top of page
Reproductive & Endocrine Systems
Cortisol synthesis and regulation (HPA axis)
Core Principle of HPA Axis and Cortisol Regulation
🧷
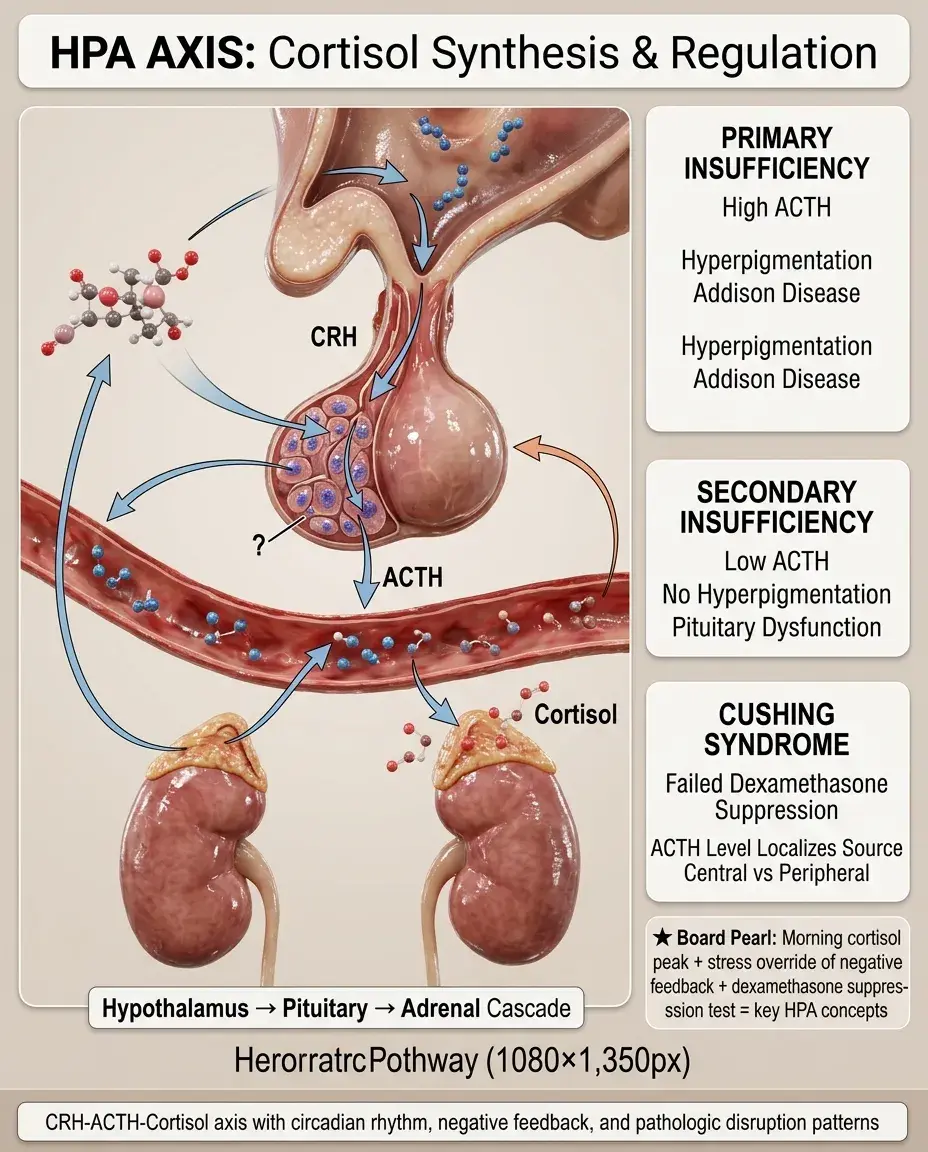
The hypothalamic-pituitary-adrenal (HPA) axis is a three-tiered neuroendocrine system that controls cortisol synthesis in response to stress, circadian rhythms, and metabolic demands.
🧷
The hypothalamus releases corticotropin-releasing hormone (CRH) → anterior pituitary releases adrenocorticotropic hormone (ACTH) → adrenal cortex zona fasciculata synthesizes and releases cortisol.
🧷
Cortisol exerts negative feedback at both the pituitary (inhibiting ACTH) and hypothalamus (inhibiting CRH), creating a self-regulating loop.
🧷
This system maintains basal cortisol levels with a pronounced circadian rhythm and mounts stress responses through rapid activation of each tier.

CRH: The Master Regulator
📍
CRH is a 41-amino acid peptide synthesized in the paraventricular nucleus (PVN) of the hypothalamus.
📍
Released into the hypophyseal portal system in response to physical stress (hypoglycemia, pain, hemorrhage), psychological stress, inflammatory cytokines (IL-1, IL-6, TNF-α), and circadian input from the suprachiasmatic nucleus.
📍
CRH binds CRH receptors on corticotroph cells in the anterior pituitary, activating the cAMP-PKA pathway → increased POMC gene transcription.
📍
Board pearl: CRH release follows a circadian pattern with peak secretion in early morning (4-6 AM) and nadir at midnight, explaining why morning cortisol is highest.

ACTH Processing and Release
🔹
ACTH is cleaved from the precursor molecule pro-opiomelanocortin (POMC) along with β-endorphin and melanocyte-stimulating hormone (MSH).
🔹
POMC processing explains why patients with high ACTH (Addison's disease, ectopic ACTH) develop hyperpigmentation — MSH binds melanocortin receptors in skin.
🔹
ACTH has a very short half-life (~10 minutes), allowing rapid on-off control of adrenal stimulation.
🔹
ACTH binds melanocortin 2 receptor (MC2R) on adrenal cortex cells → cAMP → PKA → activation of steroidogenic acute regulatory protein (StAR).
🔹
Board pearl: Primary adrenal insufficiency → high ACTH → hyperpigmentation. Secondary adrenal insufficiency → low ACTH → no hyperpigmentation.

Cortisol Biosynthesis Pathway
⭐
Cholesterol → pregnenolone (via cholesterol side-chain cleavage enzyme/CYP11A1) → 17α-hydroxypregnenolone → 17α-hydroxyprogesterone → 11-deoxycortisol → cortisol.
⭐
The rate-limiting step is cholesterol delivery to the inner mitochondrial membrane by StAR protein — this is where ACTH acts.
⭐
21-hydroxylase (CYP21A2) converts 17α-hydroxyprogesterone → 11-deoxycortisol. Deficiency causes 95% of congenital adrenal hyperplasia.
⭐
11β-hydroxylase (CYP11B1) performs the final conversion of 11-deoxycortisol → cortisol.
⭐
Board pearl: Each enzyme deficiency has a characteristic pattern — know 17α-hydroxyprogesterone accumulation = 21-hydroxylase deficiency.

Negative Feedback Mechanisms
✅
Cortisol binds glucocorticoid receptors (GR) in the hypothalamus and pituitary, directly suppressing CRH and ACTH gene transcription.
✅
Fast feedback (minutes): cortisol inhibits ACTH release from pre-formed vesicles.
✅
Intermediate feedback (hours): cortisol inhibits ACTH synthesis.
✅
Slow feedback (days): cortisol causes corticotroph cell atrophy with chronic suppression.
✅
Board distinction: Dexamethasone suppresses normal HPA axis but not autonomous sources (pituitary adenoma in low-dose test, ectopic ACTH in high-dose test).

Circadian Rhythm and Pulsatile Secretion
🧠
Cortisol secretion follows a robust circadian pattern: peak at 6-8 AM (15-20 μg/dL), nadir at midnight (< 5 μg/dL).
🧠
This rhythm is driven by the suprachiasmatic nucleus → PVN connection and is entrained by light-dark cycles.
🧠
Cortisol is released in 15-20 pulses per day, with amplitude highest in early morning.
🧠
Loss of circadian rhythm (Cushing's syndrome) or phase shift (night shift work) has diagnostic and clinical significance.
🧠
Board pearl: Random cortisol levels are worthless — always specify timing. Morning cortisol < 3 μg/dL suggests adrenal insufficiency; midnight cortisol > 7.5 μg/dL suggests Cushing's.

Stress Response Activation
⚡
Acute stress overrides negative feedback, causing massive CRH/ACTH/cortisol release within minutes.
⚡
Physical stressors activate the HPA axis via brainstem pathways → PVN. Examples: hypoglycemia (most potent), surgery, trauma, infection, hypoxia.
⚡
Psychological stress activates via limbic system (amygdala, hippocampus) → PVN.
⚡
Inflammatory cytokines (IL-1, IL-6, TNF-α) directly stimulate all three levels of the HPA axis.
⚡
Board clue: Post-surgical patient with refractory hypotension despite fluids/pressors → consider relative adrenal insufficiency from suppressed HPA axis.

Cortisol Transport and Metabolism
📌
90-95% of circulating cortisol is bound: 80% to cortisol-binding globulin (CBG/transcortin), 10-15% to albumin.
📌
Only free cortisol (5-10%) is biologically active and able to cross cell membranes.
📌
CBG increases with estrogen (pregnancy, OCPs) → total cortisol rises but free cortisol remains normal.
📌
Cortisol is metabolized in liver by 11β-hydroxysteroid dehydrogenase type 2 (11β-HSD2) → inactive cortisone.
📌
Board pearl: Pregnancy and OCPs increase total cortisol — always measure 24-hour urine free cortisol or midnight salivary cortisol to assess true cortisol status.

Tissue-Specific Cortisol Regulation
📣
11β-HSD exists in two isoforms that interconvert cortisol ↔ cortisone at tissue level.
📣
11β-HSD1 (liver, adipose, brain): converts inactive cortisone → active cortisol. Amplifies local glucocorticoid action.
📣
11β-HSD2 (kidney, colon, placenta): converts active cortisol → inactive cortisone. Protects mineralocorticoid receptor from cortisol.
📣
In kidney, 11β-HSD2 prevents cortisol from activating mineralocorticoid receptors. Deficiency (licorice, apparent mineralocorticoid excess) → hypertension, hypokalemia.
📣
Board pearl: Licorice inhibits 11β-HSD2 → cortisol acts as mineralocorticoid → HTN + hypokalemia.

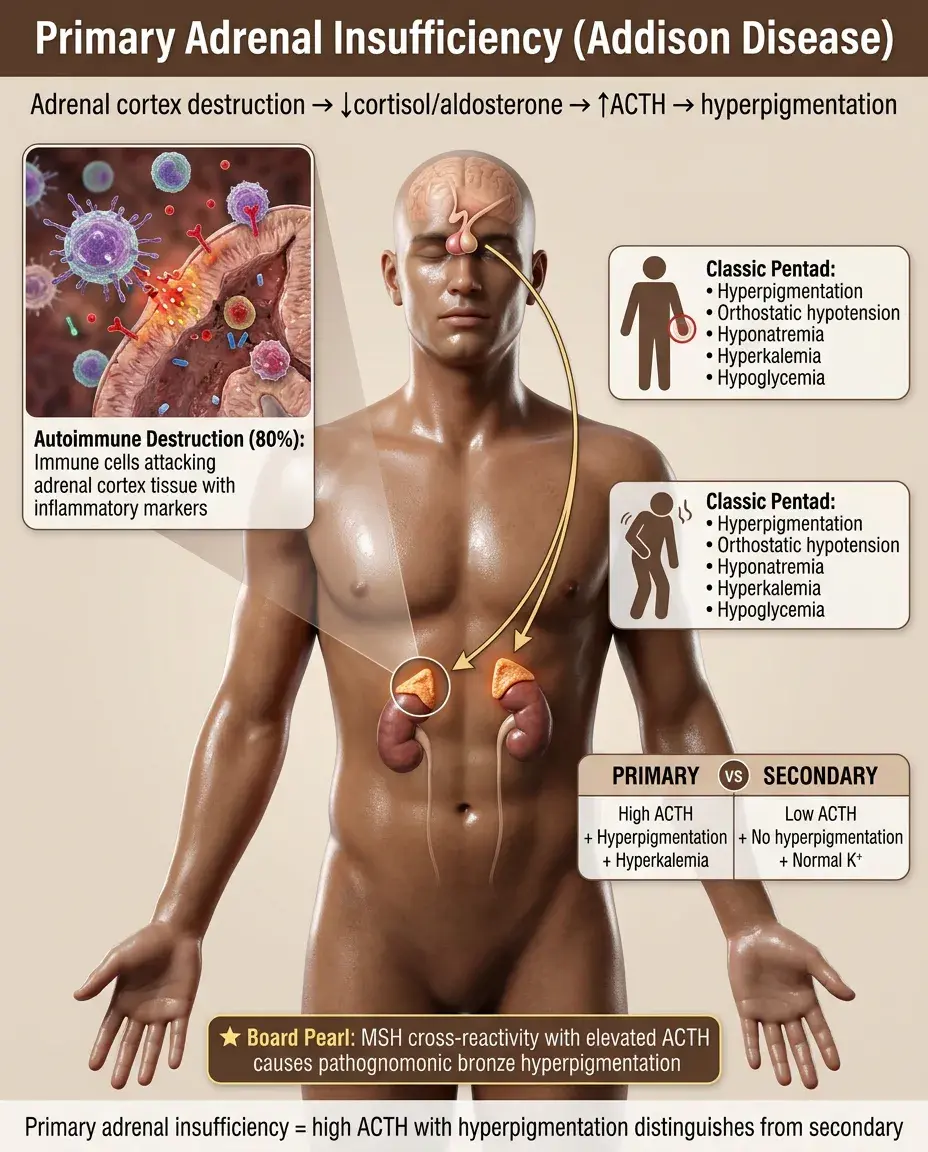
Primary Adrenal Insufficiency (Addison Disease)
🔸
Destruction of adrenal cortex → deficiency of cortisol, aldosterone, and adrenal androgens.
🔸
Causes: autoimmune (80% in developed countries), TB (most common worldwide), hemorrhage (Waterhouse-Friderichsen), metastases, HIV.
🔸
Loss of cortisol negative feedback → markedly elevated ACTH → hyperpigmentation (MSH cross-reactivity).
🔸
Classic presentation: hyperpigmentation + orthostatic hypotension + hyponatremia + hyperkalemia + hypoglycemia.
🔸
Board distinction: Primary = high ACTH + hyperpigmentation + hyperkalemia. Secondary = low ACTH + no hyperpigmentation + normal potassium.
Secondary and Tertiary Adrenal Insufficiency
🧷
Secondary: pituitary disease → ACTH deficiency. Causes: pituitary adenoma, surgery, radiation, apoplexy, infiltration.
🧷
Tertiary: hypothalamic disease or sudden withdrawal of chronic glucocorticoids → CRH deficiency.
🧷
No hyperpigmentation (low ACTH), no hyperkalemia (intact aldosterone from renin-angiotensin system).
🧷
Most common cause is iatrogenic from chronic steroid use → HPA suppression → inability to mount stress response.
🧷
Board pearl: Any patient on ≥ 20 mg prednisone daily for ≥ 3 weeks has HPA suppression and needs stress-dose steroids for surgery.

Cushing Syndrome: Classification
📍
Cushing syndrome = clinical state of cortisol excess from any cause.
📍
ACTH-dependent (80%): pituitary adenoma (Cushing disease, 70%), ectopic ACTH (10%).
📍
ACTH-independent (20%): adrenal adenoma, carcinoma, bilateral hyperplasia, exogenous steroids.
📍
Pseudo-Cushing: depression, alcoholism, severe illness → functional HPA activation without true autonomous cortisol production.
📍
Board approach: First confirm hypercortisolism (24-hour urine, midnight salivary cortisol), then measure ACTH to determine if ACTH-dependent or independent.

Diagnostic Testing: Confirming Hypercortisolism
🔹
Three first-line tests, need at least two abnormal: (1) 24-hour urine free cortisol, (2) midnight salivary cortisol, (3) 1-mg overnight dexamethasone suppression test.
🔹
24-hour urine free cortisol > 3× upper normal limit is diagnostic; 1-3× requires repeat testing.
🔹
Midnight salivary cortisol > upper normal limit on two occasions (tests loss of circadian rhythm).
🔹
1-mg dexamethasone at 11 PM → 8 AM cortisol should be < 1.8 μg/dL. Failure to suppress suggests Cushing syndrome.
🔹
Board pearl: Random serum cortisol is useless for diagnosing Cushing syndrome — too much overlap with normal stress response.

Localizing the Source: ACTH and Dexamethasone
⭐
After confirming hypercortisolism, measure ACTH: < 10 pg/mL = ACTH-independent, > 20 pg/mL = ACTH-dependent.
⭐
High-dose (8 mg) dexamethasone suppression: pituitary adenomas partially suppress (> 50% reduction), ectopic ACTH does not suppress.
⭐
CRH stimulation: pituitary adenomas show ACTH rise > 35%, ectopic ACTH shows no response.
⭐
Inferior petrosal sinus sampling (IPSS): central-to-peripheral ACTH ratio > 2 (basal) or > 3 (post-CRH) confirms pituitary source.
⭐
Board pearl: Small cell lung cancer and bronchial carcinoids are classic sources of ectopic ACTH with severe hypokalemia from cortisol's mineralocorticoid effect.

Congenital Adrenal Hyperplasia Variants
✅
All forms have impaired cortisol synthesis → loss of negative feedback → elevated ACTH → bilateral adrenal hyperplasia.
✅
21-hydroxylase deficiency (95%): accumulation of 17α-hydroxyprogesterone → shunted to androgens → virilization of females, precocious puberty in males. Salt-wasting form also lacks aldosterone.
✅
11β-hydroxylase deficiency (5%): accumulation of 11-deoxycorticosterone (DOC, mineralocorticoid activity) → hypertension + virilization.
✅
17α-hydroxylase deficiency: cannot make cortisol or sex hormones → sexual infantilism + hypertension (DOC accumulation).
✅
Board pearl: Ambiguous genitalia in newborn girl + salt wasting = 21-hydroxylase deficiency until proven otherwise.

Dynamic Testing for Adrenal Insufficiency
🧠
ACTH stimulation (cosyntropin) test: synthetic ACTH 250 μg IV → cortisol at 30 and 60 minutes should rise > 18-20 μg/dL.
🧠
Abnormal in primary adrenal insufficiency (adrenal cannot respond) and chronic secondary insufficiency (atrophic adrenals).
🧠
May be normal in acute secondary insufficiency (adrenals not yet atrophied).
🧠
Low-dose (1 μg) ACTH test: more sensitive for partial adrenal insufficiency.
🧠
Board pearl: Morning cortisol < 3 μg/dL or > 15 μg/dL is diagnostic and doesn't require stimulation testing. Intermediate values need ACTH stimulation.

Special Situations: Critical Illness and Pregnancy
⚡
Critical illness: CBG falls → total cortisol decreases but free cortisol may be adequate. "Relative adrenal insufficiency" is controversial.
⚡
Random cortisol < 10 μg/dL in severe sepsis suggests adrenal insufficiency; > 34 μg/dL suggests adequate response.
⚡
Pregnancy: CBG rises 2-3 fold → total cortisol increases but free cortisol remains normal until third trimester.
⚡
Placental CRH production in late pregnancy → further HPA activation → preparation for delivery stress.
⚡
Board pearl: In pregnancy, use trimester-specific reference ranges or measure 24-hour urine free cortisol, not total serum cortisol.

Clinical Patterns and Complications
📌
Acute adrenal crisis: profound hypotension, hyponatremia, hyperkalemia, hypoglycemia, abdominal pain. Triggered by stress in setting of adrenal insufficiency.
📌
Cushing syndrome: central obesity, moon facies, buffalo hump, purple striae, proximal myopathy, osteoporosis, glucose intolerance, hypertension.
📌
Nelson syndrome: post-bilateral adrenalectomy for Cushing disease → loss of cortisol feedback → pituitary adenoma enlargement → mass effect + hyperpigmentation.
📌
Waterhouse-Friderichsen: bilateral adrenal hemorrhage from meningococcemia → acute primary adrenal insufficiency.
📌
Board clue: Young woman with vitiligo and primary hypothyroidism develops fatigue and hyperpigmentation → autoimmune polyglandular syndrome with Addison disease.

Board Question Stem Patterns
📣
Hyperpigmentation + orthostatic hypotension + hyperkalemia → primary adrenal insufficiency.
📣
Central obesity + purple striae + proximal weakness + osteoporosis → Cushing syndrome, check 24-hour urine cortisol.
📣
Failure to suppress with low-dose but suppression with high-dose dexamethasone → pituitary Cushing disease.
📣
Severe hypokalemia + HTN + high cortisol + high ACTH + no suppression → ectopic ACTH (small cell lung cancer).
📣
Ambiguous genitalia + salt wasting in neonate → 21-hydroxylase deficiency.
📣
Post-op hypotension in patient on chronic prednisone → acute adrenal insufficiency from HPA suppression.
📣
Morning cortisol 8 μg/dL in fatigued patient → perform ACTH stimulation test.

One-Line Recap
🔸
The HPA axis (CRH → ACTH → cortisol) maintains homeostasis through negative feedback, follows circadian rhythm with morning peak, responds to stress by overriding suppression, and when disrupted causes primary insufficiency (high ACTH, hyperpigmentation), secondary insufficiency (low ACTH, no hyperpigmentation), or Cushing syndrome (confirmed by failed dexamethasone suppression, localized by ACTH level).
bottom of page

