top of page
Respiratory System
Bronchopulmonary dysplasia
Core Principle of Bronchopulmonary Dysplasia
🧷
Bronchopulmonary dysplasia (BPD) is chronic lung disease of prematurity resulting from arrested alveolar development and abnormal vascular remodeling following lung injury in the developing lung.
🧷
The pathophysiology centers on the interruption of normal lung development during the saccular and alveolar stages (24–36 weeks gestation), when premature infants require respiratory support.
🧷
Modern BPD ("new BPD") differs from classic BPD — it features simplified alveolar structures with fewer, larger alveoli rather than the fibrosis and emphysema seen historically.
🧷
Risk is inversely proportional to gestational age and birth weight, with highest incidence in infants <28 weeks or <1000g.

Pathophysiology: The Two-Hit Model
📍
First hit: prematurity interrupts normal lung development during critical alveolarization phase → lungs have fewer alveoli, thicker alveolar walls, and abnormal capillary development.
📍
Second hit: postnatal injury from mechanical ventilation, oxygen toxicity, inflammation, and infection → further disrupts alveolarization and promotes abnormal repair.
📍
Key molecular pathways: decreased VEGF signaling → impaired angiogenesis; elevated TGF-β → fibrosis; oxidative stress → cell death and arrested development.
📍
The result is a lung with reduced surface area for gas exchange, abnormal ventilation-perfusion matching, and increased work of breathing.
📍
Board pearl: BPD is not simply "scarred lungs" — it's fundamentally a disorder of lung development.

Risk Factors and Prevention Strategies
🔹
Major risk factors: extreme prematurity (<28 weeks), low birth weight (<1000g), male sex, chorioamnionitis, need for mechanical ventilation, high oxygen exposure.
🔹
Protective factors: antenatal corticosteroids (promote lung maturation), surfactant therapy (reduces ventilator needs), caffeine (reduces apnea and ventilator days).
🔹
Prevention strategies focus on minimizing lung injury: gentle ventilation strategies, permissive hypercapnia, early CPAP use, oxygen saturation targets of 90–95%.
🔹
Postnatal corticosteroids reduce BPD but increase neurodevelopmental risks — reserved for infants who cannot wean from ventilation.
🔹
Board pearl: Antenatal steroids reduce RDS severity but do not directly prevent BPD.

Clinical Definition and Severity Grading
⭐
BPD definition: oxygen requirement for ≥28 days in a premature infant, with severity assessed at 36 weeks postmenstrual age (or discharge if earlier).
⭐
Mild BPD: breathing room air at 36 weeks PMA.
⭐
Moderate BPD: requiring <30% FiO₂ at 36 weeks PMA.
⭐
Severe BPD: requiring ≥30% FiO₂ or positive pressure support at 36 weeks PMA.
⭐
This definition captures both early lung injury (28-day requirement) and persistent dysfunction (36-week assessment).
⭐
Board distinction: The 36-week PMA assessment determines severity, not the amount of support needed in the first month.

Histopathologic Features: Old vs New BPD
✅
Classic BPD (pre-surfactant era): heterogeneous lung injury with alternating areas of atelectasis and hyperinflation, severe fibrosis, squamous metaplasia, smooth muscle hyperplasia.
✅
New BPD (post-surfactant era): uniform arrest of alveolar development → fewer, larger alveoli; dysmorphic capillaries; mild fibrosis; less heterogeneity.
✅
Key finding: alveolar simplification — the hallmark of modern BPD. Alveoli are larger and fewer in number, reducing surface area for gas exchange.
✅
Pulmonary vasculature shows decreased branching and abnormal muscularization → pulmonary hypertension risk.
✅
Board pearl: Modern BPD shows alveolar simplification, not the severe fibrosis of classic BPD.

Clinical Presentation in the NICU
🧠
Early phase: persistent oxygen requirement beyond expected course of RDS, difficulty weaning ventilator support, episodes of desaturation.
🧠
Chest X-ray evolution: initial ground-glass appearance (RDS) → persistent haziness → eventually heterogeneous pattern with areas of hyperinflation and atelectasis.
🧠
Physical exam: tachypnea, retractions, crackles, wheezing in severe cases, poor growth despite adequate calories.
🧠
Laboratory findings: hypoxemia, hypercapnia (often tolerated), elevated BNP if pulmonary hypertension develops.
🧠
Board clue: An extremely premature infant who cannot wean from oxygen by 4 weeks of life likely has developing BPD.

Imaging Features and Evolution
⚡
Early (days-weeks): diffuse haziness, low lung volumes, air bronchograms — indistinguishable from RDS.
⚡
Intermediate (weeks): persistent diffuse haziness, emerging heterogeneity, subtle hyperinflation.
⚡
Established BPD (months): heterogeneous pattern with alternating areas of hyperinflation and atelectasis, prominent interstitial markings, possible cystic changes.
⚡
CT findings (rarely needed): mosaic attenuation pattern, architectural distortion, subpleural cysts.
⚡
Board pearl: The chest X-ray in BPD evolves from homogeneous (RDS-like) to heterogeneous (hyperinflation + atelectasis) over weeks to months.

Pulmonary Hypertension: Major Complication
📌
Occurs in 15–25% of infants with moderate-severe BPD due to abnormal pulmonary vascular development and remodeling.
📌
Pathophysiology: decreased vascular branching + abnormal muscularization of small arteries + chronic hypoxemia → elevated pulmonary vascular resistance.
📌
Clinical signs: loud P2, RV heave, hepatomegaly, poor growth, increased oxygen needs, feeding difficulties.
📌
Screening: echocardiogram at 36 weeks PMA for all infants with moderate-severe BPD.
📌
Treatment: optimize oxygenation (target SpO₂ >95%), diuretics if fluid overloaded, sildenafil or bosentan for confirmed PH.
📌
Board pearl: New or increasing oxygen requirement in established BPD → evaluate for pulmonary hypertension.

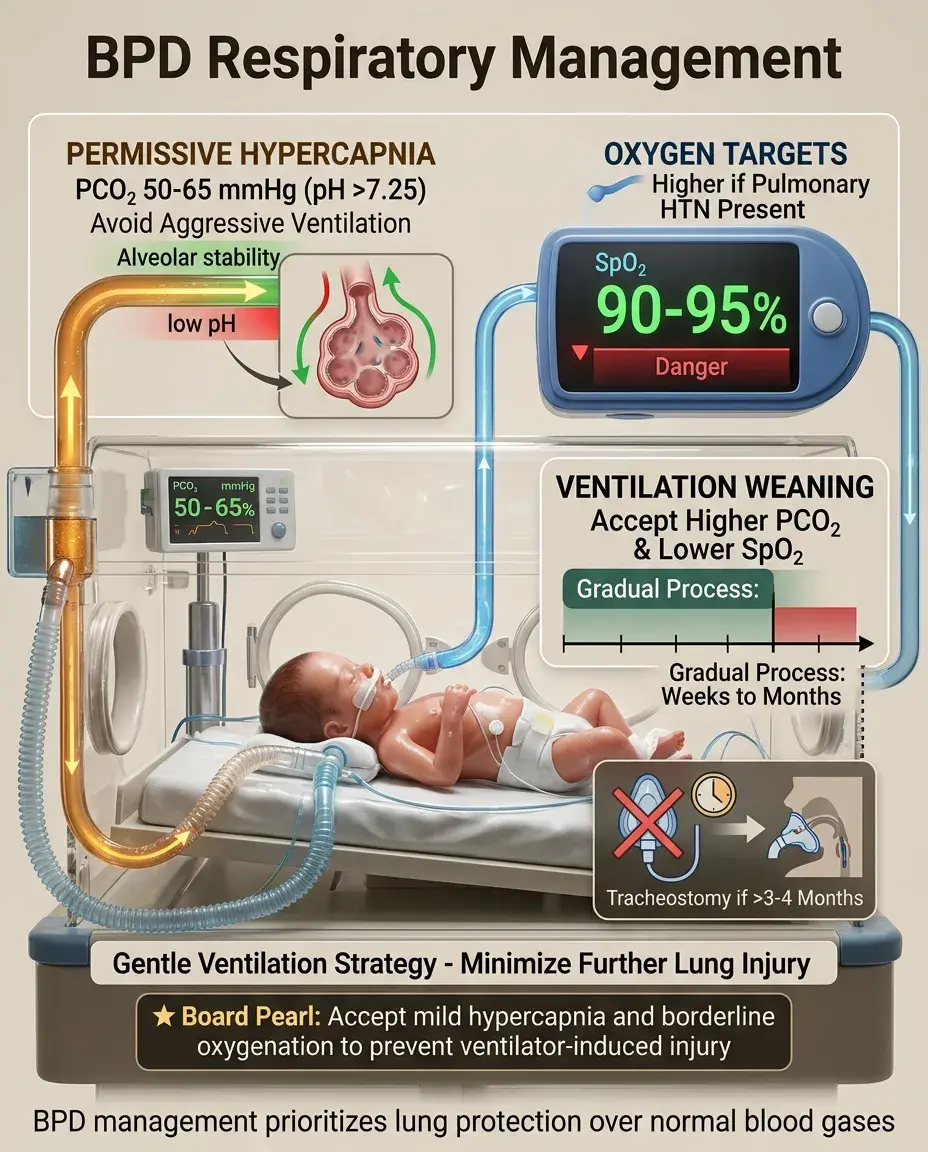
Respiratory Management Principles
📣
Goal: support gas exchange while minimizing further lung injury and allowing lung growth/repair.
📣
Permissive hypercapnia strategy: accept PCO₂ 50–65 mmHg (pH >7.25) to avoid aggressive ventilation.
📣
Oxygen targets: SpO₂ 90–95% (higher if pulmonary hypertension present).
📣
Ventilation weaning: gradual process over weeks to months; accept higher PCO₂ and lower SpO₂ to facilitate weaning.
📣
Tracheostomy considered for infants requiring ventilation beyond 3–4 months.
📣
Board pearl: In BPD, accepting mild hypercapnia and borderline oxygenation prevents ventilator-induced injury.
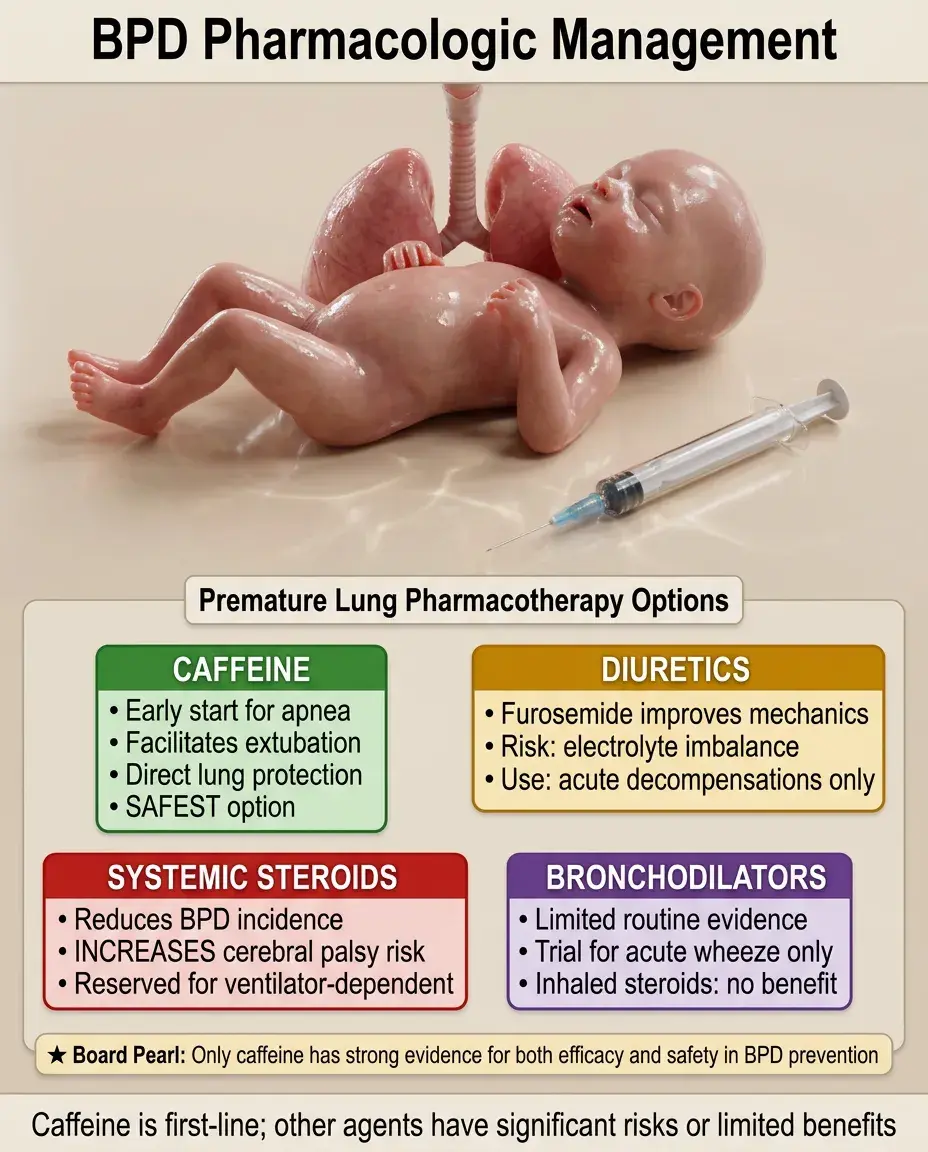
Pharmacologic Management
🔸
Caffeine: started early for apnea of prematurity, helps facilitate extubation, may have direct lung protective effects.
🔸
Diuretics: furosemide improves short-term lung mechanics but chronic use causes electrolyte imbalances and nephrocalcinosis — use intermittently for acute decompensations.
🔸
Bronchodilators: limited evidence for routine use; trial for acute wheezing episodes only.
🔸
Systemic corticosteroids: reduce BPD incidence but increase cerebral palsy risk — reserved for ventilator-dependent infants after careful risk-benefit discussion.
🔸
Inhaled corticosteroids: no proven benefit for BPD prevention or treatment.
🔸
Board pearl: Only caffeine has strong evidence for both efficacy and safety in BPD prevention.
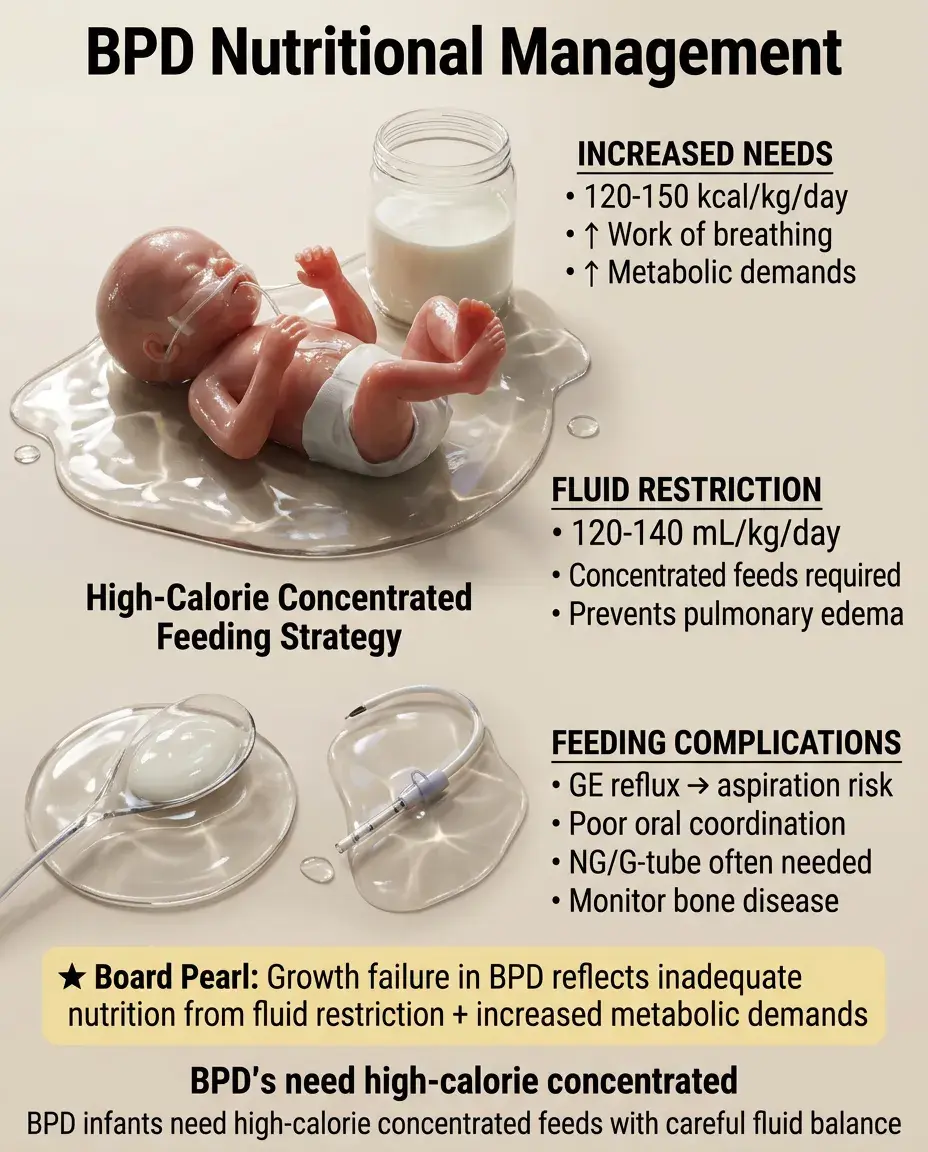
Nutritional Considerations
🧷
Increased caloric needs: 120–150 kcal/kg/day due to increased work of breathing and poor growth.
🧷
Fluid restriction often necessary (120–140 mL/kg/day) to manage pulmonary edema → requires concentrated feeds.
🧷
Gastroesophageal reflux common → increases aspiration risk and worsens lung disease.
🧷
Poor oral motor coordination → many infants require nasogastric or gastrostomy feeding.
🧷
Monitor for metabolic bone disease: premature infants have low bone mineral stores + diuretics increase calcium loss.
🧷
Board pearl: Growth failure in BPD often reflects inadequate nutrition due to fluid restriction and increased metabolic demands.
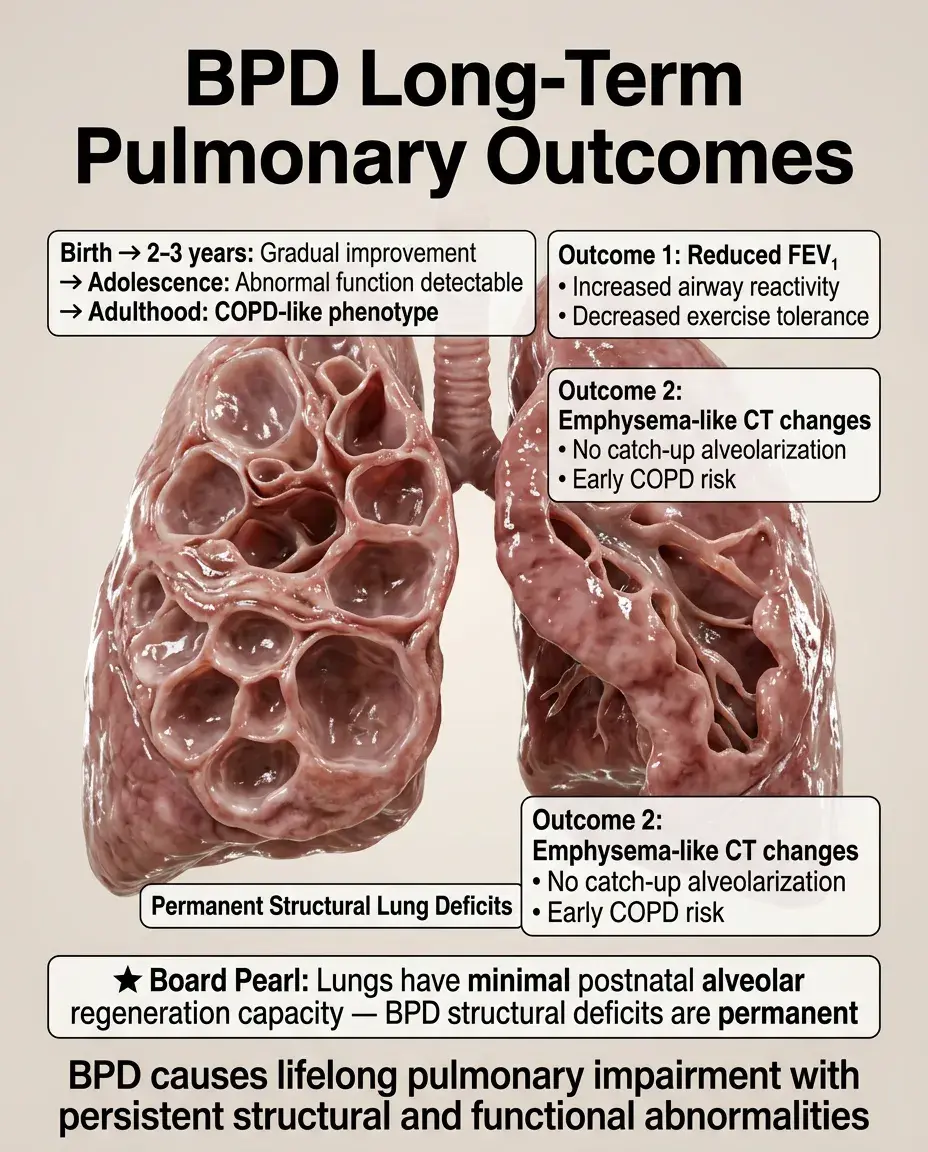
Long-Term Pulmonary Outcomes
📍
Lung function: reduced FEV₁, increased airway reactivity, decreased exercise tolerance persist into adulthood.
📍
Structural changes: emphysema-like changes on CT, reduced alveolar number persists (no catch-up alveolarization).
📍
Clinical trajectory: gradual improvement over first 2–3 years, but abnormal lung function detectable through adolescence.
📍
Adult outcomes: early COPD-like phenotype, increased risk of asthma, reduced peak lung function.
📍
Board pearl: Unlike other organs, the lung has minimal capacity for postnatal alveolar regeneration — BPD causes permanent structural deficits.
Neurodevelopmental Outcomes
🔹
BPD independently associated with worse neurodevelopmental outcomes even after controlling for prematurity.
🔹
Mechanisms: chronic hypoxemia, inflammation, nutritional deficits, prolonged hospitalization, medication effects.
🔹
Specific risks: cerebral palsy (especially with postnatal steroids), cognitive impairment, attention deficits, language delays.
🔹
Motor delays common due to prolonged ventilation, weakness, and limited mobility in NICU.
🔹
Early intervention services crucial: physical therapy, occupational therapy, speech therapy, developmental follow-up.
🔹
Board pearl: BPD is a multi-system disease — neurodevelopmental impairment is as important as respiratory morbidity.

Discharge Planning and Home Management
⭐
Discharge criteria: stable oxygen requirement, adequate growth, family education complete, medical home established.
⭐
Home oxygen: many infants require supplemental oxygen for months; wean based on sleep studies and growth.
⭐
Monitoring: home pulse oximetry for severe BPD, monthly weight checks, regular developmental screening.
⭐
Immunizations: follow standard schedule plus palivizumab (RSV prophylaxis) for first 2 RSV seasons.
⭐
Avoid smoke exposure, daycare in first year if possible, aggressive treatment of respiratory infections.
⭐
Board pearl: Palivizumab is indicated for all infants with BPD <2 years old who required treatment within 6 months of RSV season.

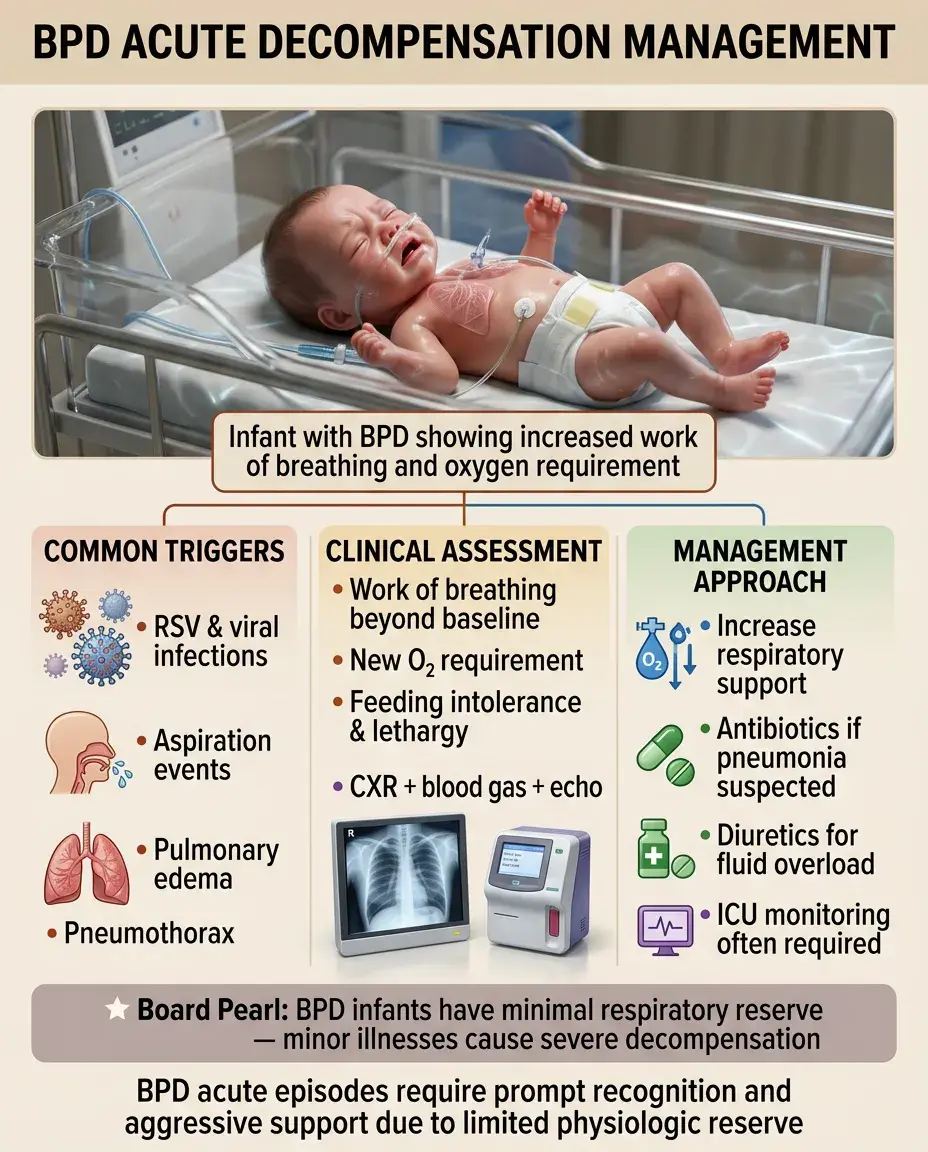
Acute Decompensation Management
✅
Common triggers: viral infections (especially RSV), aspiration, pulmonary edema, pneumothorax.
✅
Clinical signs: increased work of breathing beyond baseline, new oxygen requirement, feeding intolerance, lethargy.
✅
Initial evaluation: CXR (look for pneumonia, atelectasis, hyperinflation), blood gas, consider echo if suspect PH crisis.
✅
Management: increase respiratory support as needed, broad-spectrum antibiotics if pneumonia suspected, diuretics if fluid overloaded.
✅
Board pearl: BPD infants have little respiratory reserve — minor illnesses can cause severe decompensation requiring ICU care.
Differential Diagnosis Considerations
🧠
Congenital anomalies: pulmonary hypoplasia, congenital diaphragmatic hernia, cystic adenomatoid malformation.
🧠
Genetic conditions: surfactant protein deficiencies, ABCA3 mutations (present like severe BPD but don't improve).
🧠
Acquired conditions: pneumonia, aspiration, pulmonary hemorrhage.
🧠
Cardiac disease: PDA, congenital heart disease can worsen lung disease.
🧠
Key distinguishing feature: BPD requires prematurity + oxygen exposure for 28 days.
🧠
Board distinction: Term infants cannot have BPD by definition — consider genetic surfactant disorders in term infants with BPD-like disease.

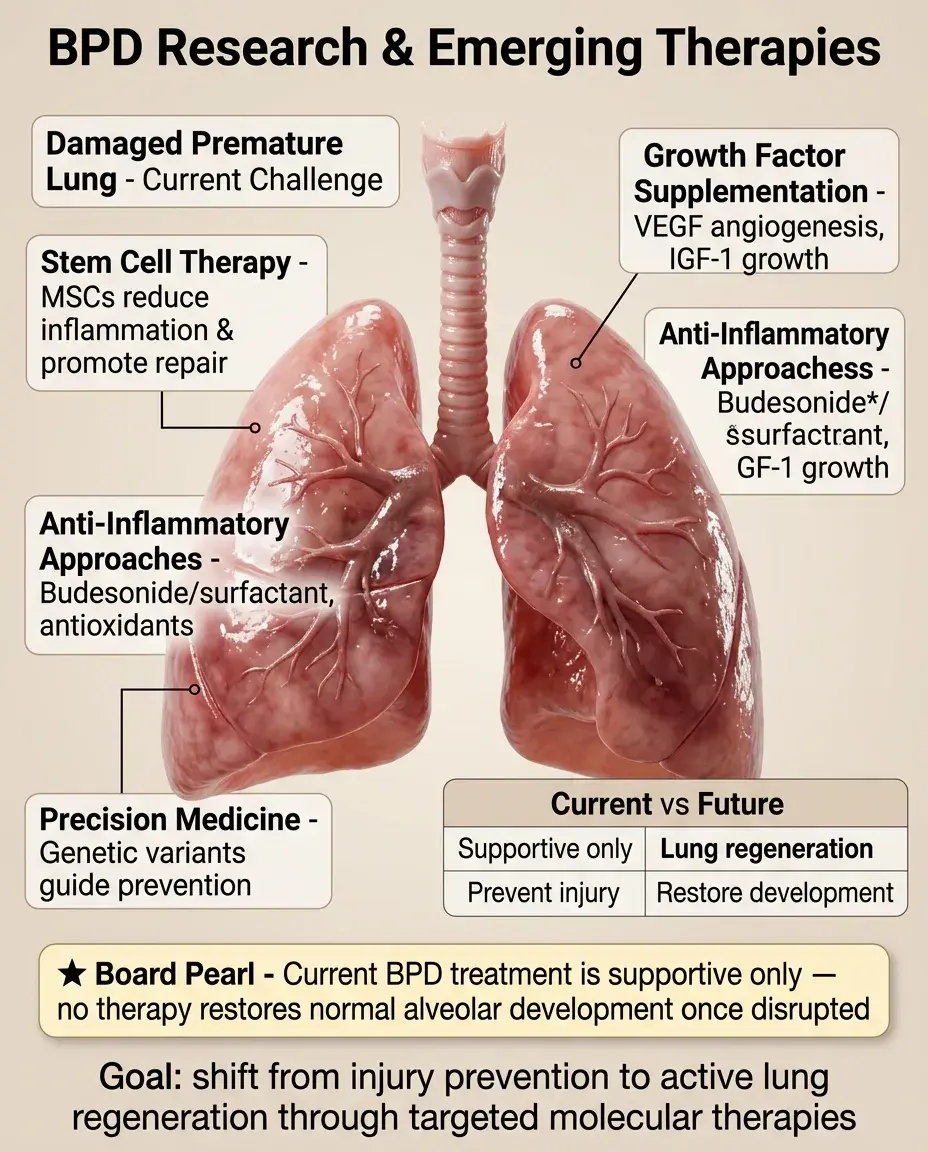
Research Directions and Emerging Therapies
⚡
Stem cell therapy: mesenchymal stem cells show promise in animal models for reducing inflammation and promoting lung repair.
⚡
Growth factors: VEGF supplementation to promote angiogenesis, IGF-1 to enhance growth.
⚡
Anti-inflammatory approaches: intratracheal budesonide/surfactant combination, antioxidant therapy.
⚡
Precision medicine: identifying genetic variants that increase BPD susceptibility to target prevention strategies.
⚡
Long-term goal: promote lung regeneration rather than just preventing injury.
⚡
Board pearl: Current BPD treatment is supportive — no therapy can restore normal alveolar development once disrupted.
Prognosis and Counseling Points
📌
Mortality: 10–15% for severe BPD in first year, usually from respiratory failure or pulmonary hypertension.
📌
Respiratory morbidity: 50% require rehospitalization in first year, usually for respiratory infections.
📌
Most infants wean off oxygen by 1–2 years, but pulmonary function abnormalities persist.
📌
Neurodevelopment: 30–50% have significant impairment, higher rates with severe BPD.
📌
Quality of life: most BPD survivors lead normal lives, but may have exercise limitations and increased respiratory symptoms.
📌
Board pearl: BPD prognosis correlates with severity at 36 weeks PMA — mild BPD has good outcomes, severe BPD has significant long-term morbidity.

Board Question Stem Patterns
📣
26-week premature infant still requiring oxygen at 30 days of life → BPD diagnosis established.
📣
Former premature infant with increased work of breathing during URI → low threshold for admission due to poor reserve.
📣
CXR showing heterogeneous lung fields with hyperinflation and atelectasis in ex-premie → classic BPD pattern.
📣
Premature infant with feeding difficulties and hepatomegaly → evaluate for pulmonary hypertension.
📣
Which intervention reduces BPD? → antenatal corticosteroids, surfactant, caffeine, gentle ventilation.
📣
Term infant with BPD-like disease → consider surfactant protein deficiency.
📣
BPD infant with RSV bronchiolitis → high risk for severe disease, ICU admission often needed.

One-Line Recap
🔸
Bronchopulmonary dysplasia is arrested lung development in premature infants exposed to mechanical ventilation and oxygen, characterized by alveolar simplification rather than fibrosis, requiring supportive care to minimize further injury while the lung slowly remodels, with long-term respiratory and neurodevelopmental sequelae proportional to disease severity at 36 weeks postmenstrual age.

bottom of page

