top of page
Multisystem Processes & Disorders
Acid–Base Classification and Compensation: Respiratory vs Metabolic
Core Principle of Acid-Base Physiology
🧷
Acid-base homeostasis maintains blood pH between 7.35-7.45 through a dynamic equilibrium: CO₂ + H₂O ⇌ H₂CO₃ ⇌ H⁺ + HCO₃⁻.
🧷
The respiratory system controls the volatile acid (CO₂) through alveolar ventilation, while the kidneys regulate the metabolic component (HCO₃⁻) through reabsorption and excretion.
🧷
Primary disorders arise from either respiratory dysfunction (altering PCO₂) or metabolic dysfunction (altering HCO₃⁻), with the opposite system providing compensation.
🧷
Board pearl: pH determines acidemia (<7.35) or alkalemia (>7.45), while PCO₂ and HCO₃⁻ identify the primary disorder.

The Henderson-Hasselbalch Equation Framework
📍
pH = 6.1 + log([HCO₃⁻]/0.03 × PCO₂) — this equation governs all acid-base physiology.
📍
The 20:1 ratio of HCO₃⁻ to dissolved CO₂ maintains normal pH; any deviation from this ratio shifts pH.
📍
Respiratory changes occur within minutes (rapid CO₂ equilibration across alveolar-capillary membrane), while metabolic compensation takes hours to days (renal HCO₃⁻ handling).
📍
Board clue: When pH and PCO₂ move in opposite directions, the primary disorder is respiratory. When they move in the same direction, the primary disorder is metabolic.

Respiratory Acidosis: Hypoventilation States
🔹
Respiratory acidosis results from decreased alveolar ventilation → CO₂ retention → increased H⁺ → pH < 7.35 with PCO₂ > 45 mmHg.
🔹
Acute causes: airway obstruction, CNS depression (opioids, sedatives), neuromuscular weakness (myasthenia crisis, Guillain-Barré), severe pneumonia.
🔹
Chronic causes: COPD, obesity hypoventilation syndrome, chronic neuromuscular disease.
🔹
Acute compensation: minimal buffering by proteins and phosphates.
🔹
Chronic compensation: kidneys increase HCO₃⁻ reabsorption — expect 3.5 mEq/L HCO₃⁻ rise per 10 mmHg chronic PCO₂ elevation.

Respiratory Alkalosis: Hyperventilation States
⭐
Respiratory alkalosis results from increased alveolar ventilation → CO₂ loss → decreased H⁺ → pH > 7.45 with PCO₂ < 35 mmHg.
⭐
Common causes: anxiety/panic, pain, hypoxemia (high altitude, pneumonia, PE), pregnancy (progesterone effect), salicylate toxicity (direct respiratory center stimulation), sepsis, liver failure.
⭐
Acute compensation: minimal — slight intracellular shift of H⁺.
⭐
Chronic compensation: kidneys decrease HCO₃⁻ reabsorption — expect 5 mEq/L HCO₃⁻ drop per 10 mmHg chronic PCO₂ decrease.
⭐
Board pearl: Hypoxemia-driven hyperventilation improves with oxygen; anxiety-driven does not.

Metabolic Acidosis: Loss of Base or Gain of Acid
✅
Metabolic acidosis presents with pH < 7.35 and HCO₃⁻ < 22 mEq/L, triggering respiratory compensation (hyperventilation) to lower PCO₂.
✅
Two categories based on anion gap: high AG (addition of unmeasured acid) vs normal AG (loss of HCO₃⁻ or impaired renal acid excretion).
✅
High AG causes: lactic acidosis, ketoacidosis (diabetic, alcoholic, starvation), uremia, toxic ingestions (methanol, ethylene glycol, salicylates).
✅
Normal AG causes: diarrhea (HCO₃⁻ loss), renal tubular acidosis, ureterosigmoidostomy.
✅
Board clue: Calculate anion gap immediately when metabolic acidosis is identified — it directs the differential diagnosis.

Metabolic Alkalosis: Gain of Base or Loss of Acid
🧠
Metabolic alkalosis presents with pH > 7.45 and HCO₃⁻ > 28 mEq/L, with compensatory hypoventilation raising PCO₂.
🧠
Generation requires initial HCO₃⁻ gain or H⁺ loss; maintenance requires impaired renal HCO₃⁻ excretion (usually volume depletion or hypokalemia).
🧠
Volume-responsive causes: vomiting (HCl loss), diuretics, post-hypercapnia.
🧠
Volume-resistant causes: hyperaldosteronism, Cushing syndrome, severe hypokalemia.
🧠
Board distinction: Urine chloride <20 mEq/L suggests volume-responsive; >20 mEq/L suggests volume-resistant alkalosis.

Winter's Formula: Predicting Respiratory Compensation
⚡
In metabolic acidosis, expected PCO₂ = 1.5 × [HCO₃⁻] + 8 (±2).
⚡
This formula predicts appropriate respiratory compensation — if actual PCO₂ differs significantly, a mixed disorder exists.
⚡
Example: HCO₃⁻ = 10 → expected PCO₂ = 23 (±2). If actual PCO₂ = 35, concurrent respiratory acidosis is present.
⚡
Respiratory compensation begins within 30 minutes and maximizes within 12-24 hours.
⚡
Board pearl: PCO₂ levels outside Winter's predicted range indicate a second primary disorder, not just incomplete compensation.

Compensation Patterns and Limits
📌
Compensation never fully normalizes pH — it only blunts the primary disorder's effect.
📌
Respiratory compensation for metabolic disorders: rapid onset (minutes to hours), limited by respiratory muscle fatigue and hypoxia/hypercapnia drives.
📌
Metabolic compensation for respiratory disorders: slow onset (hours to days), more effective for chronic than acute disorders.
📌
Maximum compensation: respiratory alkalosis can lower HCO₃⁻ to ~12-15; metabolic alkalosis rarely raises PCO₂ above 55-60 (limited by hypoxia).
📌
Board clue: "Overcompensation" does not exist — normal pH with abnormal PCO₂ and HCO₃⁻ indicates a mixed disorder.

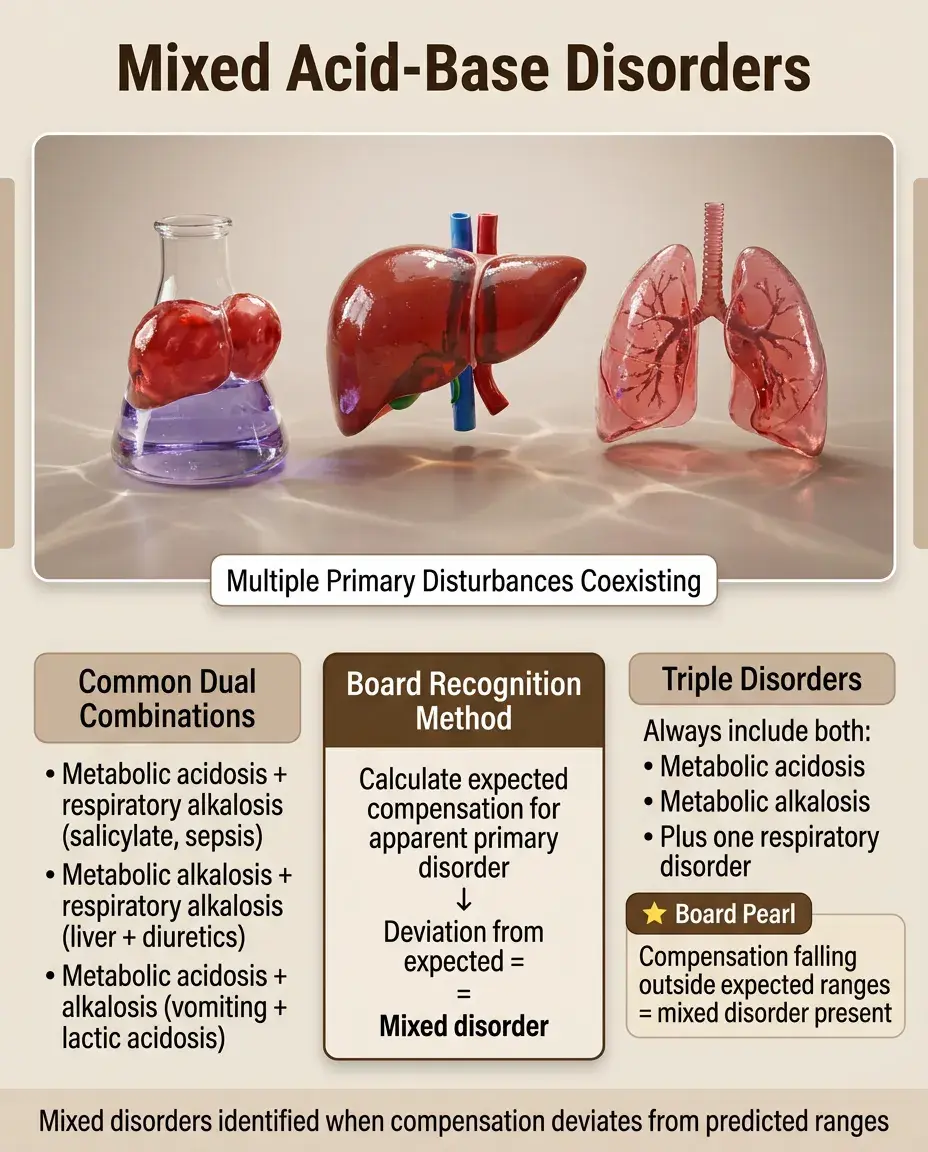
Mixed Acid-Base Disorders
📣
Mixed disorders occur when two or more primary disturbances coexist, identified by compensation falling outside expected ranges.
📣
Common combinations: metabolic acidosis + respiratory alkalosis (salicylate toxicity, sepsis), metabolic alkalosis + respiratory alkalosis (liver disease with diuretics), metabolic acidosis + metabolic alkalosis (vomiting with lactic acidosis).
📣
Triple disorders can occur but always include metabolic acidosis + metabolic alkalosis + either respiratory disorder.
📣
Board approach: Calculate expected compensation for the apparent primary disorder — deviation suggests a mixed disorder.
The Delta-Delta Calculation
🔸
Used specifically in high anion gap metabolic acidosis to detect concurrent metabolic alkalosis or normal AG acidosis.
🔸
ΔAG = measured AG - normal AG (typically 12); ΔHCO₃⁻ = normal HCO₃⁻ (24) - measured HCO₃⁻.
🔸
In pure high AG acidosis, ΔAG ≈ ΔHCO₃⁻ (1:1 relationship).
🔸
If ΔAG > ΔHCO₃⁻ → concurrent metabolic alkalosis (HCO₃⁻ higher than expected).
🔸
If ΔAG < ΔHCO₃⁻ → concurrent normal AG acidosis (HCO₃⁻ lower than expected).
🔸
Board example: AG = 20, HCO₃⁻ = 20. ΔAG = 8, ΔHCO₃⁻ = 4 → mixed high AG acidosis and metabolic alkalosis.

Clinical Manifestations by Disorder
🧷
Respiratory acidosis: confusion, somnolence, asterixis (CO₂ narcosis), papilledema (cerebral vasodilation).
🧷
Respiratory alkalosis: perioral/finger paresthesias, tetany, seizures (decreased ionized Ca²⁺ from albumin binding).
🧷
Metabolic acidosis: Kussmaul respirations (deep, rapid breathing), hypotension (decreased cardiac contractility), hyperkalemia (H⁺/K⁺ exchange).
🧷
Metabolic alkalosis: weakness, polyuria (K⁺ depletion), arrhythmias, altered mental status.
🧷
Board pearl: Tetany in alkalosis results from decreased free Ca²⁺ despite normal total calcium — alkalosis increases Ca²⁺-albumin binding.

Special Scenario: Salicylate Toxicity
📍
Salicylates directly stimulate the respiratory center → primary respiratory alkalosis.
📍
Simultaneously uncouple oxidative phosphorylation → increased oxygen consumption, heat production, and metabolic acidosis.
📍
Classic finding: mixed respiratory alkalosis and high AG metabolic acidosis with relatively normal pH.
📍
Additional features: tinnitus, altered mental status, hyperthermia, hypoglycemia (CNS glucose depletion).
📍
Board clue: Adult with tinnitus, hyperventilation, and AG = 20 with pH 7.42 → salicylate toxicity until proven otherwise.

Altitude and Acid-Base Adaptation
🔹
Acute altitude exposure → hypoxemia → hyperventilation → respiratory alkalosis.
🔹
Renal compensation over 2-3 days: decreased HCO₃⁻ reabsorption normalizes pH.
🔹
Chronic altitude dwellers maintain mild respiratory alkalosis with compensatory low HCO₃⁻.
🔹
Return to sea level → relative hyperoxia → decreased ventilatory drive → transient respiratory acidosis until readaptation.
🔹
Board application: Recent arrival at altitude with pH 7.48, PCO₂ 28, HCO₃⁻ 20 represents partial compensation — full acclimatization takes days.

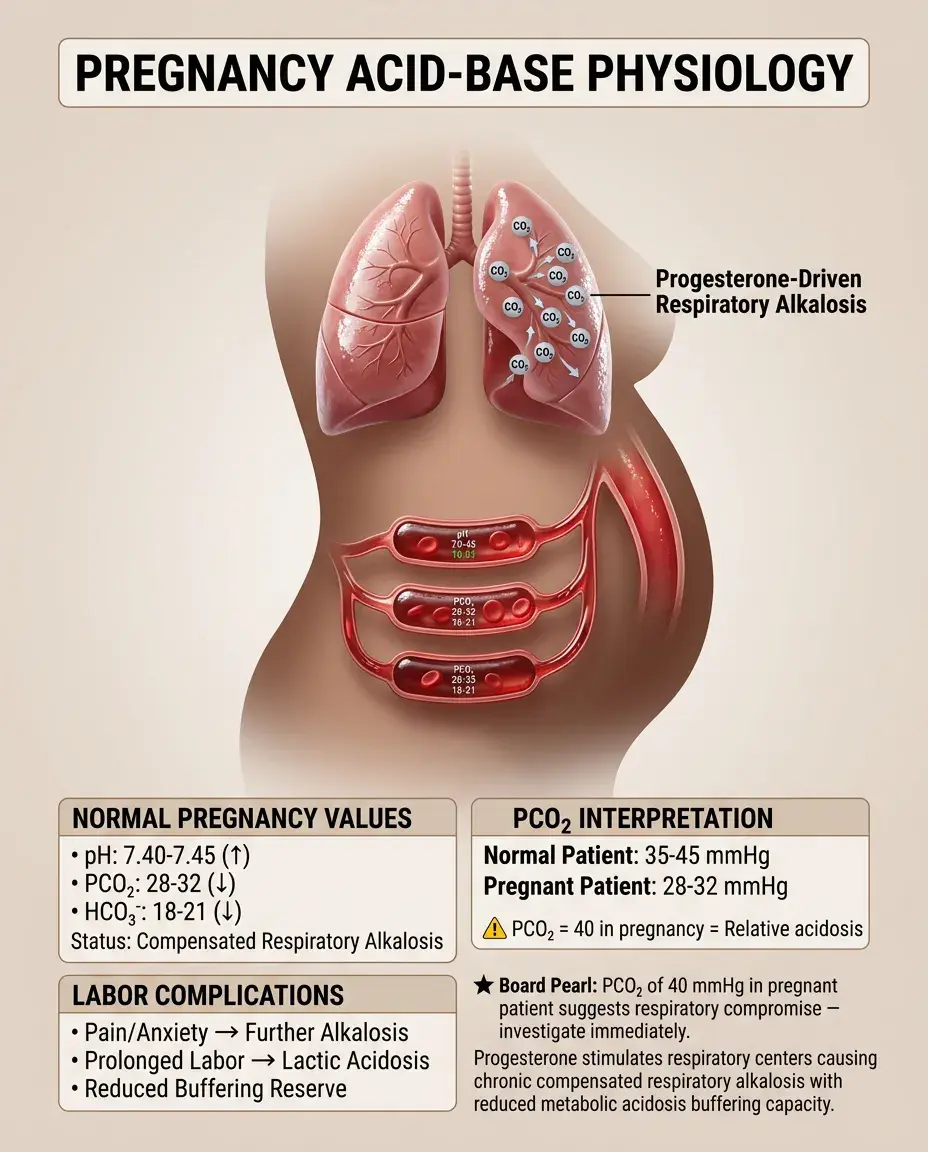
Pregnancy and Normal Acid-Base Changes
⭐
Progesterone stimulates respiratory centers → chronic respiratory alkalosis throughout pregnancy.
⭐
Normal pregnancy values: pH 7.40-7.45, PCO₂ 28-32, HCO₃⁻ 18-21 (compensated respiratory alkalosis).
⭐
This baseline shift means pregnant patients have less buffering reserve for metabolic acidosis.
⭐
Labor superimposes acute changes: pain/anxiety (further respiratory alkalosis) or prolonged labor (lactic acidosis).
⭐
Board pearl: A PCO₂ of 40 in a pregnant patient represents relative respiratory acidosis — investigate for respiratory compromise.
Diagnostic Approach: The Five-Step Method
✅
Step 1: Assess pH — acidemia or alkalemia?
✅
Step 2: Determine primary disorder — which parameter (PCO₂ or HCO₃⁻) explains the pH change?
✅
Step 3: Calculate compensation — is it appropriate for a simple disorder?
✅
Step 4: If metabolic acidosis, calculate anion gap and assess for mixed disorders.
✅
Step 5: Develop differential diagnosis based on clinical context and laboratory pattern.
✅
Board strategy: Work systematically — jumping to diagnosis without checking compensation misses mixed disorders.

Pitfalls in Interpretation
🧠
Pseudorespiratory alkalosis: delayed sample processing allows CO₂ loss → falsely low PCO₂. Always use fresh samples.
🧠
Temperature effects: hypothermia decreases CO₂ production and increases solubility — correct values for patient temperature.
🧠
Spurious lab values: check that pH, PCO₂, and HCO₃⁻ follow Henderson-Hasselbalch — if not, lab error likely.
🧠
Hidden metabolic alkalosis: may only manifest when treating metabolic acidosis (HCO₃⁻ rises more than expected).
🧠
Board warning: Post-cardiac arrest pH may not reflect tissue acidosis due to poor perfusion — venous blood better reflects tissue status.

Compensation Timing and Clinical Correlations
⚡
Respiratory compensation for metabolic disorders: begins within 30 minutes, maximal at 12-24 hours.
⚡
Metabolic compensation for respiratory disorders: begins at 6-12 hours, maximal at 3-5 days.
⚡
This timing helps distinguish acute from chronic disorders — insufficient compensation suggests acute process.
⚡
Example: COPD exacerbation shows acute-on-chronic respiratory acidosis — PCO₂ rises further but HCO₃⁻ hasn't had time to increase proportionally.
⚡
Board application: pH 7.25, PCO₂ 60, HCO₃⁻ 26 suggests acute respiratory acidosis; same PCO₂ with HCO₃⁻ 34 suggests chronic.

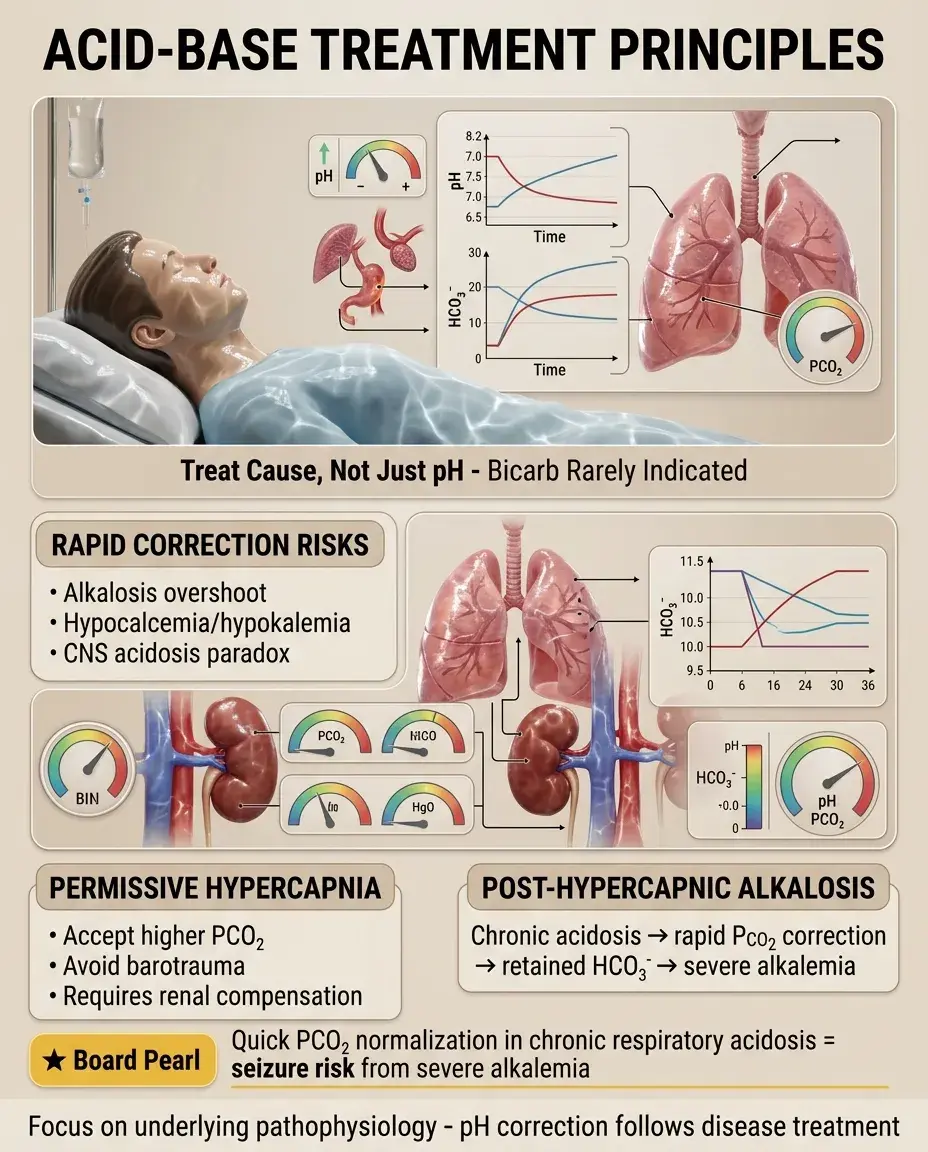
Treatment Principles and Acid-Base Goals
📌
Treat underlying cause, not just pH — bicarbonate therapy rarely indicated except severe acidemia (pH < 7.1) with hemodynamic compromise.
📌
Rapid correction risks: alkalosis overshoot, hypocalcemia, hypokalemia, paradoxical CNS acidosis (CO₂ crosses blood-brain barrier faster than HCO₃⁻).
📌
Permissive hypercapnia: accepting higher PCO₂ in mechanical ventilation to avoid barotrauma — requires intact renal compensation.
📌
Post-hypercapnic alkalosis: occurs when chronic respiratory acidosis rapidly corrected — retained HCO₃⁻ causes metabolic alkalosis.
📌
Board concept: Normalizing PCO₂ too quickly in chronic respiratory acidosis → severe alkalemia → seizures.
Board Question Stem Patterns
📣
pH 7.30, PCO₂ 50, HCO₃⁻ 24 → acute respiratory acidosis (no metabolic compensation yet).
📣
pH 7.35, PCO₂ 60, HCO₃⁻ 32 → chronic compensated respiratory acidosis.
📣
pH 7.50, PCO₂ 48, HCO₃⁻ 36 → metabolic alkalosis with appropriate compensation.
📣
pH 7.40, PCO₂ 20, HCO₃⁻ 12 → mixed disorder (respiratory alkalosis + metabolic acidosis).
📣
Post-operative patient with NG suction and pH 7.52 → metabolic alkalosis from gastric acid loss.
📣
COPD patient on diuretics with pH 7.45, PCO₂ 60, HCO₃⁻ 40 → mixed respiratory acidosis + metabolic alkalosis.
📣
Marathon runner with pH 7.32, PCO₂ 32 → metabolic acidosis with appropriate respiratory compensation.

One-Line Recap
🔸
Acid-base disorders divide into respiratory (PCO₂) and metabolic (HCO₃⁻) derangements with predictable compensation patterns, where pH determines acidemia vs alkalemia, the non-compensating parameter identifies the primary disorder, and deviations from expected compensation reveal mixed disorders requiring systematic analysis through anion gap calculation and clinical correlation.

bottom of page

