiation, or medical therapy with pasireotide.
✅
Board association: Hyperpigmentation after bilateral adrenalectomy = Nelson syndrome.

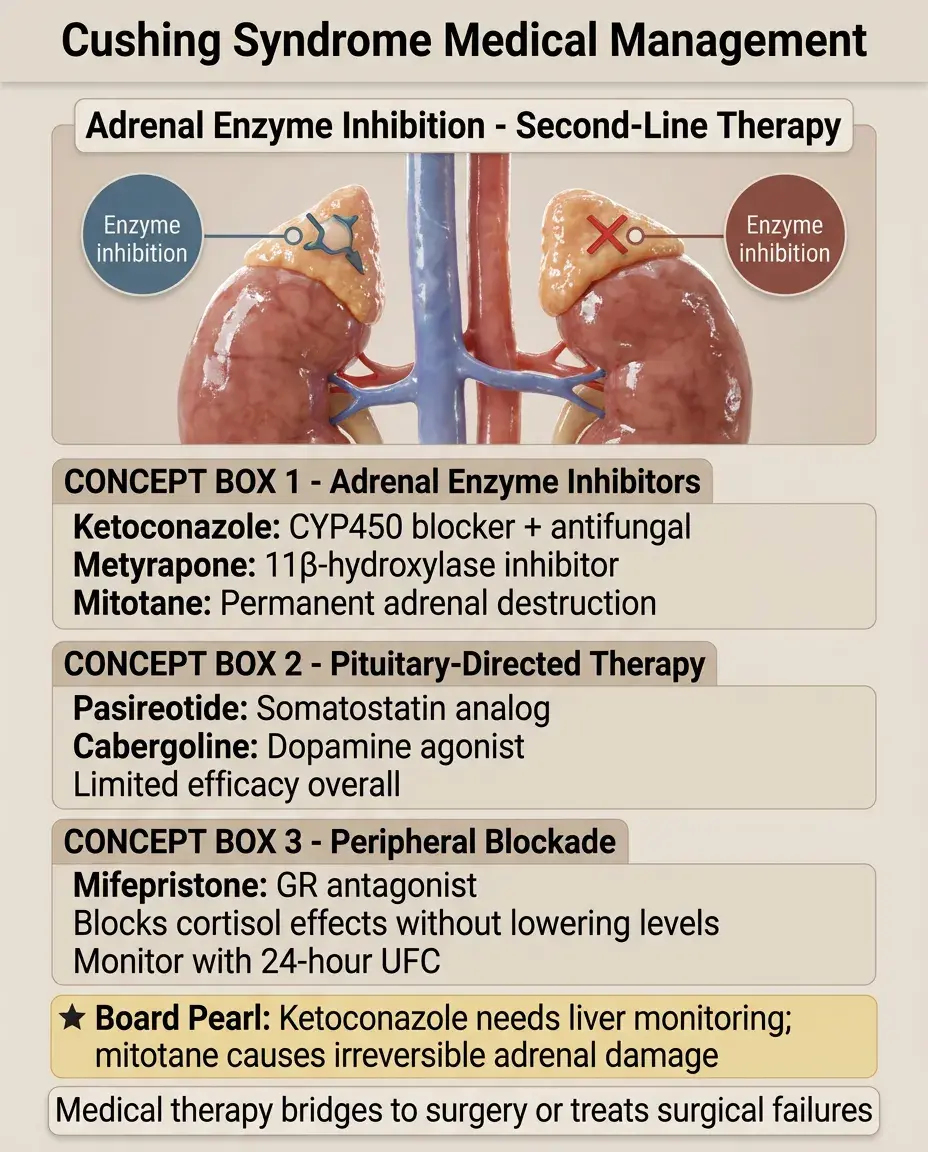
Medical Management Principles
🧠
Medical therapy is second-line, used when surgery fails or as a bridge to definitive treatment.
🧠
Adrenal enzyme inhibitors: ketoconazole (also antifungal), metyrapone (11β-hydroxylase inhibitor), mitotane (adrenolytic).
🧠
Pituitary-directed: pasireotide (somatostatin analog), cabergoline (dopamine agonist) — limited efficacy.
🧠
Glucocorticoid receptor antagonist: mifepristone — blocks peripheral cortisol effects without lowering levels.
🧠
Monitor effectiveness with 24-hour UFC; clinical improvement may lag biochemical normalization.
🧠
Board concept: Ketoconazole requires liver function monitoring; mitotane causes permanent adrenal destruction.
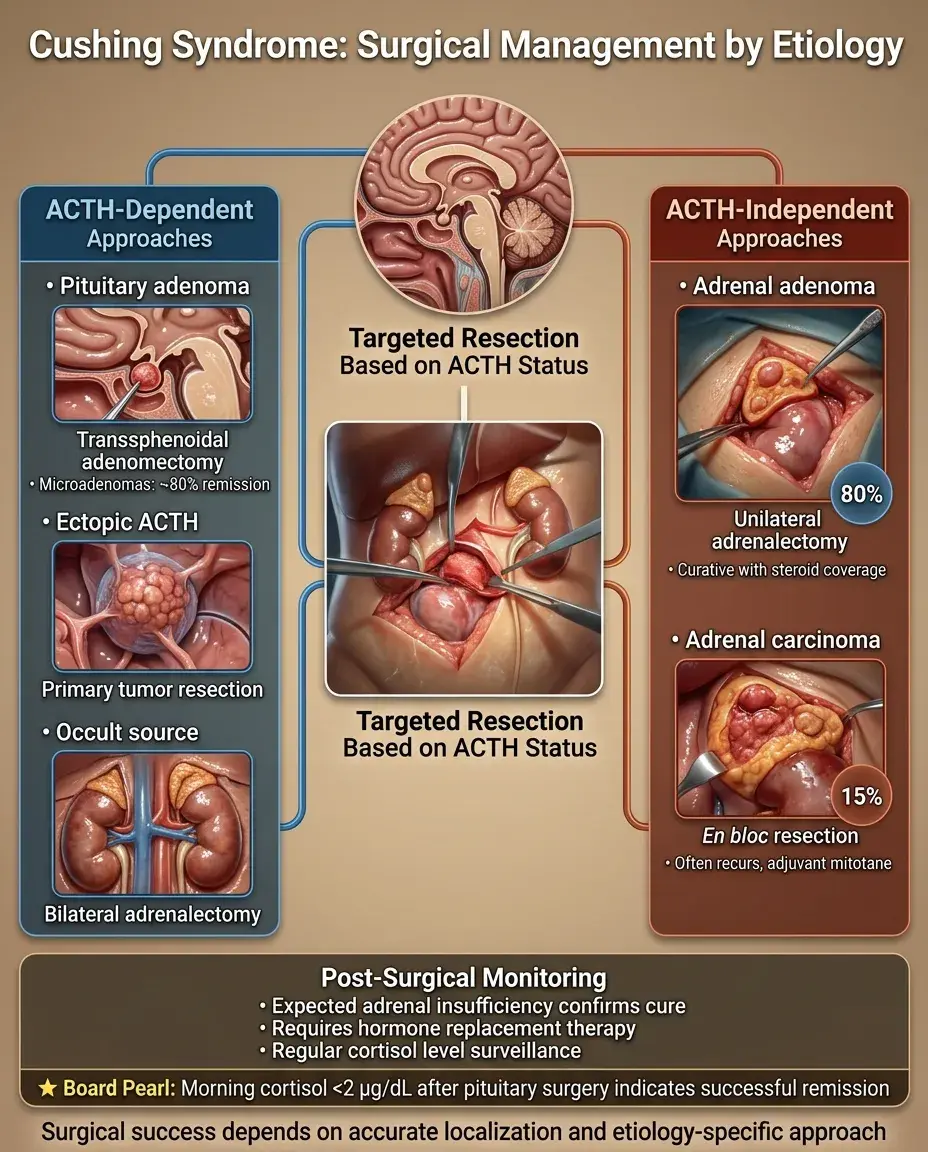
Surgical Management by Etiology
⚡
Cushing disease: transsphenoidal adenomectomy, ~80% remission rate for microadenomas, lower for macroadenomas.
⚡
Ectopic ACTH: resect primary tumor if localized and resectable; if occult, consider bilateral adrenalectomy.
⚡
Adrenal adenoma: unilateral adrenalectomy, curative; requires perioperative steroid coverage for contralateral suppression.
⚡
Adrenal carcinoma: en bloc resection if feasible; often recurs, may need adjuvant mitotane.
⚡
Post-operative adrenal insufficiency is expected after successful surgery — confirms cure but requires replacement.
⚡
Board pearl: Morning cortisol <2 μg/dL after pituitary surgery indicates remission.
Post-Treatment Monitoring
📌
After successful treatment, the HPA axis remains suppressed for 6-18 months.
📌
Glucocorticoid replacement required until axis recovers — taper based on morning cortisol and ACTH stimulation testing.
📌
Monitor for recurrence: Cushing disease recurs in 20-25% at 10 years; ectopic ACTH depends on primary tumor.
📌
Screen for persistent complications: diabetes, hypertension, osteoporosis may require ongoing management.
📌
Psychosocial support: depression may worsen initially after cure due to steroid withdrawal.
📌
Board concept: Normal morning cortisol doesn't guarantee adequate stress response — may need stimulation testing.

Board Question Stem Patterns
📣
Purple striae + proximal weakness + hyperglycemia → confirm with 24-hour UFC or overnight dexamethasone.
📣
High cortisol + low ACTH → adrenal imaging for adenoma vs carcinoma.
📣
High cortisol + high ACTH + suppression with high-dose dex → pituitary MRI for Cushing disease.
📣
High cortisol + very high ACTH + no dex suppression → chest CT for ectopic source.
📣
Severe hypokalemia + metabolic alkalosis + Cushing features → ectopic ACTH syndrome.
📣
Young woman + Cushing + virilization → adrenal carcinoma.
📣
Hyperpigmentation after bilateral adrenalectomy → Nelson syndrome.
📣
Incidental adrenal mass + mild autonomous cortisol → subclinical Cushing syndrome.

One-Line Recap
🔸
Cushing syndrome diagnosis hinges on first confirming hypercortisolism (24-hour UFC, dexamethasone suppression, or salivary cortisol), then using ACTH levels to branch between ACTH-dependent causes (high ACTH: pituitary vs ectopic) and ACTH-independent causes (low ACTH: adrenal adenoma vs carcinoma), with high-dose dexamethasone suppression and IPSS further localizing ACTH-dependent sources.

bottom of page

