top of page
Reproductive & Endocrine Systems
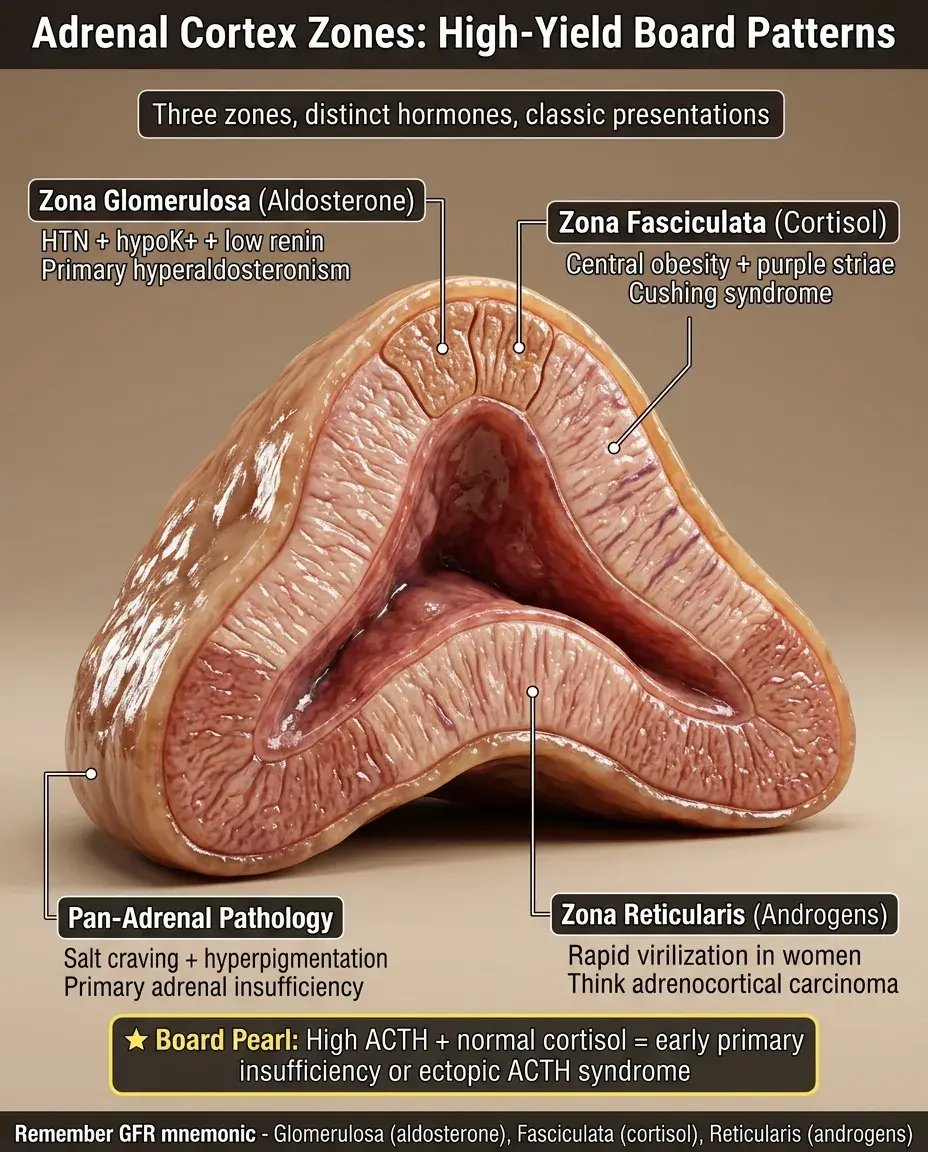
Adrenal cortex zones (glomerulosa, fasciculata, reticularis)
Core Principle of Adrenal Cortex Zonation
🧷
The adrenal cortex consists of three distinct histological zones, each producing specific steroid hormones through differential enzyme expression.
🧷
From superficial to deep: zona glomerulosa → zona fasciculata → zona reticularis, collectively comprising 80-90% of the adrenal gland's mass.
🧷
Each zone expresses a unique combination of steroidogenic enzymes that determines its hormonal output — aldosterone, cortisol, and androgens respectively.
🧷
This functional zonation reflects the differential regulation of each layer: glomerulosa by the renin-angiotensin system, fasciculata/reticularis by ACTH.
🧷
Board pearl: Zone-specific enzyme deficiencies produce predictable hormonal patterns based on which biosynthetic pathways remain intact.

Zona Glomerulosa: The Salt-Regulating Layer
📍
The outermost cortical zone, comprising ~15% of cortical volume, produces mineralocorticoids (primarily aldosterone).
📍
Contains aldosterone synthase (CYP11B2), the rate-limiting enzyme that catalyzes the final steps: corticosterone → 18-hydroxycorticosterone → aldosterone.
📍
Lacks 17α-hydroxylase, preventing synthesis of cortisol or sex hormones — this enzyme absence defines the zone's hormonal specificity.
📍
Primary regulation: angiotensin II stimulates aldosterone production; elevated K⁺ directly depolarizes glomerulosa cells.
📍
Board pearl: The glomerulosa is the only zone that can produce aldosterone and the only zone that cannot produce cortisol or androgens.

Zona Fasciculata: The Stress Response Layer
🔹
The middle and thickest zone (75% of cortex), characterized by lipid-rich cells arranged in parallel cords — the "fascicles" that give it its name.
🔹
Produces glucocorticoids (primarily cortisol) via 17α-hydroxylase and 21-hydroxylase pathways.
🔹
Contains abundant smooth ER and mitochondria with tubular cristae, reflecting high steroid synthesis capacity.
🔹
ACTH from the anterior pituitary drives cortisol production through cAMP-mediated upregulation of cholesterol uptake and steroidogenic enzymes.
🔹
Board pearl: Fasciculata atrophy occurs with chronic exogenous steroid use due to suppressed ACTH — the most common cause of adrenal insufficiency.

Zona Reticularis: The Androgen-Producing Layer
⭐
The innermost cortical zone, producing weak androgens: DHEA and androstenedione.
⭐
Expresses both 17α-hydroxylase and 17,20-lyase activity, enabling conversion of 17-hydroxypregnenolone → DHEA.
⭐
Relatively low expression of 3β-hydroxysteroid dehydrogenase favors the Δ5 pathway, producing DHEA over androstenedione.
⭐
Develops functionally at adrenarche (~age 6-8), marking the onset of adrenal androgen production before gonadal puberty.
⭐
Board pearl: In women, the reticularis produces 50% of circulating androgens — explaining virilization in adrenal tumors or 21-hydroxylase deficiency.

The Steroidogenic Cascade and Zone-Specific Enzymes
✅
All zones begin with cholesterol → pregnenolone via cholesterol desmolase (CYP11A1), the rate-limiting step of steroidogenesis.
✅
Glomerulosa pathway: pregnenolone → progesterone → 11-deoxycorticosterone → corticosterone → aldosterone.
✅
Fasciculata pathway: pregnenolone → 17-hydroxypregnenolone → 17-hydroxyprogesterone → 11-deoxycortisol → cortisol.
✅
Reticularis pathway: pregnenolone → 17-hydroxypregnenolone → DHEA (→ androstenedione).
✅
Board pearl: 17α-hydroxylase presence distinguishes fasciculata/reticularis from glomerulosa; its absence explains why glomerulosa cannot produce cortisol.

Functional Regulation: RAAS vs ACTH Control
🧠
Zona glomerulosa operates independently of pituitary control — regulated by renin-angiotensin-aldosterone system (RAAS), plasma K⁺, and to a lesser extent ACTH.
🧠
Zona fasciculata and reticularis are ACTH-dependent — chronic ACTH deficiency causes atrophy of these zones while sparing the glomerulosa.
🧠
This differential regulation explains why primary adrenal insufficiency affects all hormones while secondary (pituitary) insufficiency spares aldosterone.
🧠
Stress activates the HPA axis → ACTH release → cortisol production, while volume depletion activates RAAS → aldosterone.
🧠
Board pearl: Preserved aldosterone in secondary adrenal insufficiency means no hyperkalemia or volume depletion — key clinical distinction from primary.

Congenital Adrenal Hyperplasia and Zone Dysfunction
⚡
21-hydroxylase deficiency (90% of CAH): impaired synthesis in all zones → ↓cortisol, ↓aldosterone, ↑androgens from precursor shunting.
⚡
11β-hydroxylase deficiency: ↓cortisol, ↓aldosterone, but ↑11-deoxycorticosterone (weak mineralocorticoid) → hypertension with virilization.
⚡
17α-hydroxylase deficiency: ↓cortisol, ↓androgens, but ↑mineralocorticoids → hypertension, hypokalemia, sexual infantilism.
⚡
3β-hydroxysteroid dehydrogenase deficiency: the only CAH affecting all three pathways equally → salt wasting with ambiguous genitalia in males.
⚡
Board pearl: Enzyme location predicts the pattern — early pathway defects affect all zones; late defects show zone-specific effects.

Histological Features and Lipid Content
📌
Glomerulosa: small cells in rounded clusters beneath the capsule, moderate lipid content, round nuclei.
📌
Fasciculata: large, lipid-laden "clear cells" in parallel cords, abundant lipid droplets containing cholesterol esters — appear foamy on H&E.
📌
Reticularis: smaller, eosinophilic cells in anastomosing cords, less lipid content, occasional lipofuscin pigment.
📌
The lipid content inversely correlates with functional activity — active steroidogenesis depletes stored cholesterol.
📌
Board pearl: On histology, if asked to identify the zona fasciculata, look for the clear, foamy cells arranged in parallel columns.

Blood Supply and the Centripetal Flow Model
📣
Arterial supply: capsular arteries → subcapsular plexus → centripetal flow through cortical zones → medullary veins.
📣
This unidirectional flow creates a functional cascade: glomerulosa products can influence fasciculata/reticularis, but not vice versa.
📣
High cortisol concentrations from fasciculata bathe the medulla, inducing phenylethanolamine N-methyltransferase (PNMT) for epinephrine synthesis.
📣
The corticomedullary portal system ensures chromaffin cells are exposed to 100x higher cortisol than systemic circulation.
📣
Board pearl: Cortisol from fasciculata is required for adrenal medullary epinephrine production — explaining why Addison's patients have impaired epinephrine responses.

Primary vs Secondary Adrenal Insufficiency Patterns
🔸
Primary (Addison's): destruction of all cortical zones → ↓cortisol, ↓aldosterone, ↓androgens, with ↑ACTH causing hyperpigmentation.
🔸
Secondary (pituitary): ACTH deficiency → fasciculata/reticularis atrophy → ↓cortisol, ↓androgens, but preserved aldosterone (RAAS-regulated).
🔸
Laboratory distinction: Primary shows hyperkalemia, hyponatremia, metabolic acidosis; secondary shows only hyponatremia.
🔸
Clinical distinction: Primary presents with hyperpigmentation, salt craving, hypotension; secondary lacks these mineralocorticoid deficiency signs.
🔸
Board pearl: Normal potassium in adrenal insufficiency suggests secondary cause — the glomerulosa remains functional without ACTH.

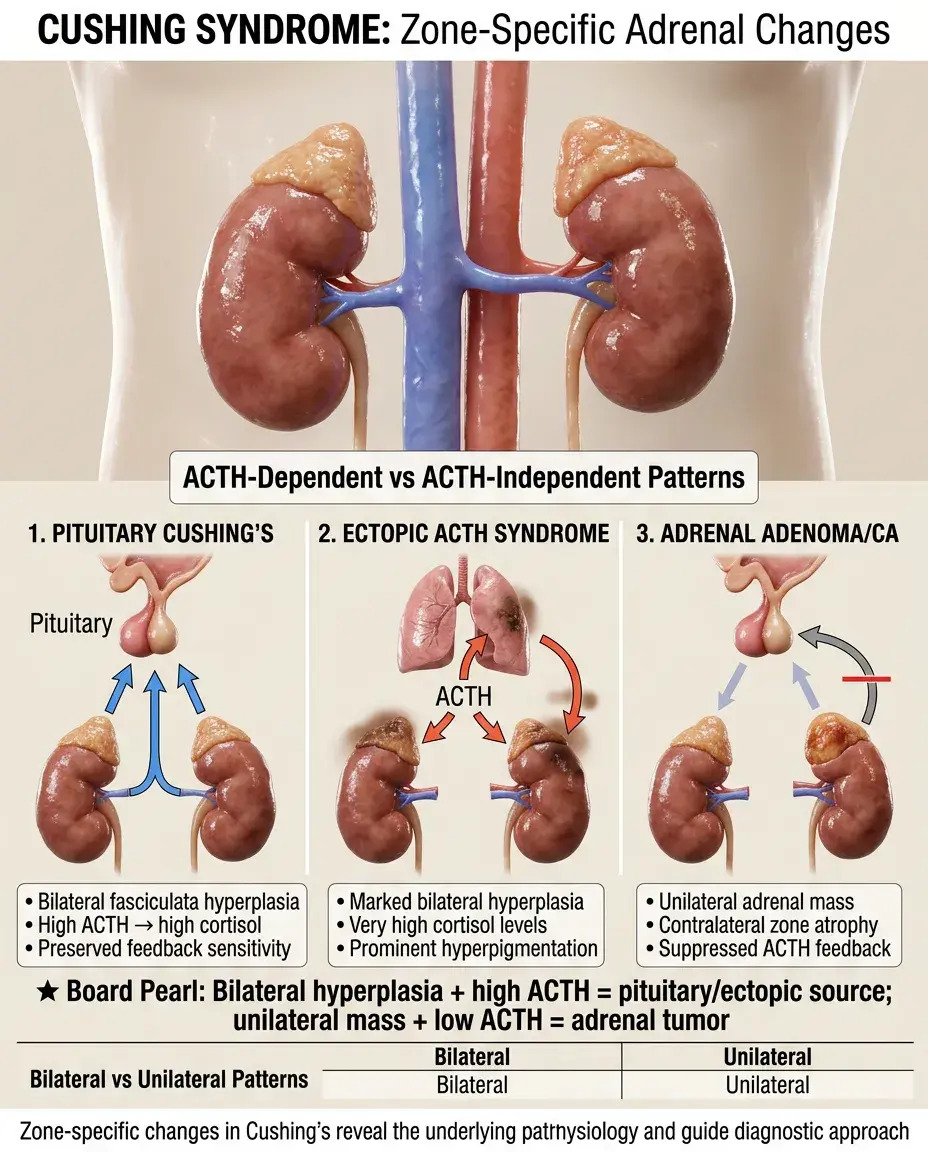
Cushing Syndrome and Zone-Specific Changes
🧷
ACTH-dependent Cushing's: bilateral fasciculata/reticularis hyperplasia with increased cortisol and androgens.
🧷
ACTH-independent Cushing's: adrenal adenoma/carcinoma with atrophy of non-tumorous zones due to feedback suppression.
🧷
Ectopic ACTH (small cell lung cancer): marked bilateral hyperplasia, very high cortisol, prominent hyperpigmentation.
🧷
Primary pigmented nodular adrenocortical disease: multiple small pigmented nodules throughout cortex, seen in Carney complex.
🧷
Board pearl: Bilateral adrenal hyperplasia with high ACTH indicates pituitary or ectopic source; unilateral mass with suppressed ACTH indicates adrenal tumor.
Hyperaldosteronism and Glomerulosa Pathology
📍
Primary hyperaldosteronism: autonomous aldosterone production from adenoma (Conn syndrome) or bilateral hyperplasia.
📍
Clinical triad: hypertension, hypokalemia, metabolic alkalosis with suppressed plasma renin activity.
📍
Aldosterone-producing adenomas typically arise from zona glomerulosa cells but may show hybrid features.
📍
Bilateral idiopathic hyperplasia affects only the glomerulosa, preserving normal fasciculata/reticularis architecture.
📍
Board pearl: Primary hyperaldosteronism shows high aldosterone with low renin; secondary shows high aldosterone with high renin.

Adrenarche and Zona Reticularis Development
🔹
Adrenarche marks functional maturation of zona reticularis around age 6-8, preceding gonadarche by several years.
🔹
Rising DHEA and DHEA-S levels stimulate early pubic and axillary hair growth before true puberty begins.
🔹
Premature adrenarche: early activation of reticularis before age 8 in girls, age 9 in boys — usually benign variant.
🔹
The reticularis continues expanding through puberty, peaks in the 20s-30s, then gradually involutes with aging (adrenopause).
🔹
Board pearl: Isolated early pubic hair without breast/testicular development suggests premature adrenarche, not precocious puberty.

Enzyme Deficiencies and Diagnostic Patterns
⭐
21-hydroxylase deficiency: ↑17-hydroxyprogesterone (diagnostic marker), ↓cortisol, ↓aldosterone, ↑androgens → virilization ± salt wasting.
⭐
11β-hydroxylase deficiency: ↑11-deoxycortisol, ↑11-deoxycorticosterone → hypertension with virilization.
⭐
17α-hydroxylase deficiency: ↑pregnenolone, ↑progesterone, ↑DOC → hypertension without virilization, males appear phenotypically female.
⭐
3β-HSD deficiency: ↑pregnenolone, ↑17-hydroxypregnenolone, ↑DHEA → only CAH with elevated DHEA.
⭐
Board pearl: The accumulated precursor before the enzyme block is the diagnostic marker — 17-OHP for 21-hydroxylase deficiency.

Stress Response and Zone Interactions
✅
Acute stress: ACTH surge → immediate cortisol release from fasciculata to mobilize glucose and suppress inflammation.
✅
Chronic stress: sustained ACTH → fasciculata hypertrophy, increased cortisol output, eventual HPA axis dysregulation.
✅
Severe illness can suppress reticularis function → decreased DHEA despite elevated cortisol (cortisol/DHEA ratio increases).
✅
Critical illness–related corticosteroid insufficiency: relative adrenal insufficiency despite "normal" cortisol levels.
✅
Board pearl: In severe stress, cortisol should be markedly elevated — a "normal" level may represent relative insufficiency.

Zona-Specific Tumors and Their Hormonal Profiles
🧠
Aldosteronoma: glomerulosa origin → isolated hyperaldosteronism without cortisol or androgen excess.
🧠
Cortisol-producing adenoma: fasciculata origin → pure Cushing syndrome without virilization, contralateral adrenal atrophy.
🧠
Adrenocortical carcinoma: often dedifferentiated → mixed hormone production (cortisol + androgens ± aldosterone).
🧠
Virilizing tumors: reticularis origin or carcinoma → marked androgen excess, rapid virilization in women.
🧠
Board pearl: Benign adenomas typically produce one hormone type; carcinomas often produce multiple hormones with predominant androgens.

Laboratory Evaluation of Zone Function
⚡
Glomerulosa: plasma aldosterone, plasma renin activity, aldosterone/renin ratio, saline suppression test.
⚡
Fasciculata: morning cortisol, ACTH, 24-hour urine free cortisol, dexamethasone suppression test, cosyntropin stimulation.
⚡
Reticularis: DHEA-S (more stable than DHEA), androstenedione, testosterone (in women).
⚡
Dynamic testing differentiates autonomous production from physiologic regulation.
⚡
Board pearl: DHEA-S is the best marker of adrenal androgen production — it has no gonadal source and a long half-life.

Clinical Manifestations by Zone
📌
Glomerulosa dysfunction: salt wasting (deficiency) → hypotension, hyperkalemia; excess → hypertension, hypokalemia, alkalosis.
📌
Fasciculata dysfunction: deficiency → fatigue, hypoglycemia, hypotension, eosinophilia; excess → central obesity, striae, osteoporosis.
📌
Reticularis dysfunction: deficiency → decreased axillary/pubic hair (women), low libido; excess → hirsutism, acne, virilization.
📌
Combined deficiencies suggest primary adrenal disease or pan-hypopituitarism.
📌
Board pearl: Female virilization (clitoromegaly, male-pattern balding, voice deepening) indicates severe androgen excess — think adrenal source.

Board Question Stem Patterns
📣
Hypertension + hypokalemia + low renin → primary hyperaldosteronism from glomerulosa pathology.
📣
Salt craving + hyperpigmentation + hypotension → primary adrenal insufficiency affecting all zones.
📣
Central obesity + purple striae + proximal weakness → Cushing syndrome from fasciculata hyperfunction.
📣
Female infant with ambiguous genitalia + salt wasting → 21-hydroxylase deficiency.
📣
Hypertension + hypokalemia + absent secondary sexual characteristics → 17α-hydroxylase deficiency.
📣
Normal cortisol with very high ACTH → consider ectopic ACTH or early primary insufficiency.
📣
Rapid virilization in adult woman → adrenocortical carcinoma until proven otherwise.
One-Line Recap
🔸
The adrenal cortex's three zones — glomerulosa (aldosterone/salt), fasciculata (cortisol/stress), and reticularis (androgens/sex) — each express specific enzymes that determine hormone production, with differential regulation (RAAS vs ACTH) explaining why mineralocorticoid function is preserved in secondary insufficiency and enzyme deficiency locations predict clinical syndromes.

bottom of page

