top of page
Cardiovascular System
Cardiac muscle ultrastructure (intercalated discs, gap junctions)
Core Principle of Cardiac Muscle Ultrastructure
🧷
Cardiac muscle cells (cardiomyocytes) are striated like skeletal muscle but uniquely interconnected through specialized cell-cell junctions called intercalated discs.
🧷
These intercalated discs contain three critical components: gap junctions for electrical coupling, adherens junctions for mechanical strength, and desmosomes for additional mechanical adhesion.
🧷
This structural arrangement allows the heart to function as a functional syncytium — individual cells contract as a coordinated unit despite being separate cellular entities.
🧷
The ultrastructural organization ensures rapid electrical propagation and synchronized contraction essential for effective cardiac pumping.

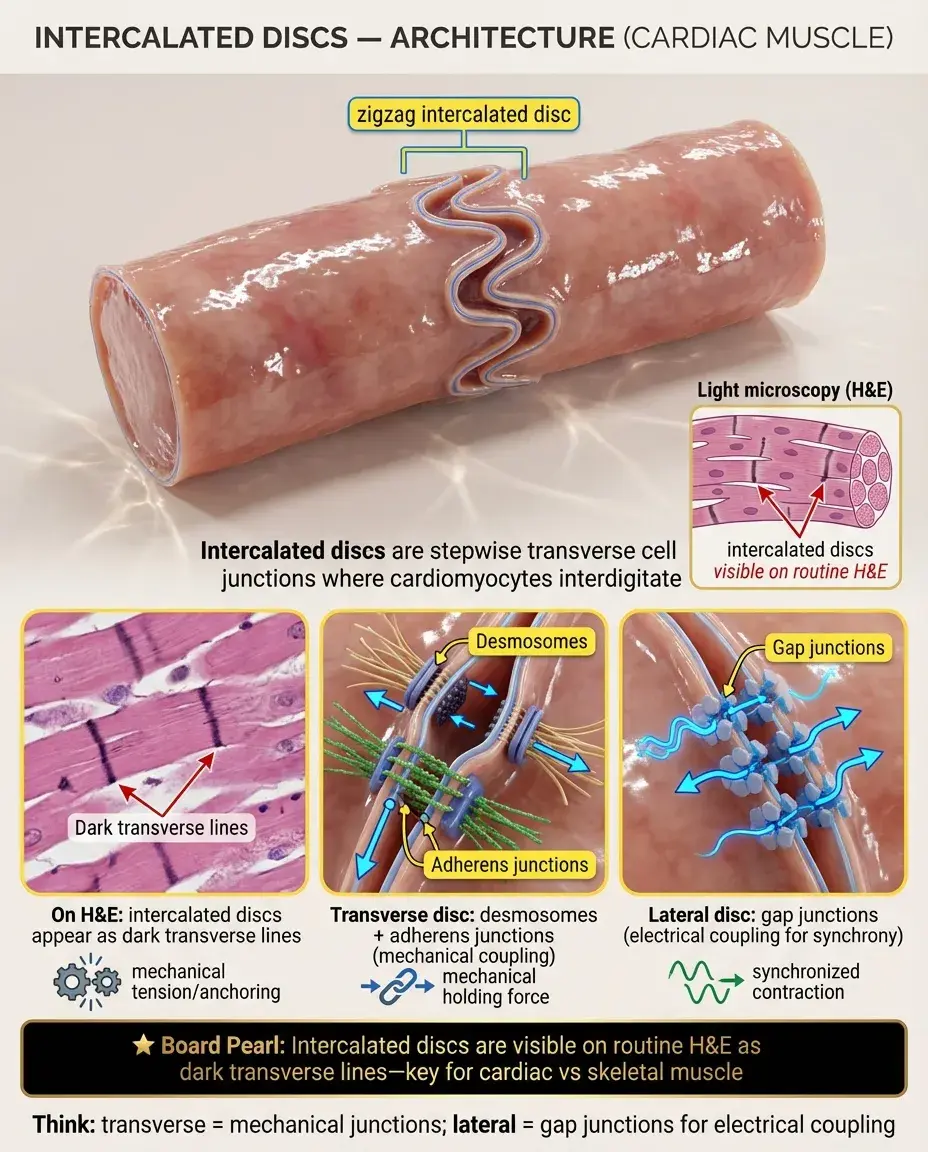
Intercalated Disc Architecture
📍
Intercalated discs appear as dark, stepwise structures at light microscopy, running transversely between adjacent cardiomyocytes.
📍
They have two distinct regions: transverse portions (perpendicular to muscle fiber axis) containing desmosomes and adherens junctions, and lateral portions (parallel to fiber axis) containing gap junctions.
📍
The disc appears as a zigzag or staircase pattern because it follows the irregular cell borders where cardiomyocytes interdigitate.
📍
Board pearl: Intercalated discs are visible with routine H&E staining as dark transverse lines — a key histological feature distinguishing cardiac from skeletal muscle.
Gap Junctions: The Electrical Coupling System
🔹
Gap junctions are clusters of intercellular channels that directly connect the cytoplasm of adjacent cardiomyocytes, allowing rapid electrical coupling.
🔹
Each gap junction channel is formed by two hemichannels (connexons), one from each cell, with each connexon composed of six connexin protein subunits.
🔹
The predominant cardiac connexin is connexin-43 (Cx43), though Cx40 and Cx45 are also present in specific regions.
🔹
These channels allow passage of ions (Na⁺, K⁺, Ca²⁺) and small molecules (<1 kDa) including cAMP and IP₃, enabling electrical and metabolic coupling.
🔹
Board pearl: Gap junctions create low-resistance pathways that allow action potentials to spread rapidly from cell to cell.

Connexin Distribution and Conduction Velocity
⭐
Connexin-43 is the most abundant connexin in ventricular and atrial working myocardium, providing rapid conduction through contractile tissue.
⭐
Connexin-40 is primarily found in atrial muscle and the rapid conduction system (His-Purkinje fibers), contributing to very fast conduction velocities.
⭐
Connexin-45 is present in the SA and AV nodes, where its lower conductance contributes to slower conduction — a protective mechanism preventing excessive heart rates.
⭐
The density and type of connexins directly correlate with conduction velocity: Purkinje fibers (highest Cx40/Cx43 density) → atrial/ventricular muscle → AV node (lowest density, mainly Cx45).

Desmosomes: The Mechanical Anchors
✅
Desmosomes (maculae adherentes) are button-like structures that mechanically couple adjacent cardiomyocytes, preventing separation during forceful contraction.
✅
They consist of transmembrane cadherins (desmoglein and desmocollin) that bind to identical proteins on the adjacent cell, linked intracellularly to intermediate filaments via desmoplakin.
✅
In cardiac muscle, the intermediate filaments are primarily desmin, which connects desmosomes to the contractile apparatus and maintains cellular architecture.
✅
Board pearl: Mutations in desmosomal proteins (especially plakophilin-2 and desmoplakin) cause arrhythmogenic right ventricular cardiomyopathy (ARVC).

Adherens Junctions: The Force Transmitters
🧠
Adherens junctions (fasciae adherentes) form continuous belt-like structures that transmit contractile force between cardiomyocytes.
🧠
They contain N-cadherin molecules that bind homophilically to N-cadherin on the adjacent cell, anchored intracellularly to actin filaments via α-actinin and vinculin.
🧠
These junctions are the primary sites where the contractile apparatus of one cell connects to the next, allowing force transmission throughout the myocardium.
🧠
Unlike in epithelial cells where adherens junctions form complete belts, in cardiac muscle they appear as discrete segments within the intercalated disc.

T-tubules and Excitation-Contraction Coupling
⚡
T-tubules (transverse tubules) are deep invaginations of the sarcolemma that penetrate into the cell interior at each Z-line.
⚡
They bring the action potential deep into the cell, ensuring simultaneous activation of all myofibrils.
⚡
T-tubules in cardiac muscle are wider (200-400 nm) and less regularly organized than in skeletal muscle, often forming longitudinal extensions.
⚡
The T-tubule membrane contains L-type Ca²⁺ channels (dihydropyridine receptors) that face ryanodine receptors on the sarcoplasmic reticulum across a 12-20 nm gap.
⚡
Board pearl: Unlike skeletal muscle, cardiac muscle requires extracellular Ca²⁺ influx through L-type channels to trigger SR Ca²⁺ release.

Sarcoplasmic Reticulum Organization
📌
The cardiac SR is less extensive than in skeletal muscle, occupying only 2-3% of cell volume (vs. 10% in skeletal muscle).
📌
It forms a network of tubules surrounding myofibrils with specialized junctional SR (jSR) regions that appose T-tubules, forming dyads.
📌
The jSR contains high concentrations of ryanodine receptors (RyR2) that release Ca²⁺ in response to Ca²⁺ influx through L-type channels — calcium-induced calcium release (CICR).
📌
Network SR contains SERCA2a pumps that actively sequester Ca²⁺ back into the SR, regulated by phospholamban.
📌
Board distinction: Cardiac muscle forms dyads (1 T-tubule + 1 SR cistern), while skeletal muscle forms triads (1 T-tubule + 2 SR cisternae).

Mitochondrial Abundance and Metabolism
📣
Cardiac muscle contains the highest mitochondrial density of any tissue — mitochondria occupy 30-35% of cell volume, reflecting enormous ATP demands.
📣
Mitochondria are arranged in rows between myofibrils and clustered beneath the sarcolemma, positioned to efficiently deliver ATP to contractile proteins and ion pumps.
📣
They preferentially utilize fatty acid oxidation (60-70% of ATP production) but maintain metabolic flexibility to use glucose, lactate, and ketones.
📣
Interfibrillar mitochondria directly supply ATP to myofibrils, while subsarcolemmal mitochondria support ion pumps and signaling.
📣
Board pearl: The heart consumes more oxygen per gram than any other organ — coronary blood flow must increase 4-5 fold during exercise.

Sarcomere Structure in Cardiac Muscle
🔸
Cardiac sarcomeres have the same basic organization as skeletal muscle: Z-lines, I-bands, A-bands, H-zones, and M-lines.
🔸
Thin filaments contain cardiac-specific isoforms of troponin (cTnI, cTnT, cTnC) and tropomyosin that regulate Ca²⁺-dependent contraction.
🔸
Thick filaments contain cardiac β-myosin heavy chain (slower ATPase than skeletal muscle) allowing sustained contraction.
🔸
Titin extends from Z-line to M-line, providing passive elasticity and maintaining sarcomere alignment during diastole.
🔸
Board pearl: Cardiac troponins (cTnI and cTnT) are specific biomarkers for myocardial injury because these isoforms are unique to heart muscle.

Pathology of Gap Junction Dysfunction
🧷
Reduced connexin-43 expression or lateralization (redistribution from intercalated discs to lateral membranes) occurs in heart failure and ischemia.
🧷
This gap junction remodeling creates heterogeneous conduction, slow conduction zones, and substrate for reentrant arrhythmias.
🧷
Mutations in GJA1 (encoding Cx43) cause oculodentodigital dysplasia with cardiac conduction abnormalities.
🧷
Acute ischemia causes rapid closure of gap junctions through acidosis and elevated intracellular Ca²⁺, electrically isolating damaged cells.
🧷
Board pearl: Gap junction uncoupling during ischemia prevents spread of injury but also creates conduction block that can trigger arrhythmias.

Intercalated Disc Proteins and Cardiomyopathies
📍
Mutations in desmosomal proteins cause arrhythmogenic right ventricular cardiomyopathy (ARVC): plakophilin-2 (most common), desmoplakin, desmoglein-2, desmocollin-2.
📍
ARVC presents with ventricular arrhythmias, sudden death in young athletes, and fibrofatty replacement of myocardium, particularly in the RV.
📍
Mutations in adherens junction proteins (N-cadherin, α-catenin) can cause dilated cardiomyopathy with conduction system disease.
📍
Loss of intercalated disc integrity leads to mechanical uncoupling, myocyte death, and replacement fibrosis.
📍
Board pearl: Epsilon waves on ECG and fibrofatty infiltration on MRI are classic findings in ARVC.

Developmental Assembly of Intercalated Discs
🔹
During fetal development, gap junctions initially distribute uniformly around cardiomyocytes before clustering at cell termini.
🔹
Adherens junctions and desmosomes assemble first, providing mechanical stability before electrical coupling matures.
🔹
Connexin expression switches during development: Cx45 predominates in early embryonic heart, then Cx43 and Cx40 increase postnatally.
🔹
T-tubule formation occurs postnatally in mammals, coinciding with the switch from hyperplastic to hypertrophic cardiac growth.
🔹
Board clue: Premature infants have immature T-tubule systems, contributing to their reduced cardiac reserve.

Calcium Handling Microdomains
⭐
Dyadic clefts (12-20 nm spaces between T-tubules and jSR) create Ca²⁺ microdomains where local [Ca²⁺] can reach 10-100 μM during excitation-contraction coupling.
⭐
Calsequestrin within the SR buffers Ca²⁺ and regulates RyR2 opening, preventing spontaneous Ca²⁺ release.
⭐
Na⁺-Ca²⁺ exchanger (NCX) and plasma membrane Ca²⁺-ATPase cluster near T-tubules to extrude Ca²⁺ during relaxation.
⭐
Mitochondria take up Ca²⁺ during systole via the mitochondrial Ca²⁺ uniporter, linking excitation-contraction coupling to ATP production.
⭐
Board pearl: Digitalis inhibits Na⁺-K⁺-ATPase → increased intracellular Na⁺ → reduced NCX activity → increased intracellular Ca²⁺ → positive inotropy.

Intercalated Disc Remodeling in Disease
✅
Heart failure shows lateralization of gap junctions from intercalated discs to lateral cell borders, disrupting anisotropic conduction.
✅
Hypertrophy increases intercalated disc convolution and length, initially maintaining cell-cell coupling despite increased cell size.
✅
Atrial fibrillation associates with reduced Cx40 expression and heterogeneous gap junction distribution in atria.
✅
Myocardial infarction border zones show reduced connexin-43 and disorganized intercalated disc structure, creating arrhythmogenic substrate.
✅
Board pearl: Angiotensin II and mechanical stretch promote gap junction remodeling, linking neurohormonal activation to arrhythmogenesis.

Unique Features of Atrial Myocyte Structure
🧠
Atrial myocytes are smaller and more spindle-shaped than ventricular myocytes, with less developed T-tubule systems.
🧠
They contain atrial-specific granules storing atrial natriuretic peptide (ANP) and brain natriuretic peptide (BNP), released in response to stretch.
🧠
Atrial intercalated discs have higher Cx40 content, enabling rapid atrial conduction for synchronized atrial contraction.
🧠
Sparse T-tubules mean atrial excitation-contraction coupling relies more on subsarcolemmal Ca²⁺ entry and propagated Ca²⁺ waves.
🧠
Board pearl: ANP and BNP release from stretched atrial myocytes promotes natriuresis, vasodilation, and inhibition of renin-angiotensin system.

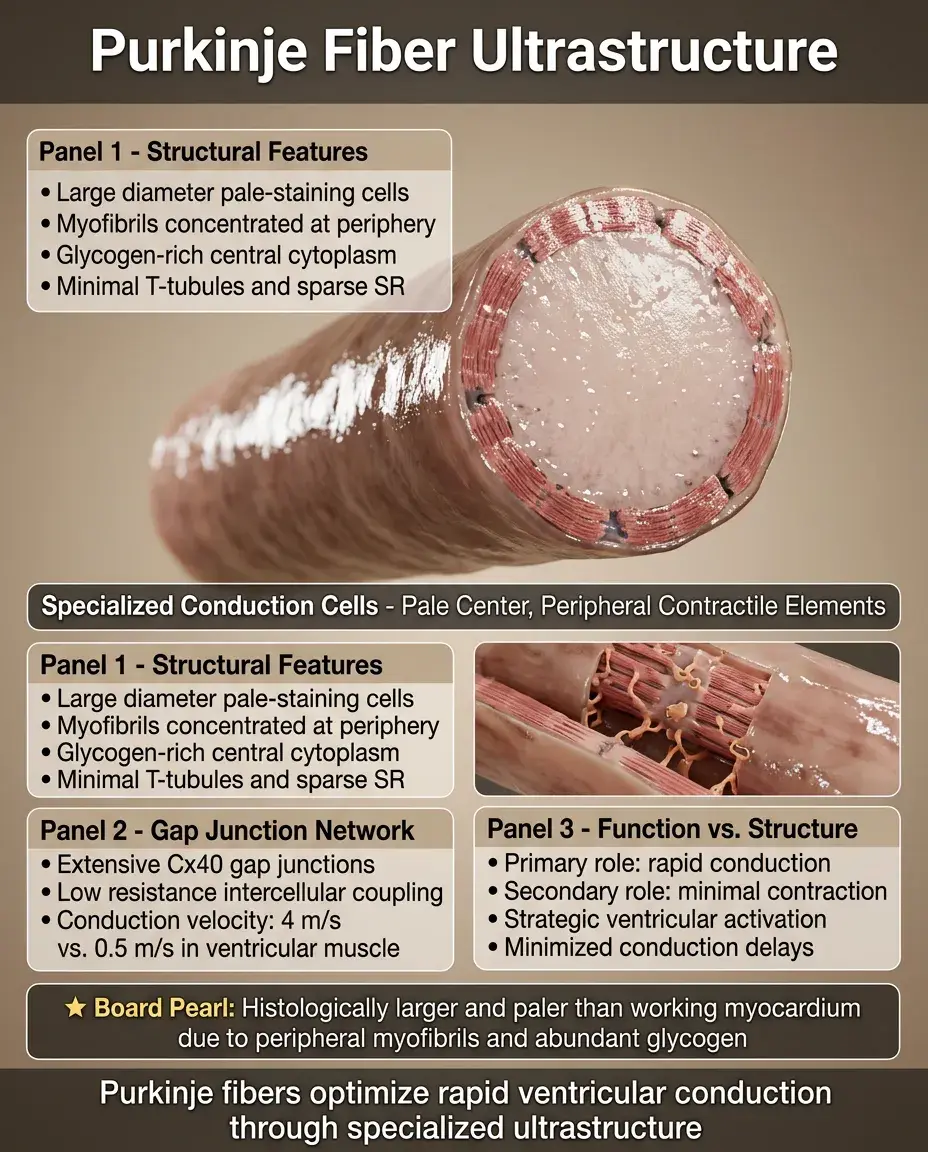
Purkinje Fiber Ultrastructure
⚡
Purkinje fibers are specialized conducting cells with fewer myofibrils concentrated at the cell periphery, leaving a pale-staining central area rich in glycogen.
⚡
They have extensive gap junction coupling with high Cx40 content, enabling conduction velocities up to 4 m/s (vs. 0.5 m/s in ventricular muscle).
⚡
Minimal T-tubule development and sparse SR reflect their primary conduction (not contractile) function.
⚡
Large diameter and low resistance intercellular connections minimize conduction delays in the ventricular activation sequence.
⚡
Board pearl: On histology, Purkinje fibers appear larger and paler than working myocardium due to peripheral myofibrils and abundant glycogen.
Clinical Correlations of Ultrastructural Defects
📌
Duchenne muscular dystrophy: dystrophin deficiency disrupts costamere linkage between sarcolemma and contractile apparatus → cardiomyopathy.
📌
Catecholaminergic polymorphic VT: RyR2 mutations cause spontaneous SR Ca²⁺ release during sympathetic stimulation → triggered arrhythmias.
📌
Timothy syndrome: L-type Ca²⁺ channel mutations prevent inactivation → prolonged QT, autism, syndactyly.
📌
Danon disease: LAMP2 deficiency → autophagic vacuoles in cardiomyocytes → hypertrophic cardiomyopathy, pre-excitation.
📌
Board distinction: Skeletal muscle symptoms often precede cardiac involvement in dystrophinopathies, while RyR2 mutations cause isolated cardiac phenotypes.

Board Question Stem Patterns
📣
Histology showing branched cells with central nuclei and transverse dark lines → cardiac muscle with intercalated discs.
📣
Young athlete with palpitations, family history of sudden death, epsilon waves → ARVC from desmosomal mutations.
📣
Decreased conduction velocity between cardiac cells in heart failure → gap junction remodeling and connexin downregulation.
📣
Positive inotropic effect of cardiac glycosides → inhibition of Na⁺-K⁺-ATPase → increased intracellular Ca²⁺.
📣
Pale-staining cells with peripheral myofibrils in the ventricular conduction system → Purkinje fibers.
📣
Exercise-induced ventricular tachycardia in a child → catecholaminergic polymorphic VT from RyR2 mutation.
📣
Loss of transverse striations in dilated cardiomyopathy → disruption of sarcomere and intercalated disc organization.

One-Line Recap
🔸
Cardiac muscle achieves synchronized contraction through intercalated discs containing gap junctions (connexin channels for electrical coupling), desmosomes (mechanical adhesion), and adherens junctions (force transmission), working with T-tubules and SR to enable calcium-induced calcium release, with mutations in disc proteins causing arrhythmogenic cardiomyopathy and gap junction remodeling contributing to heart failure arrhythmogenesis.

bottom of page

